Chloroquine Phosphate - Chloroquine Phosphate tablet, Coated prescribing information
INDICATIONS AND USAGE
Chloroquine phosphate tablets are indicated for the:
- Treatment of uncomplicated malaria due to susceptible strains of P. falciparum, P. malariae, P. ovale, and P. vivax .
- Prophylaxis of malaria in geographic areas where resistance to chloroquine is not present.
- Treatment of extraintestinal amebiasis.
Chloroquine phosphate tablets do not prevent relapses in patients with vivax or ovale malaria because it is not effective against exoerythrocytic forms of the parasites.
Limitations of Use in Malaria:
- Do not use Chloroquine phosphate tablets for the treatment of complicated malaria (high-grade parasitemia and/or complications e.g., cerebral malaria or acute renal failure).
- Do not use Chloroquine phosphate tablets for malaria prophylaxis in areas where chloroquine resistance occurs, Resistance to Chloroquine phosphate tablets is widespread in P. falciparum , and is reported in P. vivax (see WARNINGS ).
- Concomitant therapy with an 8-aminoquinoline drug is necessary for treatment of the hypnozoite liver stage forms of P. vivax and P. ovale (see DOSAGE AND ADMINISTRATION ).
DOSAGE & ADMINISTRATION
The dosage of chloroquine phosphate is often expressed in terms of equivalent chloroquine base. Each 500 mg tablet of Chloroquine phosphate contains the equivalent of 300 mg chloroquine base. In infants and children the dosage is preferably calculated by body weight.
Prophylaxis against chloroquine-sensitive Plasmodium species
Adult Dose: The dosage for prophylaxis is 500 mg (= 300 mg base) administered once per week on exactly the same day of each week.
Pediatric Dose: The dosage for prophylaxis is 5 mg calculated as base, per kg of body weight, administered once per week on exactly the same day of each week. The pediatric dose should never exceed the adult dose regardless of weight.
If circumstances permit, suppressive therapy should begin two weeks prior to exposure. However, failing this in adults, an initial double (loading) dose of 1 g (= 600 mg base), or in children 10 mg base/kg may be taken in two divided doses, six hours apart. The suppressive therapy should be continued for eight weeks after leaving the endemic area.
Treatment of uncomplicated malaria due to chloroquine-sensitive Plasmodium species
Adults: An initial dose of 1 g salt (= 600 mg base) followed by an additional 500 mg (= 300 mg base) after six to eight hours and a single dose of 500 mg (= 300 mg base) on each of two consecutive days. This represents a total dose of 2.5 g chloroquine phosphate or 1.5 g base in three days.
Infants and Children: In infants and children, the recommended dose is 10 mg base/kg followed by 5 mg based/kg at 6, 24 and 36 hours (total dose 25 mg based/kg). The pediatric dose should never exceed the adult dose regardless of weight.
The dosage for adults of low body weight and for infants and children should be determined as follows:
First dose: 10 mg base per kg (but not exceeding a single dose of 600 mg base).
Second dose: (6 hours after first dose) 5 mg base per kg (but not exceeding a single dose of 300 mg base).
Third dose: (24 hours after first dose) 5 mg base per kg.
Fourth dose: (36 hours after first dose) 5 mg base per kg.
P. vivax and P. ovale : Concomitant therapy with an 8-aminoquinoline compound is necessary for treatment of the hypnozoite liver stage forms of the parasites.
Extraintestinal Amebiasis: Adults Dosage: 1 g salt (600 mg base) daily for two days, followed by 500 mg (300 mg base) daily for at least two to three weeks. Treatment is usually combined with an effective intestinal amebicide.
Geriatric Use
See PRECAUTIONS , Geriatric Use .
CONTRAINDICATIONS
Use of Chloroquine phosphate tablets for indications other than acute malaria is contraindicated in the presence of retinal or visual field changes of any etiology.
Use of Chloroquine phosphate tablets is contraindicated in patients with known hypersensitivity to 4- aminoquinoline compounds.
ADVERSE REACTIONS
The following adverse reactions have been identified during post-approval use of chloroquine or other 4-aminoqunoline compounds. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Ocular disorders: Maculopathy and macular degeneration have been reported and may be irreversible. Irreversible retinopathy with retinal pigmentation changes (bull’s eye appearance) and visual field defects (paracentral scotomas) in patients receiving long-term or high-dosage 4-aminoquinoline therapy have been reported (see WARNINGS ). Visual disturbances (blurring of vision and difficulty of focusing or accommodation); nyctalopia; scotomatous vision with field defects of paracentral, pericentral ring types, and typically temporal scotomas (e.g., difficulty in reading with words tending to disappear, seeing half an object, misty vision, and fog before the eyes) have been reported. Reversible corneal opacities have also been reported.
Immune system disorders: Urticaria, anaphylactic reaction including angioedema.
Ear and labyrinth disorders: Nerve type deafness; tinnitus, reduced hearing in patients with preexisting auditory damage.
Musculoskeletal and connective tissue-disorders: Sensorimotor disorders, skeletal muscle myopathy or neuromyopathy leading to progressive weakness and atrophy of proximal muscle groups, depression of tendon reflexes and abnormal nerve conduction.
Gastrointestinal disorders: Hepatitis, increased liver enzymes, anorexia, nausea, vomiting, diarrhea, abdominal cramps.
Skin and subcutaneous tissue disorders: Erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis, exfoliative dermatitis. Pleomorphic skin eruptions, skin and mucosal pigmentary changes; lichen planus-like eruptions, pruritus,; drug rash with eosinophilia and systemic symptoms (DRESS syndrome); photosensitivity and hair loss and bleaching of hair pigment.
Blood and lymphatic system disorders: Pancytopenia, aplastic anemia, reversible agranulocytosis, thrombocytopenia and neutropenia. Hemolytic anemia in G6PD deficient patients (see PRECAUTIONS ).
Nervous system disorders: Convulsions, mild and transient headache, polyneuropathy, acute extrapyramidal disorders (such as dystonia, dyskinesia, tongue protrusion, torticollis) (see WARNINGS and OVERDOSAGE ).
Neuropsychiatric disorders: Neuropsychiatric changes including psychosis, delirium, anxiety, agitation, insomnia, confusion, hallucinations, personality changes, depression, suicidal behavior, suicidal ideation, affect/emotional lability, irritability, nervousness, nightmares/sleep terrors, depression, delusions, paranoia, and mania.
Cardiac disorders: Hypotension, electrocardiographic changes (particularly, inversion or depression of the T-wave with widening of the QRS complex), and cardiomyopathy (which may result in cardiac failure and in some cases a fatal outcome).
Cardiac arrhythmias, conduction disorders such as bundle branch block / atrio-ventricular block, QT interval prolongation, torsade de pointes, ventricular tachycardia and ventricular fibrillation have been reported with therapeutic doses of chloroquine as well as with overdose. The risk is greater if chloroquine is administered at high doses. Fatal cases have been reported (see WARNINGS, Cardiac Effects and OVERDOSAGE ).
Metabolic and Nutritional disorders: Hypoglycemia (see WARNINGS ).
To report SUSPECTED ADVERSE REACTIONS, contact Chartwell RX, LLC. at 1-845-262-1683, or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
Chloroquine Phosphate Tablet, USP, is a 4-aminoquinoline compound for oral administration. It is a white, odorless, bitter tasting, crystalline substance, freely soluble in water.
Chloroquine phosphate tablet is an antimalarial and amebicidal drug.
Chemically, it is 7-chloro-4-[[4-(diethylamino)-1-methylbutyl]amino]quinoline phosphate (1:2) and has the following structural formula:
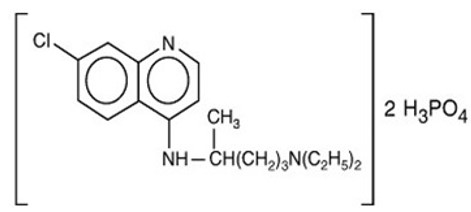
Each tablet contains 500 mg of chloroquine phosphate USP, equivalent to 300 mg chloroquine base.
Inactive Ingredients:
microcrystalline cellulose, dibasic calcium phosphate, pregelatinzed starch, sodium starch glycolate, colloidal silicon dioxide, magnesium stearate, polyethylene glycol, titanium dioxide, hydroxy propyl methyl cellulose and polysorbate 80.
CLINICAL PHARMACOLOGY
Chloroquine is rapidly and almost completely absorbed from the gastrointestinal tract, and only a small proportion of the administered dose is found in the stools. Approximately 55% of the drug in the plasma is bound to nondiffusible plasma constituents. Excretion of chloroquine is quite slow but is increased by acidification of the urine. Chloroquine is deposited in the tissues in considerable amounts. In animals, from 200 to 700 times the plasma concentration may be found in the liver, spleen, kidney, and lung; leukocytes also concentrate the drug. The brain and spinal cord, in contrast, contain only 10 to 30 times the amount present in plasma.
Chloroquine undergoes appreciable degradation in the body. The main metabolite is desethylchloroquine, which accounts for one fourth of the total material appearing in the urine; bisdesethylchloroquine, a carboxylic acid derivative, and other metabolic products as yet uncharacterized are found in small amounts. Slightly more than half of the urinary drug products can be accounted for as unchanged chloroquine.
Cardiac Electrophysiology
QTc interval prolongation was studied in a randomized, placebo-controlled parallel trial in 116 healthy subjects who received either chloroquine (1000 mg) alone or in combination with oral azithromycin (500 mg, 1000 mg, and 1500 mg once daily). Co-administration of azithromycin increased the QTc interval in a dose- and concentration- dependent manner. In comparison to chloroquine alone, the maximum mean (95% upper confidence bound) increases in QTcF were 5 ms, 7 (12) ms and 9 (14) ms with the co-administration of 500 mg, 1000 mg and 1500 mg azithromycin, respectively.
Microbiology
Mechanism of Action: Chloroquine, a 4-aminoquinoline, is an anti-protozoal agent. The precise mechanism by which chloroquine exhibits activity is not known. Chloroquine, may exert its effect against Plasmodium species by concentrating in the acid vesicles of the parasite and by inhibiting polymerization of heme. It can also inhibit certain enzymes by its interaction with DNA.
Activity in Vitro and in Clinical Infections: Chloroquine is active against the erythrocytic forms of susceptible strains of Plasmodium falciparum, Plasmodium malariae, Plasmodium ovale , and Plasmodium vivax . Chloroquine is not active against the gametocytes and the exoerythrocytic forms including the hypnozoite stage ( P. vivax and P. ovale ) of the Plasmodium parasites.
In vitro studies with Chloroquine demonstrated that it is active against the trophozoites of Entamoeba histolytica.
Drug Resistance: Resistance of Plasmodium parasites to chloroquine is widespread (see INDICATIONS AND USAGE, Limitations of Use in Malaria and WARNINGS ).
Plasmodium parasites exhibiting reduced susceptibility to hydroxychloroquine also show reduced susceptibility to chloroquine.
Patients in whom chloroquine or hydroxychloroquine have failed to prevent or cure clinical malaria or parasitemia, or patients who acquired malaria in a geographic area where chloroquine resistance is known to occur should be treated with another form of antimalarial therapy (see WARNINGS and INDICATIONS AND USAGE, Limitations of Use ).
HOW SUPPLIED
Chloroquine Phosphate Tablets, USP contains 500 mg chloroquine phosphate USP, equivalent to 300 mg of chloroquine base
Bottle of 25 (NDC 62135-717-26)
White, round, biconvex film-coated tablets, with debossing of ‘CP 500’ on one side and plain surface on the other side.
Dispense in tight, light-resistant container as defined in the USP/NF.
Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Protect from light and moisture.