Get your patient on Chlordiazepoxide Hydrochloride And Clidinium Bromide - Chlordiazepoxide Hydrochloride, Clidinium Bromide capsule (Chlordiazepoxide Hydrochloride, Clidinium Bromide)
Chlordiazepoxide Hydrochloride And Clidinium Bromide - Chlordiazepoxide Hydrochloride, Clidinium Bromide capsule prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS
Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death (see WARNINGS and PRECAUTIONS).
- Reserve concomitant prescribing of these drugs for use in patients for whom alternative treatment options are inadequate.
- Limit dosages and durations to the minimum required.
- Follow patients for signs and symptoms of respiratory depression and sedation.
INDICATIONS AND USAGE
Chlordiazepoxide Hydrochloride and Clidinium Bromide is indicated to control emotional and somatic factors in gastrointestinal disorders. Chlordiazepoxide Hydrochloride and Clidinium Bromide may also be used as adjunctive therapy in the treatment of peptic ulcer and in the treatment of the irritable bowel syndrome (irritable colon, spastic colon, mucous colitis) and acute enterocolitis.
DOSAGE AND ADMINISTRATION
Because of the varied individual responses to tranquilizers and anticholinergics, the optimum dosage of Chlordiazepoxide Hydrochloride and Clidinium Bromide varies with the diagnosis and response of the individual patient. The dosage therefore should be individualized for maximum beneficial effects. The usual maintenance dose is 1 or 2 capsules, 3 or 4 times a day administered before meals and at bedtime.
Geriatric Dosing
Dosage should be limited to the smallest effective amount to preclude the development of ataxia, oversedation or confusion. The initial dose should not exceed 2 Chlordiazepoxide Hydrochloride and Clidinium Bromide capsules per day, to be increased gradually as needed and tolerated.
CONTRAINDICATIONS
Chlordiazepoxide Hydrochloride and Clidinium Bromide is contraindicated in the presence of glaucoma (since the anticholinergic component may produce some degree of mydriasis) and in patients with prostatic hypertrophy and benign bladder neck obstruction. It is contraindicated in patients with known hypersensitivity to chlordiazepoxide hydrochloride and/or clidinium bromide.
ADVERSE REACTIONS
No side effects or manifestation not seen with either compound alone have been reported with the administration of Chlordiazepoxide Hydrochloride and Clidinium Bromide. However, since the Chlordiazepoxide Hydrochloride and Clidinium Bromide capsule contains chlordiazepoxide hydrochloride and clidinium bromide, the possibility of untoward effects which may be seen with either of these two compounds cannot be excluded.
When chlordiazepoxide hydrochloride has been used alone the necessity of discontinuing therapy because of undesirable effects has been rare. Drowsiness, ataxia and confusion have been reported in some patients – particularly the elderly and debilitated. While these effects can be avoided in almost all instances by proper dosage adjustment, they have occasionally been observed at the lower dosage ranges. In a few instances syncope has been reported.
Other adverse reactions reported during therapy with chlordiazepoxide hydrochloride include isolated instances of skin eruptions, edema, minor menstrual irregularities, nausea and constipation, extrapyramidal symptoms, as well as increased and decreased libido. Such side effects have been infrequent and are generally controlled with reduction of dosage. Changes in EEG patterns (low-voltage fast activity) have been observed in patients during and after chlordiazepoxide hydrochloride treatment.
Blood dyscrasias, including agranulocytosis, jaundice and hepatic dysfunction have occasionally been reported during therapy with chlordiazepoxide hydrochloride. When chlordiazepoxide hydrochloride treatment is protracted, periodic blood counts and liver function tests are advisable.
Adverse effects reported with use of Chlordiazepoxide Hydrochloride and Clidinium Bromide are those typical of anticholinergic agents, i.e., dryness of the mouth, blurring of vision, urinary hesitancy and constipation. Constipation has occurred most often when Chlordiazepoxide Hydrochloride and Clidinium Bromide therapy has been combined with other spasmolytic agents and/or low residue diet.
To report SUSPECTED ADVERSE REACTIONS, contact Misemer Pharmaceuticals, Inc at 1-732-762-6577 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
DESCRIPTION
Chlordiazepoxide Hydrochloride and Clidinium Bromide combines in a single capsule formulation the antianxiety action of chlordiazepoxide hydrochloride and the anticholinergic/spasmolytic effects of clidinium bromide.
Each Chlordiazepoxide Hydrochloride and Clidinium Bromide capsule contains the active ingredients 5 mg chlordiazepoxide hydrochloride and 2.5 mg clidinium bromide.
Each capsule also contains the inactive ingredients lactose monohydrate, pregelatinized starch, talc. Gelatin capsule shells may contain titanium dioxide with the following dye systems: D&C Yellow #10, FD&C Green #3 and gelatin.
Chlordiazepoxide hydrochloride is a versatile, therapeutic agent of proven value for the relief of anxiety and tension. It is indicated when anxiety, tension or apprehension are significant components of the clinical profile. It is among the safer of the effective psychopharmacologic compounds.
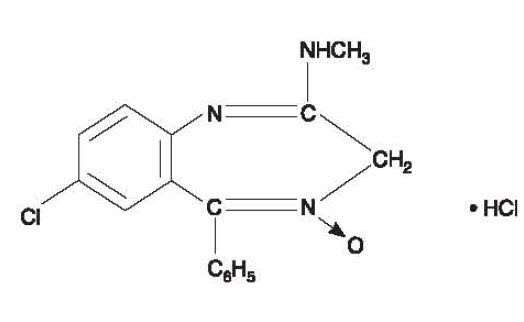
Chlordiazepoxide hydrochloride is 7-chloro-2-methylamino-5-phenyl-3H-1,4-benzodiazepine 4-oxide hydrochloride. A colorless, crystalline substance, it is soluble in water. It is unstable in solution and the powder must be protected from light. The molecular weight is 336.22. The structural formula of chlordiazepoxide hydrochloride is as follows:
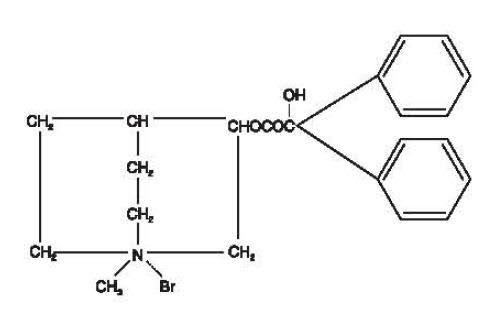
Clidinium bromide is a synthetic anticholinergic agent which has been shown in experimental and clinical studies to have a pronounced antispasmodic and antisecretory effect on the gastrointestinal tract. Structurally clidinium bromide is:
HOW SUPPLIED
Chlordiazepoxide Hydrochloride and Clidinium Bromide capsules are available in light green opaque capsules, each containing 5 mg chlordiazepoxide hydrochloride and 2.5 mg clidinium bromide - bottles of 100 capsules (NDC 0276-0500-10), with “VP302” imprinted on the cap of capsule.
Store at 25°C (77°F): excursions permitted to 15° to 30°C (59° to 86°F).
Keep out of reach of children. Dispense in a tight, light-resistant container as defined in the USP/NF.
Manufactured for:
Mismer Pharmaceuticals Inc Ripley, MS 38663.
D-2020