Get your patient on Cromolyn Sodium Oral Solution (concentrate)- Cromolyn Sodium Oral Solution (concentrate) solution, Concentrate (Cromolyn Sodium Oral Solution (Concentrate))
Cromolyn Sodium Oral Solution (Concentrate)- Cromolyn Sodium Oral Solution (Concentrate) solution, Concentrate prescribing information
Cromolyn Sodium Oral Solution (Concentrate) is indicated in the management of patients with mastocytosis. Use of this product has been associated with improvement in diarrhea, flushing, headaches, vomiting, urticaria, abdominal pain, nausea, and itching in some patients.
NOT FOR INHALATION OR INJECTION. SEE DIRECTIONS FOR USE.
The usual starting dose is as follows:
Adults and Adolescents (13 Years and Older): Two ampules four times daily, taken one-half hour before meals and at bedtime.
Children 2-12 Years: One ampule four times daily, taken one-half hour before meals and at bedtime.
Pediatric Patients Under 2 Years: Not recommended.
If satisfactory control of symptoms is not achieved within two to three weeks, the dosage may be increased but should not exceed 40 mg/kg/day. Patients should be advised that the effect of Cromolyn Sodium Oral Solution (Concentrate) therapy is dependent upon its administration at regular intervals, as directed.
Maintenance Dose: Once a therapeutic response has been achieved, the dose may be reduced to the minimum required to maintain the patient with a lower degree of symptomatology. To prevent relapses, the dosage should be maintained.
Administration: Cromolyn Sodium Oral Solution (Concentrate) should be administered as a solution at least 1/2 hour before meals and at bedtime after preparation according to the following directions:
1. Break open ampule(s) and squeeze liquid contents of ampule(s) into a glass of water. 2. Stir solution. 3. Drink all of the liquid.
Cromolyn Sodium Oral Solution (Concentrate) is contraindicated in those patients who have shown hypersensitivity to cromolyn sodium.
Most of the adverse events reported in mastocytosis patients have been transient and could represent symptoms of the disease. The most frequently reported adverse events in mastocytosis patients who have received Cromolyn Sodium Oral Solution (Concentrate) during clinical studies were headache and diarrhea, each of which occurred in 4 of the 87 patients. Pruritus, nausea, and myalgia were each reported in 3 patients and abdominal pain, rash, and irritability in 2 patients each. One report of malaise was also recorded.
To report SUSPECTED ADVERSE REACTIONS, contact Ritedose Pharmaceuticals, LLC at 1-855-806-3300 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Other Adverse Events: Additional adverse events have been reported during studies in other clinical conditions and from worldwide postmarketing experience. In most cases the available information is incomplete and attribution to the drug cannot be determined. The majority of these reports involve the gastrointestinal system and include: diarrhea, nausea, abdominal pain, constipation, dyspepsia, flatulence, glossitis, stomatitis, vomiting, dysphagia, esophagospasm.
Other less commonly reported events (the majority representing only a single report) include the following:
| Skin: | pruritus, rash, urticaria/angioedema, erythema/ burning, photosensitivity |
| Musculoskeletal: | arthralgia, myalgia, stiffness/weakness of legs |
| Neurologic: | headache, dizziness, hypoesthesia, paresthesia, migraine, convulsions, flushing |
| Psychiatric: | psychosis, anxiety, depression, hallucinations, behavior change, insomnia, nervousness |
| Heart Rate: | tachycardia, premature ventricular contractions (PVCs), palpitations |
| Respiratory: | pharyngitis, dyspnea |
| Miscellaneous: | fatigue, edema, unpleasant taste, chest pain, postprandial lightheadedness and lethargy, dysuria, urinary frequency, purpura, hepatic function test abnormal, polycythemia, neutropenia, pancytopenia, tinnitus, lupus erythematosus (LE) syndrome |
Each 5 mL ampule of Cromolyn Sodium Oral Solution (Concentrate) contains 100 mg cromolyn sodium, USP, in purified water. Cromolyn sodium is a hygroscopic, white powder having little odor. It may leave a slightly bitter aftertaste. Cromolyn Sodium Oral Solution (Concentrate) is clear, colorless, and sterile. It is intended for oral use.
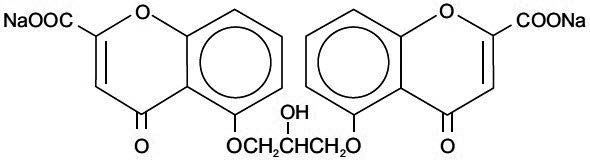
Chemically, cromolyn sodium is disodium 5,5’-[(2-hydroxy-trimethylene) dioxy]bis[4-oxo-4H-1-benzopyran-2-carboxylate]. The empirical formula is C 23 H 14 Na 2 O 11 ; the molecular weight is 512.34. Its chemical structure is:
Pharmacologic Category: Mast cell stabilizer
Therapeutic Category: Antiallergic
In vitro and in vivo animal studies have shown that cromolyn sodium inhibits the release of mediators from sensitized mast cells. Cromolyn sodium acts by inhibiting the release of histamine and leukotrienes (SRS-A) from the mast cell.
Cromolyn sodium has no intrinsic vasoconstrictor, antihistamine, or glucocorticoid activity.
Cromolyn sodium is poorly absorbed from the gastrointestinal tract. No more than 1% of an administered dose is absorbed by humans after oral administration, the remainder being excreted in the feces. Very little absorption of cromolyn sodium was seen after oral administration of 500 mg by mouth to each of 12 volunteers. From 0.28 to 0.50% of the administered dose was recovered in the first 24 hours of urinary excretion in 3 subjects. The mean urinary excretion of an administered dose over 24 hours in the remaining 9 subjects was 0.45%.
Four randomized, controlled clinical trials were conducted with Cromolyn Sodium Oral Solution (Concentrate) in patients with either cutaneous or systemic mastocytosis; two of which utilized a placebo-controlled crossover design, one utilized an active-controlled (chlorpheniramine plus cimetidine) crossover design, and one utilized a placebo-controlled parallel group design. Due to the rare nature of this disease, only 36 patients qualified for study entry, of whom 32 were considered evaluable. Consequently, formal statistical analyses were not performed. Clinically significant improvement in gastrointestinal symptoms (diarrhea, abdominal pain) were seen in the majority of patients with some improvement also seen for cutaneous manifestations (urticaria, pruritus, flushing) and cognitive function. The benefit seen with Cromolyn Sodium Oral Solution (Concentrate) 200 mg QID was similar to chlorpheniramine (4 mg QID) plus cimetidine (300 mg QID) for both cutaneous and systemic symptoms of mastocytosis.
Clinical improvement occurred within 2-6 weeks of treatment initiation and persisted for 2-3 weeks after treatment withdrawal. Cromolyn Sodium Oral Solution (Concentrate) did not affect urinary histamine levels or peripheral eosinophilia, although neither of these variables appeared to correlate with disease severity. Positive clinical benefits were also reported for 37 of 51 patients who received Cromolyn Sodium Oral Solution (Concentrate) in United States and foreign humanitarian programs.
Cromolyn Sodium Oral Solution (Concentrate) is an unpreserved, colorless solution supplied in a low density polyethylene plastic unit dose ampule with 8 ampules per foil pouch.
Each 5 mL ampule contains 100 mg cromolyn sodium, USP, in purified water.
NDC 76204-025-96 96 ampules x 5 mL (12 pouches x 8 ampules)
Cromolyn Sodium Oral Solution (Concentrate) should be stored between 20° – 25°C (68° – 77°F) [see USP Controlled Room Temperature] and protected from light. Do not use if it contains a precipitate or becomes discolored. Keep out of the reach of children.
Store ampules in foil pouch until ready for use.
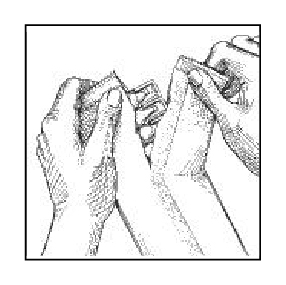
1. Open foil pouch by tearing at serrated edge as shown.
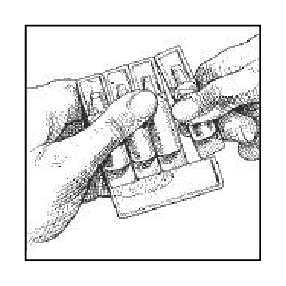
2. Remove ampule(s) from the strip.
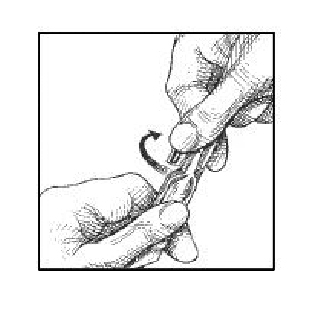
3. Open the ampule by twisting off the tabbed top section
4. Squeeze liquid contents into a glass of water. Stir Solution. Drink all of the liquid. Discard the empty ampule.