Chlordiazepoxide And Amitriptyline Hydrochloride - Chlordiazepoxide And Amitriptyline Hydrochloride tablet, Film Coated prescribing information
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; DEPENDENCE AND WITHDRAWAL REACTIONS; and SUICIDAL THOUGHTS AND BEHAVIORS
- Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of these drugs for patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation (see WARNINGS and PRECAUTIONS ).
- The use of benzodiazepines, including chlordiazepoxide and amitriptyline hydrochloride tablets, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing chlordiazepoxide and amitriptyline hydrochloride tablets and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction (see WARNINGS ).
- The continued use of benzodiazepines, including chlordiazepoxide and amitriptyline hydrochloride tablets, may lead to clinically significant physical dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Abrupt discontinuation or rapid dosage reduction of chlordiazepoxide and amitriptyline hydrochloride tablets after continued use may precipitate acute withdrawal reactions, which can be life-threatening. To reduce the risk of withdrawal reactions, use a gradual taper to discontinue chlordiazepoxide and amitriptyline hydrochloride tablets or reduce the dosage (see DOSAGE AND ADMINISTRATION and WARNINGS ).
- Antidepressants increased the risk of suicidal thoughts and behaviors in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors (see WARNINGS ). Chlordiazepoxide and amitriptyline hydrochloride tablets are not approved for use in pediatric patients (see PRECAUTIONS ).
INDICATIONS AND USAGE
Chlordiazepoxide and amitriptyline hydrochloride tablets are indicated for the treatment of patients with moderate to severe depression associated with moderate to severe anxiety.
The therapeutic response to chlordiazepoxide and amitriptyline hydrochloride tablets occurs earlier and with fewer treatment failures than when either amitriptyline or chlordiazepoxide is used alone.
Symptoms likely to respond in the first week of treatment include: insomnia, feelings of guilt or worthlessness, agitation, psychic and somatic anxiety, suicidal ideation and anorexia.
DOSAGE AND ADMINISTRATION
Optimum dosage varies with the severity of the symptoms and the response of the individual patient. When a satisfactory response is obtained, dosage should be reduced to the smallest amount needed to maintain the remission. The larger portion of the total daily dose may be taken at bedtime. In some patients, a single dose at bedtime may be sufficient. In general, lower dosages are recommended for elderly patients.
Chlordiazepoxide and amitriptyline hydrochloride tablets 10 mg/25 mg are recommended in an initial dosage of 3 or 4 tablets daily in divided doses; this may be increased to 6 tablets daily as required. Some patients respond to smaller doses and can be maintained on 2 tablets daily.
Chlordiazepoxide and amitriptyline hydrochloride tablets 5 mg/12.5 mg in an initial dosage of 3 or 4 tablets daily in divided doses may be satisfactory in patients who do not tolerate higher doses.
Screen for Bipolar Disorder Prior to Starting Chlordiazepoxide and Amitriptyline Hydrochloride Tablets
Prior to initiating treatment with chlordiazepoxide and amitriptyline hydrochloride tablets or another antidepressant, screen patients for a personal or family history of bipolar disorder, mania, or hypomania (see WARNINGS: Activation of Mania or Hypomania ).
Discontinuation or Dosage Reduction of Chlordiazepoxide and Amitriptyline Hydrochloride Tablets
To reduce the risk of withdrawal reactions, use a gradual taper to discontinue chlordiazepoxide and amitriptyline hydrochloride tablets or reduce the dosage. If a patient develops withdrawal reactions, consider pausing the taper or increasing the dosage to the previous tapered dosage level. Subsequently decrease the dosage more slowly (see WARNINGS: Dependence and Withdrawal Reactions and DRUG ABUSE AND DEPENDENCE: Dependence ).
CONTRAINDICATIONS
Chlordiazepoxide and amitriptyline hydrochloride tablets are contraindicated in patients with hypersensitivity to either benzodiazepines or tricyclic antidepressants. It should not be given concomitantly with a monoamine oxidase inhibitor. Hyperpyretic crises, severe convulsions and deaths have occurred in patients receiving a tricyclic antidepressant and a monoamine oxidase inhibitor simultaneously. When it is desired to replace a monoamine oxidase inhibitor with chlordiazepoxide and amitriptyline hydrochloride tablets, a minimum of 14 days should be allowed to elapse after the former is discontinued. Chlordiazepoxide and amitriptyline hydrochloride tablets should then be initiated cautiously with gradual increase in dosage until optimum response is achieved.
This drug is contraindicated during the acute recovery phase following myocardial infarction.
ADVERSE REACTIONS
Adverse reactions to chlordiazepoxide and amitriptyline hydrochloride are those associated with the use of either component alone. Most frequently reported were drowsiness, dry mouth, constipation, blurred vision, dizziness and bloating. Other side effects occurring less commonly included vivid dreams, impotence, tremor, confusion and nasal congestion. Many symptoms common to the depressive state, such as anorexia, fatigue, weakness, restlessness and lethargy, have been reported as side effects of treatment with both chlordiazepoxide and amitriptyline hydrochloride tablets and amitriptyline.
Granulocytopenia, jaundice and hepatic dysfunction of uncertain etiology have also been observed rarely with chlordiazepoxide and amitriptyline hydrochloride tablets. When treatment with chlordiazepoxide and amitriptyline hydrochloride tablets is prolonged, periodic blood counts and liver function tests are advisable.
Note
Included in the listing which follows are adverse reactions which have not been reported with chlordiazepoxide and amitriptyline hydrochloride. However, they are included because they have been reported during therapy with one or both of the components or closely related drugs.
Cardiovascular: Hypotension, hypertension, tachycardia, palpitations, myocardial infarction, arrhythmias, heart block, stroke.
Psychiatric: Euphoria, apprehension, poor concentration, delusions, hallucinations, hypomania and increased or decreased libido.
Neurologic: Incoordination, ataxia, numbness, tingling and paresthesias of the extremities, extrapyramidal symptoms, syncope, changes in EEG patterns.
Anticholinergic: Disturbance of accommodation, paralytic ileus, urinary retention, dilatation of urinary tract.
Allergic: Skin rash, urticaria, photosensitization, edema of face and tongue, pruritus, drug reaction with eosinophilia and systemic symptoms (DRESS).
Hematologic: Bone marrow depression including agranulocytosis, eosinophilia, purpura, thrombocytopenia.
Gastrointestinal: Nausea, epigastric distress, vomiting, anorexia, stomatitis, peculiar taste, diarrhea, black tongue.
Endocrine: Testicular swelling and gynecomastia in the male, breast enlargement, galactorrhea and minor menstrual irregularities in the female, elevation and lowering of blood sugar levels, and syndrome of inappropriate ADH (antidiuretic hormone) secretion.
Other: Headache, weight gain or loss, increased perspiration, urinary frequency, mydriasis, jaundice, alopecia, parotid swelling, hyponatremia.
DESCRIPTION
Chlordiazepoxide and amitriptyline hydrochloride tablets, USP combine for oral administration, chlordiazepoxide, an agent for the relief of anxiety and tension, and amitriptyline, an antidepressant.
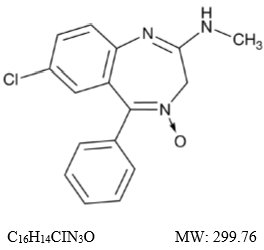
Chlordiazepoxide, USP is a benzodiazepine with the formula 7-chloro-2-(methylamino)-5-phenyl-3 H -1,4-benzodiazepine-4-oxide. It is a yellow crystalline powder and is insoluble in water. The chemical structure is:
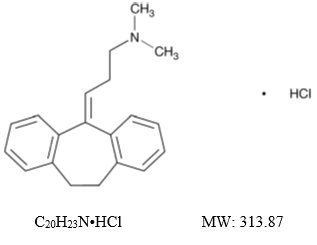
Amitriptyline hydrochloride, USP is a dibenzocycloheptadiene derivative. The formula is 10,11-dihydro- N,N -dimethyl-5 H -dibenzo[a,d]cycloheptene-Δ 5 ,γ-propylamine hydrochloride. It is a white or practically white crystalline powder that is freely soluble in water. The chemical structure is:
Each film-coated tablet for oral administration contains 5 mg of chlordiazepoxide and 14 mg of amitriptyline hydrochloride equivalent to 12.5 mg of amitriptyline or 10 mg of chlordiazepoxide and 27.98 mg of amitriptyline hydrochloride equivalent to 25 mg of amitriptyline. Each tablet contains the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, hydroxypropyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, pregelatinized starch (corn), sodium lauryl sulfate and titanium dioxide. In addition, the 5 mg/12.5 mg tablets also contain D&C Yellow No. 10 Aluminum Lake, FD&C Blue No. 1 Aluminum Lake and FD&C Yellow No. 6 Aluminum Lake and the 10 mg/25 mg tablets also contain polydextrose and triacetin.
CLINICAL PHARMACOLOGY
Both components of chlordiazepoxide and amitriptyline hydrochloride tablets exert their action in the central nervous system. Extensive studies with chlordiazepoxide in many animal species suggest action in the limbic system. Recent evidence indicates that the limbic system is involved in emotional response. Taming action was observed in some species. The mechanism of action of amitriptyline in man is not known, but the drug appears to interfere with the reuptake of norepinephrine into adrenergic nerve endings. This action may prolong the sympathetic activity of biogenic amines.
HOW SUPPLIED
Chlordiazepoxide and Amitriptyline Hydrochloride Tablets, USP are available containing 5 mg of chlordiazepoxide, USP and 14 mg of amitriptyline hydrochloride, USP equivalent to 12.5 mg of amitriptyline or 10 mg of chlordiazepoxide, USP and 27.98 mg of amitriptyline hydrochloride, USP equivalent to 25 mg of amitriptyline.
The 5 mg/12.5 mg tablets are green, film-coated, round, unscored tablets debossed with MYLAN on one side of the tablet and 211 on the other side. They are available as follows:
NDC 0378-0211-01 bottles of 100 tablets
NDC 0378-0211-05 bottles of 500 tablets
The 10 mg/25 mg tablets are white, film-coated, round, unscored tablets debossed with MYLAN on one side of the tablet and 277 on the other side. They are available as follows:
NDC 0378-0277-01 bottles of 100 tablets
NDC 0378-0277-05 bottles of 500 tablets
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Store in a dry place.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
PHARMACIST: Dispense a Medication Guide with each prescription.