Clindesse - Clindamycin Phosphate cream prescribing information
INDICATIONS AND USAGE
Clindesse is a lincosamide antibacterial indicated for the treatment of bacterial vaginosis in non-pregnant women (1.1 )
Treatment of Bacterial Vaginosis
Clindesse is indicated for the treatment of bacterial vaginosis (formerly referred to as Haemophilus vaginitis, Gardnerella vaginitis, nonspecific vaginitis, Corynebacterium vaginitis, or anaerobic vaginosis) in non-pregnant women.
DOSAGE AND ADMINISTRATION
The recommended dose is the complete contents of a single pre-filled applicator containing 5 g of Clindesse cream administered once intravaginally at any time of the day.
Not for ophthalmic, dermal, or oral use.
DOSAGE FORMS AND STRENGTHS
Clindesse is an intravaginal cream containing clindamycin phosphate 2%. Each pre-filled, single-dose applicator delivers approximately 5 g of cream containing approximately 100 mg of clindamycin.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
Clindesse has not been studied in pregnant women. The systemic exposure (based on AUC and C max ) of Clindesse administered intravaginally is substantially lower than clindamycin administered intravenously [see Clinical Pharmacology (12.3 )] ; therefore, maternal use is not likely to result in significant fetal exposure to the drug. Available data from published observational studies and randomized controlled trials over decades of use with other clindamycin products during pregnancy have not identified an increased risk of major birth defects, miscarriage, or other adverse maternal and fetal outcomes.
In animal reproduction studies, no adverse developmental outcomes were observed in pregnant rats and mice administered oral doses of clindamycin at up to 600 mg/kg/day (58 and 29 times, respectively, the recommended human dose based on a body surface area comparison) ( see Data ).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
Reproduction studies have been performed in rats and mice using oral doses of clindamycin up to 600 mg/kg/day (58 and 29 times, respectively, the recommended human dose based on body surface area comparisons) and have revealed no evidence of harm to the fetus due to clindamycin.
Lactation
Risk Summary
The systemic exposure (based on AUC and C max ) of Clindesse is substantially lower than intravenous administration of clindamycin [see Clinical Pharmacology (12.3 )] ; therefore, adverse effects on the breastfed infant are not expected from transfer of clindamycin into breastmilk. There are no data on the effect of clindamycin on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Clindesse and any potential adverse effects on the breastfed infant from Clindesse or from the underlying maternal condition.
Females and Males of Reproductive Potential
Contraception
Clindesse contains mineral oil that may weaken latex or rubber products such as condoms or vaginal contraceptive diaphragms. Therefore, the use of such barrier contraceptives is not recommended concurrently or for 5 days following treatment with Clindesse. During this time period, condoms may not be reliable for preventing pregnancy or for protecting against transmission of HIV and other sexually transmitted diseases [see Warnings and Precautions (5.2 )].
Pediatric Use
The safety and efficacy of Clindesse in the treatment of bacterial vaginosis in post-menarchal females have been established on the extrapolation of clinical trial data from adult women. The safety and efficacy of Clindesse in pre-menarchal females have not been established.
Geriatric Use
Clinical studies with Clindesse did not include sufficient numbers of subjects 65 years of age or older to determine whether they respond differently than younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
CONTRAINDICATIONS
Hypersensitivity
Clindesse is contraindicated in individuals with a history of hypersensitivity to clindamycin or other lincosamides. Reported reactions to other formulations of clindamycin include rashes, urticaria, erythema multiforme, and anaphylactoid reactions [see Adverse Reactions (6.2 )] .
History of Bowel Disease
Clindesse is contraindicated in patients with regional enteritis, ulcerative colitis, or a history of Clostridioides difficile -associated diarrhea.
WARNINGS AND PRECAUTIONS
- Clostridioides difficile -Associated Diarrhea: Discontinue and evaluate if diarrhea occurs (5.1 )
- Use with Condoms and Vaginal Contraceptive Diaphragms: The use of barrier contraceptives (condoms or vaginal contraceptive diaphragms) is not recommended concurrently or for 5 days following treatment. Condoms may not be reliable for preventing pregnancy or for protecting against transmission of HIV and other sexually transmitted diseases during this time period (5.2 , 8.3 )
Clostridioides difficile -Associated Diarrhea (CDAD)
Clostridioides difficile -associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including clindamycin, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile .
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile , and surgical evaluation should be instituted as clinically indicated [see Adverse Reactions (6.2 )] .
Use with Condoms and Vaginal Contraceptive Diaphragms
This cream contains mineral oil that may weaken latex or rubber products such as condoms or vaginal contraceptive diaphragms. Therefore, the use of such barrier contraceptives is not recommended concurrently or for 5 days following treatment with Clindesse. During this time period, condoms may not be reliable for preventing pregnancy or for protecting against transmission of HIV and other sexually transmitted diseases [see Use in Specific Populations (8.3 )] .
ADVERSE REACTIONS
Most common adverse reactions reported in ≥2% of patients and at a higher rate in the Clindesse group than in the placebo group are vaginosis fungal (14%), headache (7%), back pain (5%), constipation (2%), and urinary tract infection (2%) (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Padagis ® at 1-866-634-9120 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Clinical Study Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to Clindesse in 368 patients. Clindesse was studied in three clinical studies: placebo-controlled (n=85), active-controlled (n=263), and single-arm (n=20). The population was female, aged 18 to 78, who were diagnosed with bacterial vaginosis. Patient demographics in the trials were 51% Caucasian, 36% Black, 10% Hispanic, and 3% Asian, other or unknown. All patients received 100 mg clindamycin phosphate cream intravaginally in a single dose.
Of the 368 women treated with a single dose of Clindesse, 1.6% of the patients discontinued therapy due to adverse reactions. Adverse reactions occurred in 126 of 368 patients (34%) treated with Clindesse and in 32 of 85 patients (38%) treated with placebo.
Adverse reactions occurring in ≥2% of patients receiving Clindesse in the placebo-controlled clinical trial are shown in Table 1.
Table 1. Adverse Reactions Occurring in ≥2% of Clindesse-Treated Patients and at a Higher Rate than Placebo-Treated Patients
Adverse Reactions | Clindesse N=85 n (%) | Placebo N=85 n (%) |
Vaginosis fungal NOS• | 12 (14) | 7 (8) |
Headache NOS | 6 (7) | 2 (2) |
Back Pain | 4 (5) | 1 (1) |
Constipation | 2 (2) | 0 (0) |
Urinary tract infection NOS | 2 (2) | 0 (0) |
N = number of patients in intent-to-treat population
n (%) = number and percentage of patients with reported adverse reaction
NOS = not otherwise specified
- •The use of clindamycin may result in the overgrowth of non-susceptible fungal organisms in the vagina and may require antifungal treatment
- Other reactions reported by <1% of those women treated with Clindesse include:
- Dermatologic: Pruritic rash
- Gastrointestinal: Diarrhea, vomiting
- General: Fatigue
- Immune System: Hypersensitivity
- Nervous System: Dizziness
- Reproductive System: Dysfunctional uterine bleeding, dysmennorrhea, intermenstrual bleeding, pelvic pain, vaginal burning, vaginal irritation, vulvar erythema, vulvitis, vulvovaginal discomfort, vulvovaginal dryness, vulvovaginitis
Other Clindamycin Formulations
Clindesse affords minimal peak serum levels and systemic exposure (AUCs) of clindamycin compared to an oral or intravenous dose of clindamycin [see Clinical Pharmacology (12.3 )] . Data from well-controlled trials directly comparing clindamycin administered orally to clindamycin administered vaginally are not available.
The following additional adverse reactions and altered laboratory tests have been reported with the oral or parenteral use of clindamycin:
Gastrointestinal: Abdominal pain, esophagitis, nausea, Clostridioides difficile -associated diarrhea [see Warnings and Precautions (5.1 )] .
Hematopoietic: Transient neutropenia (leukopenia), eosinophilia, agranulocytosis, and thrombocytopenia have been reported. No direct etiologic relationship to concurrent clindamycin therapy could be made in any of these reports.
Hypersensitivity Reactions: Maculopapular rash, vesiculobullous rash, and urticaria have been observed during drug therapy. Generalized mild to moderate morbilliform-like skin rashes are the most frequently reported of all adverse reactions. Cases of erythema multiforme, some resembling Stevens-Johnson syndrome, have been associated with clindamycin. A few cases of anaphylactoid reactions have been reported.
Liver: Jaundice and abnormalities in liver function tests have been observed during clindamycin therapy.
Musculoskeletal: Cases of polyarthritis have been reported.
Renal: Although no direct relationship of clindamycin to renal damage has been established, renal dysfunction as evidenced by azotemia, oliguria, and/or proteinuria has been observed in rare instances.
Immune System: Drug reaction with eosinophilia and systemic symptoms (DRESS) cases have been reported.
Postmarketing Experience
The following adverse reactions have been identified during postapproval use of Clindesse. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Dermatologic: Rash
Gastrointestinal: Hematochezia
Reproductive System: Vaginal erythema, vulvovaginal pruritis, vaginal discharge, vaginal swelling, vaginal bleeding, vaginal pain
DRUG INTERACTIONS
No formal drug interaction studies have been conducted for Clindesse.
Neuromuscular Blocking Agents
Orally or intravenously administered clindamycin has neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, it should be used with caution in patients receiving such agents.
DESCRIPTION
Clindamycin phosphate, a lincosamide, is a water soluble ester of the semi-synthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent antibiotic lincomycin. The chemical name for clindamycin phosphate is methyl 7-chloro- 6,7,8-trideoxy-6-(1-methyl- trans - 4-propyl-L-2-pyrrolidinecarboxamido)-1-thio-L- threo-(alpha)-D-galacto- octopyranoside 2-(dihydrogen phosphate). It has a molecular weight of 504.96, and the molecular formula is C 18 H 34 CIN 2 O 8 PS. The structural formula is represented below:
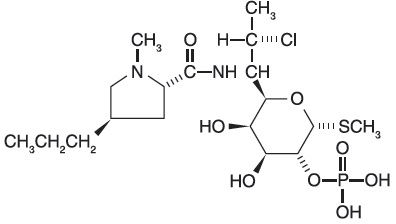
Clindesse is a semi-solid, white cream, which contains clindamycin phosphate, USP, at a concentration equivalent to 20 mg clindamycin base per gram. The cream also contains edetate disodium, glycerol monoisostearate, lecithin, methylparaben, microcrystalline wax, mineral oil, polyglyceryl-3-oleate, propylparaben, purified water, silicon dioxide and sorbitol solution.
Clindesse does not comply with the pH test of the USP monograph for clindamycin phosphate vaginal cream.
CLINICAL PHARMACOLOGY
Mechanism of Action
Clindamycin is an antibacterial drug [see Clinical Pharmacology, Microbiology (12.4 )] .
Pharmacokinetics
Following a single intravaginal application of Clindesse cream to 20 healthy women, the mean (range) AUC 0-inf and C max estimates were 175 (38.6 to 541) ng/mL•hr and 6.6 (0.8 to 39) ng/mL, respectively. The mean C max of clindamycin for Clindesse was approximately 0.3%, 0.1%, and 7.6% of that observed after the administration of a 150 mg Cleocin oral capsule (2.5 mcg/mL), a 600 mg Cleocin intravenous injection (10.9 mcg/mL), and a single dose of 100 mg of Cleocin Vaginal Cream (86.5 ng/mL), respectively. The peak serum concentration of clindamycin was attained approximately 20 hours post dosing for Clindesse.
Microbiology
Mechanism of Action
Clindamycin inhibits bacterial protein synthesis at the level of the bacterial ribosome. The antibiotic binds preferentially to the 50S ribosomal subunit and affects the process of peptide chain initiation. Although clindamycin phosphate is inactive in vitro, in vivo hydrolysis converts this compound to the antibacterially active clindamycin.
Activity In Vitro
Clindamycin is an antibacterial agent active in vitro against most strains of the following organisms that have been reported to be associated with bacterial vaginosis:
Bacteroides spp.
Gardnerella vaginalis
Mobiluncus spp.
Mycoplasma hominis
Peptostreptococcus spp.
Standard methodology for the susceptibility testing of the potential bacterial vaginosis pathogens has not been defined. Culture and sensitivity testing of bacteria are not routinely performed to establish the diagnosis of bacterial vaginosis [see Clinical Studies (14 )] .
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed with clindamycin to evaluate carcinogenic potential. Genotoxicity tests performed included a rat micronucleus test and an Ames test. Both tests were negative. Fertility studies in rats treated orally with up to 300 mg/kg/day (29 times the recommended human dose based on body surface area comparisons) revealed no effects on fertility or mating ability.
CLINICAL STUDIES
Two clinical studies were conducted to evaluate the efficacy of Clindesse for the treatment of bacterial vaginosis. A clinical diagnosis of bacterial vaginosis was defined by the presence of a homogeneous vaginal discharge that (a) has a pH of greater than 4.5, (b) emits a “fishy” amine odor when mixed with a 10% KOH solution, and (c) contains clue cells on microscopic examination. Gram’s stain results consistent with a diagnosis of bacterial vaginosis include (a) markedly reduced or absent Lactobacillus morphology, (b) predominance of Gardnerella morphotype, and (c) absent or few white blood cells.
In a randomized, double-blind, placebo-controlled, clinical study involving 144 non-pregnant female patients aged 18 to 64 with a baseline Nugent score ≥4, Clindesse demonstrated statistically significantly higher cure rates over placebo as measured by therapeutic cure, clinical cure, and Nugent score cure (Table 2) assessed at 21-30 days after administration of the drug. Therapeutic cure was a composite endpoint which required both clinical cure and Nugent score cure. Clinical cure required normal vaginal discharge, vaginal pH < 4.7, < 20% clue cells on wet mount preparation, and negative “whiff” test (detection of amine odor on addition of 10% KOH solution to sample of the vaginal discharge). A Nugent score of 0-3 was considered a Nugent score cure. The Nugent scoring is based on microscopic examination of the Gram’s stained vaginal smears for quantification of specific bacterial morphotypes. Cure rates were consistently higher for Clindesse compared to placebo for the following demographic subsets: age, race, height, weight, sexual behavior, and recalcitrant infection status.
Table 2. Efficacy of Clindesse for Treatment of Bacterial Vaginosis in a Randomized, Double-Blind, Placebo-Controlled, Parallel Group Study
Outcome | Clindesse N=78 % Cure | Placebo N=66 % Cure | Treatment Difference † (%) [97.5% Confidence Interval] |
Therapeutic Cure | 29.5 | 3.0 | 26.5 [14.0, 39.0] |
Clinical Cure | 41.0 | 19.7 | 21.3 [4.7, 38.0] |
Nugent Score Cure | 44.9 | 6.1 | 38.8 [24.6, 53.1] |
N = number of patients in treatment group (modified intent-to-treat population defined as all subjects randomized who received at least one dose of study medication, and who had a baseline Nugent score of at least 4)
† Treatment difference = Clindesse minus placebo cure rates
In a second controlled clinical study involving 432 patients aged 18 to 78 with a baseline Nugent score of ≥4, 221 women self-administered a single dose of Clindesse, and 211 women self-administered a single daily dose of a formulation of clindamycin vaginal cream for 7 days. A single dose of Clindesse was shown to be similar to 7 daily doses of the clindamycin vaginal cream for treatment of bacterial vaginosis as measured by therapeutic cure, clinical cure or Nugent score cure assessed at 21-30 days after administration of the drug in the modified intent-to-treat population (Table 3) and for the per protocol population (Table 4). The study endpoints were identical to those described above for the placebo-controlled study. Statistical analyses did not reveal any significant differences when controlling for the following demographic variables: age, race, height, weight, sexual behavior, and recalcitrant infection status.
The cure rates reported in the clinical studies with Clindesse were based on resolution of 4 out of 4 Amsel criteria and a Nugent score of < 4, while the criteria for cure in previous clinical studies with the clindamycin vaginal cream were based solely on resolution of 2 out of 4 Amsel criteria, resulting in higher reported rates of cure for bacterial vaginosis.
Table 3. Efficacy of Clindesse in Treatment of Bacterial Vaginosis in a Randomized, Investigator-Blind, Active-Controlled Comparative Study – Modified-Intent-to-Treat
Outcome | Clindesse Single Dose N=221 % Cure | Clindamycin Vaginal Cream (7 doses) N=211 % Cure | Treatment Difference † (%) [95% Confidence Interval] |
Therapeutic Cure | 33.0 | 37.0 | -3.9 [-12.9, 5.1] |
Clinical Cure | 53.4 | 54.0 | -0.6 [-10.0, 8.8] |
Nugent Score Cure | 45.7 | 49.3 | -3.6 [-13.1, 5.8] |
† Treatment difference = Clindesse minus clindamycin vaginal cream cure rates
N = number of patients in treatment group (modified intent-to-treat population defined as all subjects randomized who received at least one dose of study medication, and who had a baseline Nugent score of at least 4)
Table 4. Efficacy of Clindesse in Treatment of Bacterial Vaginosis in a Randomized, Investigator-Blind, Active-Controlled Comparative Study – Per Protocol
Outcome | Clindesse Single Dose N=126 % Cure | Clindamycin Vaginal Cream (7 doses) N=125 % Cure | Treatment Difference † (%) [95% Confidence Interval] |
Therapeutic Cure | 42.1 | 45.6 | -3.5 [-15.8, 8.7] |
Clinical Cure | 64.3 | 63.2 | 1.1 [-10.8, 13.0] |
Nugent Score Cure | 56.5 ‡ | 57.7 ‡ | -1.3 [-13.6, 11.1] |
† Treatment difference = Clindesse minus clindamycin vaginal cream cure rates
N = number of patients in treatment group (per protocol population defined as all subjects included in the modified intent-to-treat population who completed the study without significant protocol violation)
‡ Four subjects (2 from each treatment group) did not have complete Nugent scores and were not included in the Nugent Score cure analysis
HOW SUPPLIED/STORAGE AND HANDLING
Clindesse (clindamycin phosphate) Vaginal Cream, 2%, is available in cartons containing one single-dose, pre-filled disposable applicator (NDC 45802-042-01). Each applicator delivers approximately 5 g of vaginal cream containing approximately 100 mg of clindamycin.
Store at 20°-25°C (68°-77°F). [See USP Controlled Room Temperature.]
Avoid heat above 30°C (86°F).
Instructions for Use
Clindesse (clin-DESS)
(clindamycin phosphate)
Vaginal Cream, 2%
For vaginal use only.
Do not put Clindesse in your eyes, mouth, or on your skin.
It is important that you read and follow these directions on how to use Clindesse vaginal cream properly.
Clindesse comes in a single-dose, pre-filled, disposable applicator that gives you a certain amount of clindamycin cream to be inserted into your vagina.
Step 1. Prepare the applicator.
Peel back the protective foil and remove the pre-filled applicator. Do not remove the tip.
The applicator is made to be used with the tip in place.
Do not use the applicator if the tip has been removed (see Figure 1).
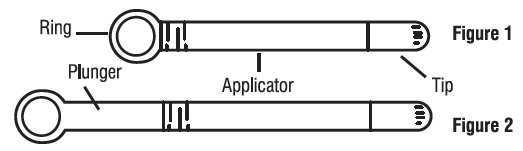
Activate the plunger before you use it. To activate the plunger, pull the ring back to fully extend the plunger while you firmly hold the applicator (see Figure 2).
Step 2. Insert the applicator.
Gently insert the applicator into your vagina as far as it will comfortably go (see Figure 3).
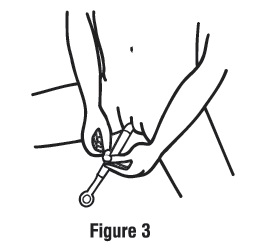
Step 3. Apply the cream.
Push the plunger in until all of the cream goes into your vagina (see Figures 4 and 5).
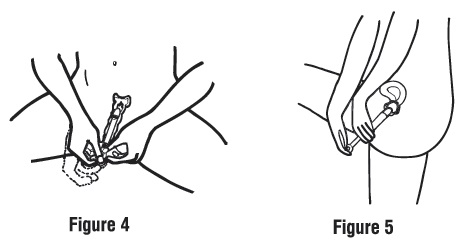
Step 4. Remove the empty applicator from your vagina and throw it away in the trash.
This Instructions for Use has been approved by the U.S. Food and Drug Administration
01/2025
Mechanism of Action
Clindamycin is an antibacterial drug [see Clinical Pharmacology, Microbiology (12.4 )] .