Get your patient on Cupric Chloride
Cupric Chloride prescribing information
INDICATIONS AND USAGE
Cupric chloride injection is indicated for use as a supplement to intravenous solutions given for TPN. Administration helps to maintain copper serum levels and to prevent depletion of endogenous stores and subsequent deficiency symptoms.
DOSAGE AND ADMINISTRATION
- Cupric chloride injection contains 0.4 mg of copper/mL and is administered intravenously only after dilution. The additive should be diluted in a volume of fluid not less than 100 mL.
- For the adult receiving TPN, the suggested additive dosage of copper is 0.3 mg/day to 0.5 mg/day.
- For pediatric patients, the suggested additive dosage of copper is 20 mcg/kg/day (0.05 mL/kg/day) up to a maximum of 500 mcg/day.
- This product is not appropriate for patients weighing less than 4 kg due to the inability to measure the appropriate amount of the product.
- Do not administer cupric chloride injection intramuscularly because the acidic pH of the solution may cause considerable tissue irritation.
Cupric chloride injection should only be used in conjunction with a pharmacy directed admixture program using aseptic technique in a laminar flow environment; it should be used promptly and in a single operation without any repeated penetrations. The solution contains no preservatives; discard the unused portion immediately after the admixture procedure is completed.
Cupric chloride injection should be inspected visually for particulate matter and discoloration prior to administration. Do not use unless the solution is clear and the seal is intact.
Cupric ion may degrade ascorbic acid in TPN solutions. In order to avoid this loss of ascorbate, multivitamin additives should be added to TPN solutions immediately prior to infusion. Alternatively, the multivitamin additive may be added to one container of TPN solution, followed by copper in a subsequent container.
CONTRAINDICATIONS
Cupric chloride injection is contraindicated in patients with hypersensitivity to copper (see WARNINGS: Hypersensitivity Reactions ).
ADVERSE REACTIONS
None known.
DESCRIPTION
Cupric chloride injection USP, 0.4 mg/mL is a sterile, clear, colorless to light blue color, nonpyrogenic solution intended for use as an additive to intravenous solutions for total parenteral nutrition (TPN). Each mL of solution contains 1.07 mg cupric chloride dihydrate; 9 mg sodium chloride and water for injection.
The solution contains no bacteriostat, antimicrobial agent or added buffer. The pH is 2.0 (1.5 to 2.5); product may contain hydrochloric acid and sodium hydroxide for pH adjustment. The osmolarity is 0.327 mOsmol/mL (calc.).
Cupric chloride, USP is chemically designated cupric chloride, dihydrate. Its molecular weight is 170.48 g/mol.
The molecular formula is CuCl 2 • 2H 2 O and the structural formula is:
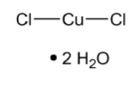
Cupric chloride, USP is a blue to blue-green crystalline compound freely soluble in water, soluble in alcohol and slightly soluble in ether.
Sodium chloride, USP is chemically designated NaCl, a white crystalline compound freely soluble in water.
The semi-rigid vial is fabricated from a specially formulated polyolefin. It is a copolymer of ethylene and propylene. The small amount of water vapor that can pass through the plastic container wall will not significantly alter the drug concentration.
CLINICAL PHARMACOLOGY
Copper is an essential nutrient which serves as a cofactor for serum ceruloplasmin, an oxidase necessary for proper formation of the iron carrier protein, transferrin. Copper also helps maintain normal rates of red and white blood cell formation.
Providing copper during TPN helps prevent development of the following deficiency symptoms: Leukopenia, neutropenia, anemia, depressed ceruloplasmin levels, impaired transferrin formation, secondary iron deficiency and osteoporosis.
Normal serum copper values range from 80 mcg/dl to 163 mcg/dl (mean, approximately 110 mcg/dl). The serum copper level at which deficiency symptoms appear is not precisely defined. In the plasma, about 7% of copper is bound to albumin and amino acids. In the liver, about 93% of copper is bound to ceruloplasmin and released to the serum. The daily turnover of copper through ceruloplasmin is approximately 0.5 mg. Copper is primarily excreted through the bile and into the gastrointestinal tract where it is not reabsorbed. Copper is also eliminated through the kidneys.
HOW SUPPLIED
Cupric Chloride Injection USP, 0.4 mg/mL is available as sterile, clear, colorless to light blue color solution filled in 10 mL polypropylene vial with gray rubber stopper and orange flip-off seal.
It is supplied as follows:
4 mg/10 mL (0.4 mg/mL)
1 Single-Dose Vial: NDC 80830-2301-1
25 Vials in 1 Carton: NDC 80830-2301-5
Store at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature].
Manufactured by:
Amneal Pharmaceuticals Pvt. Ltd.
Ahmedabad 382110, INDIA
Distributed by:
Amneal Pharmaceuticals LLC
Bridgewater, NJ 08807
Rev. 10-2025-03