Clorazepate Dipotassium - Clorazepate Dipotassium tablet prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; and DEPENDENCE AND WITHDRAWAL REACTIONS
- Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of these drugs in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation (See WARNINGS and PRECAUTIONS ).
- The use of benzodiazepines, including Clorazepate dipotassium tablets, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing Clorazepate dipotassium tablets and throughout out treatment, assess each patient’s risk for abuse, misuse, and addiction (See WARNINGS ).
- The continued use of benzodiazepines, including Clorazepate dipotassium tablets, may lead to clinically significant physical dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Abrupt discontinuation or rapid dosage reduction of Clorazepate dipotassium tablets after continued use may precipitate acute withdrawal reactions, which can be life- threatening. To reduce the risk of withdrawal reactions, use a gradual taper to discontinue Clorazepate dipotassium tablets or reduce the dosage (See DOSAGE AND ADMINISTRATION and WARNINGS ).
INDICATIONS AND USAGE
Clorazepate dipotassium tablets are indicatedfor the management of anxiety disordersorfortheshort-term relief of the symptoms of anxiety. Anxiety or tensionassociatedwith the stress of everyday life usually does not require treatmentwith an anxiolytic.
Clorazepate dipotassium tablets are indicated as adjunctivetherapy in themanagement of partialseizures.
Theeffectiveness of Clorazepate dipotassium tablets in long-term management of anxiety, that is,more than 4 months, has not beenassessed by systematicclinicalstudies.Long-term studies in epilepticpatients,however, have showncontinuedtherapeuticactivity.Thephysician should reassessperiodically the usefulnessof the drug for the individualpatient.
Clorazepate dipotassium tabletsare indicatedfor the symptomaticrelief of acute alcoholwithdrawal.
DOSAGE AND ADMINISTRATION
FortheSymptomatic ReliefofAnxiety: Clorazepate dipotassium tablets are administeredorally in divided doses. The usual daily dose is 30mg.The dose should beadjustedgraduallywithintherange of 15 to 60 mg daily in accordancewith the response of the patient. In elderly or debilitatedpatientsit is advisable to initiate treatment at a daily dose of 7.5 to 15 mg.
Clorazepate dipotassium tabletsmay also be administeredin a singledosedaily at bedtime; the recommendedinitialdose is 15 mg.After the initial dose, the response of the patient mayrequireadjustment of subsequent dosage.Lower doses may be indicated in the elderlypatient. Drowsinessmay occur at the initiation of treatment and withdosageincrement.
For the Symptomatic Relief of Acute Alcohol Withdrawal:
Thefollowing dosage scheduleisrecommended:
| 1st 24 hours (Day 1) | 30 mginitially;followed by 30 to 60 mg in divideddoses |
| 2nd 24 hours (Day 2) | 45 to 90 mg in divideddoses |
| 3rd 24 hours (Day 3) | 22.5 to 45 mg in divided doses |
| Day 4 | 15 to 30 mg in divideddoses |
Thereafter,graduallyreduce the daily dose to 7.5 to 15 mg.Discontinuedrugtherapy as soon as patient’s condition is stable.
Themaximum recommendedtotaldailydose is 90 mg. Avoid excessive reductionsin the totalamount of drug administeredonsuccessive days.
As an Adjunct to Antiepileptic Drugs: In order to minimizedrowsiness, the recommendedinitialdosages and dosageincrementsshould not be exceeded.
Adults: Themaximum recommendedinitialdose in patients over 12 years old is 7.5 mg three times a day. Dosage should be increased by no more than 7.5mg every week and should not exceed 90 mg/day.
Children (9-12 years): Themaximum recommendedinitial dose is 7.5 mgtwotimes a day. Dosage should be increased by no more than 7.5mg every week and should not exceed 60 mg/day.
Discontinuation or DosageReduction of Clorazepate dipotassium tablets: To reduce the risk of withdrawalreactions,use a gradualtaper to discontinueClorazepate dipotassium tablets or reduce the dosage. If a patient develops withdrawalreactions,consider pausing the taper or increasingthe dosage to the previous tapereddosage level. Subsequentlydecreasethedosagemoreslowly (see WARNINGS and DRUG ABUSE AND DEPENDENCE ).
CONTRAINDICATIONS
Clorazepate dipotassium tabletsare contraindicated in patientswith a knownhypersensitivityto the drug and in those withacutenarrow angle glaucoma.
ADVERSE REACTIONS
The side effectmostfrequentlyreported wasdrowsiness.Lesscommonlyreported (in descending order of occurrence)were:dizziness, various gastrointestinal complaints,nervousness, blurred vision, dry mouth,headache, and mentalconfusion.Other side effects included insomnia,transientskinrashes,fatigue,ataxia,genitourinarycomplaints,irritability,diplopia,depression,tremor, and slurredspeech.
There have been reports of abnormalliver and kidneyfunction tests and of decrease in hematocrit.
Decrease in systolicbloodpressurehas been observed.
To report SUSPECTED ADVERSE REACTIONS, contact Aurobindo Pharma USA, Inc. at 1-866-850-2876 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DRUG INTERACTIONS
Theconcomitant use of benzodiazepines and opioidsincreases the riskof respiratorydepressionbecause of actionsatdifferentreceptor sites in the CNS that control respiration.Benzodiazepinesinteract at GABAA sites and opioidsinteractprimarilyatmu receptors. Whenbenzodiazepinesand opioids arecombined, the potentialforbenzodiazepinestosignificantlyworsenopioid-relatedrespiratorydepressionexists.Limitdosageandduration of concomitant use of benzodiazepines and opioids, and monitorpatients closelyforrespiratorydepressionandsedation.
If Clorazepate dipotassium tablets is to be combined with other drugs acting on the central nervous system, careful consideration should be given to thepharmacology of the agentsto be employed.Animalexperienceindicates that clorazepatedipotassium prolongs thesleepingtimeafterhexobarbital or afterethylalcohol,increases the inhibitoryeffects of chlorpromazine, but does not exhibitmonoamineoxidaseinhibition.Clinicalstudies have shownincreasedsedationwithconcurrent hypnotic medications.Theactions of the benzodiazepines may be potentiated by barbiturates,narcotics,phenothiazines,monoamine oxidase inhibitors or other antidepressants.
If Clorazepate dipotassium tablets are used to treatanxietyassociatedwithsomatic disease states,carefulattentionmust be paid to possible druginteractionwith concomitantmedication.
In bioavailabilitystudieswithnormalsubjects,theconcurrentadministration of antacidsattherapeuticlevelsdid not significantlyinfluencethebioavailability of Clorazepate dipotassium tablets.
DESCRIPTION
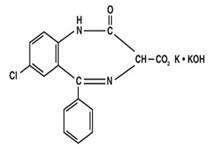
Chemically, Clorazepate dipotassium is a benzodiazepine. The empirical formula is C16H11ClK2N2O4; the molecular weight is 408.92; 1 H -1, 4 Benzodiazepine-3-carboxylic acid, 7-chloro-2,3-dihydro-2-oxo-5-phenyl-, potassium salt compound with potassium hydroxide (1:1) and the structural formula may be represented as follows:
The compound occurs as a fine, light yellow, practically odorless powder. It is insoluble in the common organic solvents, but very soluble in water. Aqueous solutions are unstable, clear, light yellow, and alkaline.
Clorazepate dipotassium tablets, USP contain 3.75 mg, 7.5 mg or 15 mg of clorazepate dipotassium, USP for oral administration. Inactive ingredients for Clorazepate dipotassium tablets: croscarmellose sodium, magnesium oxide, magnesium stearate, microcrystalline cellulose, potassium carbonate, sodium chloride and sodium lauryl sulfate. The 3.75 mg tablets also contain FD&C Blue No. 2 Aluminum Lake and the 7.5 mg tablets also contain FD&C Yellow No. 6 Aluminum Lake.
CLINICAL PHARMACOLOGY
Pharmacologically, clorazepate dipotassium has the characteristics of the benzodiazepines. It has depressant effects on the central nervous system. The primary metabolite, nordiazepam, quickly appears in the blood stream. The serum half-life is about 2 days. The drug is metabolized in the liver and excreted primarily in the urine.
Studies in healthy men have shown that clorazepate dipotassium has depressant effects on the central nervous system. Prolonged administration of single daily doses as high as 120 mg was without toxic effects. Abrupt cessation of high doses was followed in some patients by nervousness, insomnia, irritability, diarrhea, muscle aches, or memory impairment.
Since orally administered clorazepate dipotassium is rapidly decarboxylated to form nordiazepam, there is essentially no circulating parent drug. Nordiazepam, the primary metabolite, quickly appears in the blood and is eliminated from the plasma with an apparent half-life of about 40 to 50 hours. Plasma levels of nordiazepam increase proportionally with Clorazepate dipotassium tablets dose and show moderate accumulation with repeated administration. The protein binding of nordiazepam in plasma is high (97-98%).
Within 10 days after oral administration of a 15 mg (50 µCi) dose of 14 C-Clorazepate dipotassium tablets to two volunteers, 62-67% of the radioactivity was excreted in the urine and 15-19% was eliminated in the feces. Both subjects were still excreting measurable amounts of radioactivity in the urine (about 1% of the 14 C-dose) on day ten.
Nordiazepam is further metabolized by hydroxylation. The major urinary metabolite is conjugated oxazepam (3-hydroxynordiazepam), and smaller amounts of conjugated p-hydroxynordiazepam and nordiazepam are also found in the urine.
HOW SUPPLIED
Clorazepate Dipotassium Tablets, USP are available containing 3.75 mg, 7.5 mg or 15 mg of clorazepate dipotassium, USP.
The 3.75 mg tablets are blue, round, scored tablets debossed with M above the score and 30 below the score on one side of the tablet and blank on the other side. They are available as follows:
Bottles of 100 NDC 13107-319-01
Bottles of 500 NDC 13107-319-05
The 7.5 mg tablets are peach, round, scored tablets debossed with M above the score and 40 below the score on one side of the tablet and blank on the other side. They are available as follows:
Bottles of 100 NDC 13107-320-01
Bottles of 500 NDC 13107-320-05
The 15 mg tablets are white, round, scored tablets debossed with M above the score and 70 below the score on one side of the tablet and blank on the other side. They are available as follows:
Bottles of 100 NDC 13107-321-01
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from light and moisture.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
PHARMACIST: Dispense a Medication Guide with each prescription.
Dispense with Medication Guide available at : www.aurobindousa.com/medication-guides
Manufactured by: Alphapharm Pty Ltd 15 Garnet Street Carole Park QLD 4300 Australia Distributed by: Aurobindo Pharma USA, Inc. 279 Princeton-Hightstown Road East Windsor, NJ 08520 Revised: 04/2023