Get your patient on Clemastine Fumarate - Clemastine Fumarate syrup (Clemastine Fumarate)
Clemastine Fumarate - Clemastine Fumarate syrup prescribing information
INDICATIONS AND USAGE
Clemastine Fumarate Syrup is indicated for the relief of symptoms associated with allergic rhinitis such as sneezing, rhinorrhea, pruritus and lacrimation. Clemastine Fumarate Syrup is indicated for use in pediatric populations (age 6 years through 12) and adults (see DOSAGE AND ADMINISTRATION ).
It should be noted that clemastine is indicated for the relief of mild uncomplicated allergic skin manifestations of urticaria and angioedema at the 2 mg dosage level only.
DOSAGE AND ADMINISTRATION
DOSAGE SHOULD BE INDIVIDUALIZED ACCORDING TO THE NEEDS AND RESPONSE OF THE PATIENT.
Pediatric
Children aged 6 to 12 years
For Symptoms of Allergic Rhinitis
The starting dose is 1 teaspoonful (0.5 mg clemastine) twice daily. Since single doses of up to 2.25 mg clemastine were well tolerated by this age group, dosage may be increased as required, but not to exceed 6 teaspoonfuls daily (3 mg clemastine).
For Urticaria and Angioedema
The starting dose is 2 teaspoonfuls (1 mg clemastine) twice daily, not to exceed 6 teaspoonfuls daily (3 mg clemastine).
Adults and Children 12 Years and Over
For Symptoms of Allergic Rhinitis
The starting dose is 2 teaspoonfuls (1 mg clemastine) twice daily. Dosage may be increased as required, but not to exceed 12 teaspoonfuls daily (6 mg clemastine).
For Urticaria and Angioedema
The starting dose is 4 teaspoonfuls (2 mg clemastine) twice daily, not to exceed 12 teaspoonfuls daily (6 mg clemastine).
CONTRAINDICATIONS
Antihistamines are contraindicated in patients hypersensitive to the drug or to other antihistamines of similar chemical structure (see PRECAUTIONS- Drug Interactions ).
Antihistamines should not be used in newborn or premature infants . Because of the higher risk of antihistamines for infants generally and for newborns and prematures in particular, antihistamine therapy is contraindicated in nursing mothers (see PRECAUTIONS-Nursing Mothers ).
ADVERSE REACTIONS
The most frequent adverse reactions are italicized:
Nervous System
Sedation , sleepiness , dizziness , disturbed coordination , fatigue, confusion, restlessness, excitation, nervousness, tremor, irritability, insomnia, euphoria, paresthesia, blurred vision, diplopia, vertigo, tinnitus, acute labyrinthitis, hysteria, neuritis, convulsions.
Gastrointestinal System
Epigastric distress , anorexia, nausea, vomiting, diarrhea, constipation.
Respiratory System
Thickening of bronchial secretions , tightness of chest and wheezing, nasal stuffiness.
Cardiovascular System
Hypotension, headache, palpitations, tachycardia, extrasystoles.
Hematologic System
Hemolytic anemia, thrombocytopenia, agranulocytosis.
Genitourinary System
Urinary frequency, difficult urination, urinary retention, early menses.
General
Urticaria, drug rash, anaphylactic shock, photosensitivity, excessive perspiration, chills, dryness of mouth, nose and throat.
To report SUSPECTED ADVERSE REACTIONS, contact Allucent at 1-866-511-6754 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Additive CNS depression may occur when antihistamines are administered concomitantly with other CNS depressants including barbiturates, tranquilizers, and alcohol. Patients receiving antihistamines should be advised against the concurrent use of other CNS depressant drugs. Monoamine oxidase (MAO) inhibitors prolong and intensify the anticholinergic effects of antihistamines.
DESCRIPTION
Each 5 mL (teaspoonful) of Clemastine Fumarate Syrup for oral administration contains clemastine 0.5 mg (present as clemastine fumarate 0.67 mg). Other ingredients: Alcohol 5.5%, Flavors, Maleic Acid, Methylparaben, Propylene Glycol, Propylparaben, Purified Water, Saccharin Sodium, Sodium Hydroxide, and Sorbitol. Clemastine fumarate belongs to the benzhydryl ether group of antihistaminic compounds. The chemical name is (+)-(2R)-2-[2-[[(R)-p-Chloro-α-methyl-α-phenylbenzyl]-oxy]ethyl]-1-methylpyrrolidine fumarate and has the following structural formula:
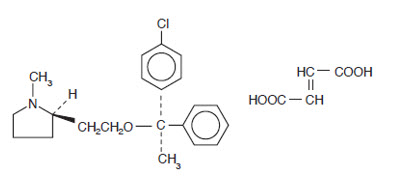 | |
| C 21 H 26 ClNO∙C 4 H 4 O 4 | M.W. 459.97 |
Clemastine fumarate occurs as a colorless to faintly yellow, odorless, crystalline powder. Clemastine Fumarate Syrup has an approximate pH of 6.2.
CLINICAL PHARMACOLOGY
Clemastine fumarate is an antihistamine with anticholinergic (drying) and sedative side effects. Antihistamines competitively antagonize various physiological effects of histamine including increased capillary permeability and dilatation, the formation of edema, the "flare" and "itch" response, and gastrointestinal and respiratory smooth muscle constriction. Within the vascular tree, H1-receptor antagonists inhibit both the vasoconstrictor and vasodilator effects of histamine. Depending on the dose, H1-receptor antagonists can produce CNS stimulation or depression.
Most antihistamines exhibit central and/or peripheral anticholinergic activity. Antihistamines act by competitively blocking H1-receptor sites. Antihistamines do not pharmacologically antagonize or chemically inactivate histamine, nor do they prevent the release of histamine.
Pharmacokinetics
Antihistamines are well-absorbed following oral administration. Chlorpheniramine maleate, clemastine fumarate, and diphenhydramine hydrochloride achieve peak blood levels within 2-5 hours following oral administration. The absorption of antihistamines is often partially delayed by the use of controlled release dosage forms. In these instances, plasma concentrations from identical doses of the immediate and controlled release dosage forms will not be similar. Tissue distribution of the antihistamines in humans has not been established. Antihistamines appear to be metabolized in the liver chiefly via mono- and didemethylation and glucuronide conjugation.
Antihistamine metabolites and small amounts of unchanged drug are excreted in the urine. Small amounts of the drugs may also be excreted in breast milk.
In normal human subjects who received histamine injections over a 24-hour period, the antihistaminic activity of clemastine reached a peak at 5-7 hours, persisted for 10-12 hours and, in some cases, for as long as 24 hours. Pharmacokinetic studies in man utilizing 3H and 14C labeled compound demonstrates that: clemastine is rapidly absorbed from the gastrointestinal tract, peak plasma concentrations are attained in 2-4 hours, and urinary excretion is the major mode of elimination.
HOW SUPPLIED
Clemastine Fumarate Syrup: clemastine 0.5 mg/5 mL (present as clemastine fumarate 0.67 mg/5 mL). A clear, colorless liquid with a citrus flavor (passion fruit), in 120 mL bottle.
120 mL bottle (NDC 64950-324-12)
Store at controlled room temperature, between 20° and 25°C (68° and 77°F) (see USP).