Cromolyn Sodium - Cromolyn Sodium solution/ Drops prescribing information
INDICATIONS AND USAGE
Cromolyn Sodium Ophthalmic Solution USP, 4% is indicated in the treatment of vernal keratoconjunctivitis, vernal conjunctivitis, and vernal keratitis.
DOSAGE AND ADMINISTRATION
The dose is 1 or 2 drops in each eye 4 to 6 times a day at regular intervals. One drop contains approximately 1.6 mg cromolyn sodium.
Patients should be advised that the effect of cromolyn sodium ophthalmic solution therapy is dependent upon its administration at regular intervals, as directed.
Symptomatic response to therapy (decreased itching, tearing, redness, and discharge) is usually evident within a few days, but longer treatment for up to six weeks is sometimes required. Once symptomatic improvement has been established, therapy should be continued for as long as needed to sustain improvement.
If required, corticosteroids may be used concomitantly with Cromolyn Sodium Ophthalmic Solution USP, 4%.
CONTRAINDICATIONS
Cromolyn Sodium Ophthalmic Solution USP, 4% is contraindicated in those patients who have shown hypersensitivity to cromolyn sodium or to any of the other ingredients.
ADVERSE REACTIONS
The most frequently reported adverse reaction attributed to the use of cromolyn sodium ophthalmic solution, on the basis of reoccurrence following readministration, is transient ocular stinging or burning upon instillation.
The following adverse reactions have been reported as infrequent events. It is unclear whether they are attributable to the drug:
Conjunctival injection; watery eyes; itchy eyes; dryness around the eye; puffy eyes; eye irritation; and styes.
Immediate hypersensitivity reactions have been reported rarely and include dyspnea, edema and rash.
DESCRIPTION
Cromolyn Sodium Ophthalmic Solution USP, 4% is a clear, colorless, sterile solution intended for topical ophthalmic use.
Cromolyn sodium is represented by the following structural formula:
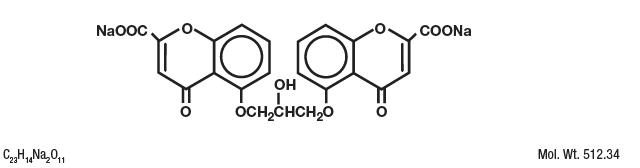
Chemical Name: Disodium 5-5'-[(2-hydroxytrimethylene)dioxy]bis[4-oxo-4H-1-benzopyran-2-carboxylate]
Pharmacologic Category: Mast cell stabilizer.
EACH mL CONTAINS: Active: cromolyn sodium 40 mg (4%); Preservative: benzalkonium chloride 0.01%. Inactives: edetate disodium 0.1% and purified water. Hydrochloric acid and/or sodium hydroxide may be added to adjust pH (4.0-7.0).
CLINICAL PHARMACOLOGY
In vitro and in vivo animal studies have shown that cromolyn sodium inhibits the degranulation of sensitized mast cells which occurs after exposure to specific antigens. Cromolyn sodium acts by inhibiting the release of histamine and SRS-A (slow-reacting substance of anaphylaxis) from the mast cell.
Another activity demonstrated in vitro is the capacity of cromolyn sodium to inhibit the degranulation of non-sensitized rat mast cells by phospholipase A and the subsequent release of chemical mediators. Another study showed that cromolyn sodium did not inhibit the enzymatic activity of released phospholipase A on its specific substrate.
Cromolyn sodium has no intrinsic vasoconstrictor, antihistaminic or anti-inflammatory activity.
Cromolyn sodium is poorly absorbed. When multiple doses of cromolyn sodium ophthalmic solution are instilled into normal rabbit eyes, less than 0.07% of the administered dose of cromolyn sodium is absorbed into the systemic circulation (presumably by way of the eye, nasal passages, buccal cavity and gastrointestinal tract). Trace amounts (less than 0.01%) of the cromolyn sodium dose penetrate into the aqueous humor and clearance from this chamber is virtually complete within 24 hours after treatment is stopped.
In normal volunteers, analysis of drug excretion indicates that approximately 0.03% of cromolyn sodium is absorbed following administration to the eye.
HOW SUPPLIED
Cromolyn Sodium Ophthalmic Solution USP, 4% is supplied in a white, opaque, plastic ophthalmic dispenser in the following size:
10 mL NDC 61314-237-10
Storage: Store between 15° - 30°C (59°- 86°F). Protect from light-store in original carton. Keep tightly closed and out of the reach of children.
Rx Only