Clindamycin Phosphate And Tretinoin - Clindamycin Phosphate And Tretinoin gel prescribing information
INDICATIONS AND USAGE
Clindamycin phosphate and tretinoin gel is indicated for the topical treatment of acne vulgaris in patients 12 years and older.
DOSAGE AND ADMINISTRATION
Clindamycin phosphate and tretinoin gel should be applied once daily in the evening, gently rubbing the medication to lightly cover the entire affected area. Approximately a pea-sized amount will be needed for each application. Avoid the eyes, lips, and mucous membranes.
Clindamycin phosphate and tretinoin gel is not for oral, ophthalmic, or intravaginal use.
DOSAGE FORMS AND STRENGTHS
Clindamycin phosphate and tretinoin gel, 1.2%/0.025%, containing clindamycin phosphate 1.2% and tretinoin 0.025%, is a yellow, opaque topical gel. Each gram of clindamycin phosphate and tretinoin gel, 1.2%/0.025% contains, as dispensed, 10 mg (1%) clindamycin as clindamycin phosphate, and 0.25 mg (0.025%) tretinoin solubilized in an aqueous-based gel.
USE IN SPECIFIC POPULATIONS
- Pediatric Use: The efficacy and safety have not been established in pediatric patients below the age of 12 years. (8.4 )
Pregnancy
Pregnancy Category C.
There are no well-controlled studies in pregnant women treated with clindamycin phosphate and tretinoin gel. Clindamycin phosphate and tretinoin gel should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. A limit teratology study performed in Sprague Dawley rats treated topically with clindamycin phosphate and tretinoin gel or 0.025% tretinoin gel at a dose of 2 mL/kg during gestation days 6 to 15 did not result in teratogenic effects. Although no systemic levels of tretinoin were detected, craniofacial and heart abnormalities were described in drug-treated groups. These abnormalities are consistent with retinoid effects and occurred at 16 times the recommended clinical dose assuming 100% absorption and based on body surface area comparison. For purposes of comparison of the animal exposure to human exposure, the recommended clinical dose is defined as 1 g of clindamycin phosphate and tretinoin gel applied daily to a 50-kg person.
Clindamycin: Reproductive developmental toxicity studies performed in rats and mice using oral doses of clindamycin up to 600 mg/kg/day (480 and 240 times the recommended clinical dose based on body surface area comparison, respectively) or subcutaneous doses of clindamycin up to 180 mg/kg/day (140 and 70 times the recommended clinical dose based on body surface area comparison, respectively) revealed no evidence of teratogenicity.
Tretinoin: Oral tretinoin has been shown to be teratogenic in mice, rats, hamsters, rabbits, and primates. It was teratogenic and fetotoxic in Wistar rats when given orally at doses greater than 1 mg/kg/day (32 times the recommended clinical dose based on body surface area comparison). However, variations in teratogenic doses among various strains of rats have been reported. In the cynomologous monkey, a species in which tretinoin metabolism is closer to humans than in other species examined, fetal malformations were reported at oral doses of 10 mg/kg/day or greater, but none were observed at 5 mg/kg/day (324 times the recommended clinical dose based on body surface area comparison), although increased skeletal variations were observed at all doses. Dose-related teratogenic effects and increased abortion rates were reported in pigtail macaques.
With widespread use of any drug, a small number of birth defect reports associated temporally with the administration of the drug would be expected by chance alone. Thirty cases of temporally associated congenital malformations have been reported during 2 decades of clinical use of another formulation of topical tretinoin. Although no definite pattern of teratogenicity and no causal association have been established from these cases, 5 of the reports describe the rare birth defect category, holoprosencephaly (defects associated with incomplete midline development of the forebrain). The significance of these spontaneous reports in terms of risk to fetus is not known.
Nursing Mothers
It is not known whether clindamycin is excreted in human milk following use of clindamycin phosphate and tretinoin gel. However, orally and parenterally administered clindamycin has been reported to appear in breast milk. Because of the potential for serious adverse reactions in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. It is not known whether tretinoin is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when clindamycin phosphate and tretinoin gel is administered to a nursing woman.
Pediatric Use
Safety and effectiveness of clindamycin phosphate and tretinoin gel in pediatric patients below the age of 12 years have not been established.
Clinical trials of clindamycin phosphate and tretinoin gel included 2,086 patients aged 12 - 17 years of age with acne vulgaris. [See Clinical Studies (14 ).]
Geriatric Use
Clinical studies of clindamycin phosphate and tretinoin gel did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
CONTRAINDICATIONS
Clindamycin phosphate and tretinoin gel is contraindicated in patients with regional enteritis, ulcerative colitis, or history of antibiotic-associated colitis.
WARNINGS AND PRECAUTIONS
- Colitis: Clindamycin can cause severe colitis, which may result in death. Diarrhea, bloody diarrhea, and colitis (including pseudomembranous colitis) have been reported with the use of clindamycin. Clindamycin phosphate and tretinoin gel should be discontinued if significant diarrhea occurs. (5.1 )
- Ultraviolet Light and Environmental Exposure: Avoid exposure to sunlight, sunlamps, and weather extremes. Wear sunscreen daily. (5.2 )
Colitis
Systemic absorption of clindamycin has been demonstrated following topical use. Diarrhea, bloody diarrhea, and colitis (including pseudomembranous colitis) have been reported with the use of topical clindamycin. If significant diarrhea occurs, clindamycin phosphate and tretinoin gel should be discontinued.
Severe colitis has occurred following oral or parenteral administration of clindamycin with an onset of up to several weeks following cessation of therapy. Antiperistaltic agents such as opiates and diphenoxylate with atropine may prolong and/or worsen severe colitis. Severe colitis may result in death.
Studies indicate a toxin(s) produced by clostridia is one primary cause of antibiotic-associated colitis. The colitis is usually characterized by severe persistent diarrhea and severe abdominal cramps and may be associated with the passage of blood and mucus. Stool cultures for Clostridium difficile and stool assay for C. difficile toxin may be helpful diagnostically.
Ultraviolet Light and Environmental Exposure
Exposure to sunlight, including sunlamps, should be avoided during the use of clindamycin phosphate and tretinoin gel. Patients with sunburn should be advised not to use the product until fully recovered because of heightened susceptibility to sunlight as a result of the use of tretinoin. Patients who may be required to have considerable sun exposure due to occupation and those with inherent sensitivity to the sun should exercise particular caution. Daily use of sunscreen products and protective apparel (e.g., a hat) are recommended. Weather extremes, such as wind or cold, also may be irritating to patients under treatment with clindamycin phosphate and tretinoin gel.
ADVERSE REACTIONS
- Observed local treatment-related adverse reactions (≥ 1%) in clinical studies with clindamycin phosphate and tretinoin gel were application site reactions, including dryness, irritation, exfoliation, erythema, pruritus, and dermatitis. Sunburn was also reported. (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Solaris Pharma Corporation at 1-833-919-0527 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Adverse Reactions in Clinical Studies
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
The safety data reflect exposure to clindamycin phosphate and tretinoin gel in 1,104 patients with acne vulgaris. Patients were 12 years or older and were treated once daily in the evening for 12 weeks. Adverse reactions that were reported in ≥1% of patients treated with clindamycin phosphate and tretinoin gel are presented in Table 1 .
| Clindamycin Phosphate and Tretinoin Gel N = 1,104 n (%) | Clindamycin Gel N = 1,091 n (%) | Tretinoin Gel N = 1,084 n (%) | Vehicle Gel N = 552 n (%) | |
| Patients with at least one adverse reaction | 140 (13) | 38 (3) | 141 (13) | 17 (3) |
| Application site dryness | 64 (6) | 12 (1) | 62 (6) | 3 (1) |
| Application site irritation | 50 (5) | 4 (<1) | 57 (5) | 5 (1) |
| Application site exfoliation | 50 (5) | 2 (<1) | 56 (5) | 2 (<1) |
| Application site erythema | 40 (4) | 6 (1) | 39 (4) | 3 (1) |
| Application site pruritus | 26 (2) | 7 (1) | 23 (2) | 6 (1) |
| Sunburn | 11 (1) | 6 (1) | 7 (1) | 3 (1) |
| Application site dermatitis | 6 (1) | 0 (0) | 8 (1) | 1 (<1) |
Local skin reactions actively assessed at baseline and end of treatment with a score >0 are presented in Table 2 .
| Clindamycin Phosphate and Tretinoin Gel | Vehicle Gel | |||
| Local Reaction | Baseline N = 476 (%) | End of Treatment N = 409 (%) | Baseline N = 219 (%) | End of Treatment N = 209 (%) |
| Erythema | 24% | 21% | 31% | 35% |
| Scaling | 8% | 19% | 14% | 12% |
| Dryness | 11% | 22% | 18% | 13% |
| Burning | 8% | 13% | 8% | 4% |
| Itching | 17% | 15% | 22% | 14% |
During the 12 weeks of treatment, each local skin reaction peaked at Week 2 and gradually reduced thereafter.
DRUG INTERACTIONS
- Clindamycin phosphate and tretinoin gel should not be used in combination with erythromycin-containing products because of its clindamycin component. (7.1 )
Erythromycin
Clindamycin phosphate and tretinoin gel should not be used in combination with erythromycin-containing products due to possible antagonism to the clindamycin component. In vitro studies have shown antagonism between these 2 antimicrobials. The clinical significance of this in vitro antagonism is not known.
Neuromuscular Blocking Agents
Clindamycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, clindamycin phosphate and tretinoin gel should be used with caution in patients receiving such agents.
DESCRIPTION
Clindamycin phosphate and tretinoin gel, 1.2%/0.025%, is a fixed combination of 2 solubilized active ingredients in an aqueous-based gel. Clindamycin phosphate is a water soluble ester of the semi-synthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent antibiotic lincomycin.
The chemical name for clindamycin phosphate is methyl 7-chloro-6,7,8-trideoxy-6-(1-methyl-trans-4-propyl-L-2-pyrrolidinecarboxamido)-1-thio-L-threo-α-D-galacto-octopyranoside 2-(dihydrogen phosphate). The structural formula for clindamycin phosphate is represented below:
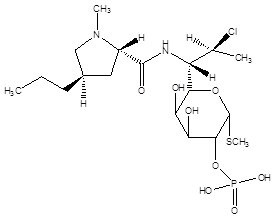
Molecular Formula: C 18 H 34 ClN 2 O 8 PS
Molecular Weight: 504.97
The chemical name for tretinoin is all-trans 3,7-dimethyl-9-(2,6,6-trimethyl-1-cyclohexen-1-yl)-2,4,6,8-nonatetraenoic acid. It is a member of the retinoid family of compounds.
The structural formula for tretinoin is represented below:
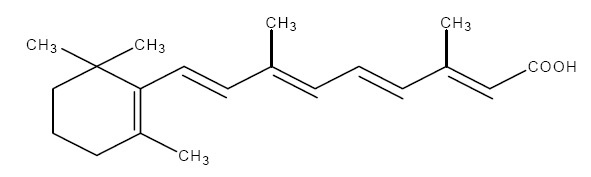
Molecular Formula: C 20 H 28 O 2
Molecular Weight: 300.44
Clindamycin phosphate and tretinoin gel contains the following inactive ingredients: anhydrous citric acid, butylated hydroxytoluene, carbomer homopolymer (type C), edetate disodium, laureth 4, methylparaben, propylene glycol, purified water, and tromethamine.
CLINICAL PHARMACOLOGY
Mechanism of Action
Clindamycin: [See Microbiology (12.4 ).] Tretinoin: Although the exact mode of action of tretinoin is unknown, current evidence suggests that topical tretinoin decreases cohesiveness of follicular epithelial cells with decreased microcomedone formation. Additionally, tretinoin stimulates mitotic activity and increased turnover of follicular epithelial cells causing extrusion of the comedones.
Pharmacokinetics
In an open-label study of 17 patients with moderate-to-severe acne vulgaris, topical administration of approximately 3 grams of clindamycin phosphate and tretinoin gel once daily for 5 days, clindamycin concentrations were quantifiable in all 17 patients starting from 1 hour post-dose. All plasma clindamycin concentrations were ≤5.56 ng/mL on Day 5, with the exception of 1 subject who had a maximum clindamycin concentration of 8.73 ng/mL at 4 hours post-dose. There was no appreciable increase in systemic exposure to tretinoin, as compared with the baseline value. The average tretinoin concentration across all sampling times on Day 5 ranged from 1.19 to 1.23 ng/mL compared with the corresponding baseline mean tretinoin concentration range of 1.16 to 1.30 ng/mL.
Microbiology
No microbiology studies were conducted in the clinical trials with this product.
Mechanism of Action: Clindamycin binds to the 50S ribosomal subunit of susceptible bacteria and prevents elongation of peptide chains by interfering with peptidyl transfer, thereby suppressing protein synthesis. Clindamycin has been shown to have in vitro activity against Propionibacterium acnes (P. acnes) , an organism that has been associated with acne vulgaris; however, the clinical significance of this activity against P. acnes was not examined in clinical studies with clindamycin phosphate and tretinoin gel. P. acnes resistance to clindamycin has been documented. Inducible Clindamycin Resistance: The treatment of acne with antimicrobials is associated with the development of antimicrobial resistance in P. acnes as well as other bacteria (e.g., Staphylococcus aureus, Streptococcus pyogenes ). The use of clindamycin may result in developing inducible resistance in these organisms. This resistance is not detected by routine susceptibility testing. Cross Resistance: Resistance to clindamycin is often associated with resistance to erythromycin.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential of clindamycin phosphate and tretinoin gel or the effect of clindamycin phosphate and tretinoin gel on fertility. Clindamycin phosphate and tretinoin gel was negative for mutagenic potential when evaluated in an in vitro Ames Salmonella reversion assay. Clindamycin phosphate and tretinoin gel was equivocal for clastogenic potential in the absence of metabolic activation when tested in an in vitro chromosomal aberration assay.
Clindamycin: Once-daily dermal administration of 1% clindamycin as clindamycin phosphate in the gel vehicle (32 mg/kg/day, 13 times the recommended clinical dose based on body surface area comparison) to mice for up to 2 years did not produce evidence of tumorigenicity.
Fertility studies in rats treated orally with up to 300 mg/kg/day of clindamycin (240 times the recommended clinical dose based on body surface area comparison) revealed no effects on fertility or mating ability.
Tretinoin: In 2 independent mouse studies where tretinoin was administered topically (0.025% or 0.1%) 3 times per week for up to 2 years no carcinogenicity was observed, with maximum effects of dermal amyloidosis. However, in a dermal carcinogenicity study in mice, tretinoin applied at a dose of 5.1 mcg (1.4 times the recommended clinical dose based on body surface area comparison) 3 times per week for 20 weeks acted as a weak promoter of skin tumor formation following a single application of dimethylbenz[α]anthracene (DMBA).
In a study in female SENCAR mice, papillomas were induced by topical exposure to DMBA followed by promotion with 12-O-tetradecanoylphorbol-13-acetate or mezerein for up to 20 weeks. Topical application of tretinoin prior to each application of promoting agent resulted in a reduction in the number of papillomas per mouse. However, papillomas resistant to topical tretinoin suppression were at higher risk for pre-malignant progression.
Tretinoin has been shown to enhance photoco-carcinogenicity in properly performed specific studies, employing concurrent or intercurrent exposure to tretinoin and UV radiation. The photoco-carcinogenic potential of the clindamycin tretinoin combination is unknown. Although the significance of these studies to humans is not clear, patients should avoid exposure to sun.
The genotoxic potential of tretinoin was evaluated in an in vitro Ames Salmonella reversion test and an in vitro chromosomal aberration assay in Chinese hamster ovary cells. Both tests were negative.
In oral fertility studies in rats treated with tretinoin, the no-observed-effect-level was 2 mg/kg/day (64 times the recommended clinical dose based on body surface area comparison).
CLINICAL STUDIES
The safety and efficacy of clindamycin phosphate and tretinoin gel, applied once daily for the treatment of acne vulgaris, was evaluated in 12-week multi-center, randomized, blinded studies in subjects 12 years and older.
Treatment response was defined as the percent of subjects who had a 2-grade improvement from baseline to Week 12 based on the Investigator’s Global Assessment (IGA) and a mean absolute change from baseline to Week 12 in 2 out of 3 (total, inflammatory and non-inflammatory) lesion counts. The IGA scoring scale used in all the clinical trials for clindamycin phosphate and tretinoin gel is as follows:
| 0 | Clear | Normal, clear skin with no evidence of acne vulgaris. |
| 1 | Almost Clear | Skin almost clear; rare non-inflammatory lesions present, with rare non-inflamed papules (papules must be resolving and may be hyperpigmented, though not pink-red) requiring no further treatment in the Investigator’s opinion. |
| 2 | Mild | Some non-inflammatory lesions are present, with few inflammatory lesions (papules/pustules only, no nodulo-cystic lesions). |
| 3 | Moderate | Non-inflammatory lesions predominate, with multiple inflammatory lesions evident; several-to-many comedones and papules/pustules, and there may or may not be 1 small nodulo-cystic lesion. |
| 4 | Severe | Inflammatory lesions are more apparent; many comedones and papules/pustules, there may or may not be a few nodulo-cystic lesions. |
| 5 | Very Severe | Highly inflammatory lesions predominate; variable numbers of comedones, many papules/pustules and nodulo-cystic lesions. |
In Study 1, 1,649 subjects were randomized to clindamycin phosphate and tretinoin gel, clindamycin gel, tretinoin gel, and vehicle gel. The median age of subjects was 17 years and 58% were females. At baseline, subjects had an average of 71 total lesions of which the mean number of inflammatory lesions was 25.5 lesions and the mean number of non-inflammatory lesions was 45.1 lesions. The majority of subjects enrolled with a baseline IGA score of 3. The efficacy results at Week 12 are presented in Table 3 .
| Study 1 | Clindamycin Phosphate and Tretinoin Gel N = 476 | Clindamycin Gel N = 467 | Tretinoin Gel N = 464 | Vehicle Gel N = 242 |
| Investigator’s Global Assessment | ||||
| Percentage of subjects achieving 2-Grade Improvement | 36.3% | 26.6% | 26.1% | 20.2% |
| Percentage of subjects achieving an IGA of 0 or 1 with a 2-Grade Improvement | 33.2% | 24.0% | 22.6% | 17.8% |
| Inflammatory Lesions: Mean absolute reduction Mean percentage (%) reduction | 15.5 60.4% | 14.5 56.5% | 13.9 54.5% | 11.1 43.3% |
| Non-inflammatory Lesions: Mean absolute reduction Mean percentage (%) reduction | 23.2 51.0% | 19.5 42.9% | 22.1 47.3% | 17.0 36.0% |
| Total Lesions: Mean absolute reduction Mean percentage (%) reduction | 38.7 55.0% | 34.0 49.0% | 36.0 50.5% | 28.1 39.1% |
The safety and efficacy of clindamycin-tretinoin gel was also evaluated in two additional 12-week, multi-centered, randomized, blinded, studies in patients 12 years and older. A total of 2,219 subjects with mild-to-moderate acne vulgaris were treated once daily for 12 weeks. Of the 2,219 subjects, 634 subjects were treated with clindamycin-tretinoin gel. These studies demonstrated consistent outcomes.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Clindamycin phosphate and tretinoin gel, 1.2%/0.025% is supplied as follows:
- 30 g aluminum tubes NDC 73473-306-30
- 60 g aluminum tubes NDC 73473-306-60
Storage and Handling
- Store at 25°C (77°F); excursions permitted from 15°C to 30°C (59°F to 86°F).
- Protect from heat.
- Protect from light.
- Protect from freezing.
- Keep out of reach of children.
- Keep tube tightly closed.
Mechanism of Action
Clindamycin: [See Microbiology (12.4 ).] Tretinoin: Although the exact mode of action of tretinoin is unknown, current evidence suggests that topical tretinoin decreases cohesiveness of follicular epithelial cells with decreased microcomedone formation. Additionally, tretinoin stimulates mitotic activity and increased turnover of follicular epithelial cells causing extrusion of the comedones.