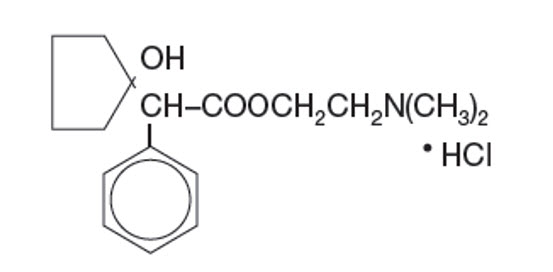
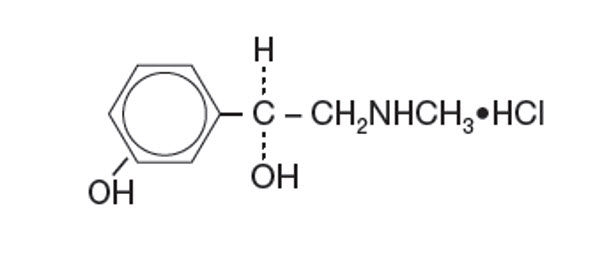
Get your patient on Cyclomydril - Cyclopentolate Hydrochloride And Phenylephrine Hydrochloride solution/ Drops (Cyclopentolate Hydrochloride And Phenylephrine Hydrochloride)
Cyclomydril - Cyclopentolate Hydrochloride And Phenylephrine Hydrochloride solution/ Drops prescribing information
Check medication interactionsReview interactions as part of your prescribing workflow
Cyclomydril - Cyclopentolate Hydrochloride And Phenylephrine Hydrochloride solution/ Drops PubMed™ news
5 most recent articles for Cyclomydril - Cyclopentolate Hydrochloride And Phenylephrine Hydrochloride solution/ Drops, view all articles on PubMed™
Show the latest PubMed™ articles for Cyclomydril - Cyclopentolate Hydrochloride And Phenylephrine Hydrochloride solution/ Drops