Ceretec - Technetium Tc-99m Exametazime injection, Powder, Lyophilized, For Solution prescribing information
INDICATIONS AND USAGE
Technetium Tc99m exametazime scintigraphy may be useful as an adjunct in the detection of altered regional cerebral perfusion in stroke.
Tc99m exametazime is indicated for leukocyte labeled scintigraphy as an adjunct in the localization of intra-abdominal infection and inflammatory bowel disease.
DOSAGE AND ADMINISTRATION
Tc99m labeled leukocytes for adjunctive localization of intra-abdominal infection or inflammation.
The normal adult (70 kg) dose is 0.259-0.925 GBq (7-25 mCi) as Tc99m labeled leukocytes by intravenous injection. Optimal planar imaging is between 2-4 hours.
Cerebral Scintigraphy
The recommended dose range for i.v. administration, of reconstituted sodium pertechnetate Tc99m exametazime in the average adult (70 kg) is 370-740 MBq (10-20 mCi).
Dynamic imaging may be performed between 0 to 10 minutes following injection. Static imaging may be performed from 15 minutes up to 6 hours after injection.
CONTRAINDICATIONS
None known.
ADVERSE REACTIONS
Rash with generalized erythema, facial edema and fever has been reported in less than 1% of patients. A transient increase in blood pressure was seen in 8% of patients.
DESCRIPTION
The Ceretec kit is supplied as a pack of 5 vials for use in the preparation of a technetium Tc99m exametazime intravenous injection as a diagnostic radiopharmaceutical for use as an adjunct in the detection of altered regional cerebral perfusion and for the radiolabeling of autologous leukocytes. Each vial of Ceretec contains a pre-dispensed sterile, non-pyrogenic, lyophilized mixture of 0.5 mg exametazime [(RR,SS)-4.8-diaza-3,6,6,9-tetramethylundecane-2, 10-dione bisoxime], 7.6 µg stannous chloride dihydrate (minimum stannous tin 0.6 µg; maximum total stannous and stannic tin 4.0 µg per vial) and 4.5 mg sodium chloride, sealed under nitrogen atmosphere with a rubber closure. The product contains no antimicrobial preservative.
Prior to publication of the USAN, exametazime was formerly known as hexamethylpropylene amine oxime (HM-PAO). The name HM-PAO appears in many publications.
The structural formula of exametazime is:
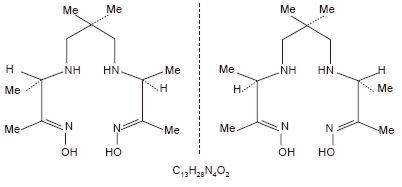
When sterile pyrogen-free sodium pertechnetate Tc99m in isotonic saline is added to the vial of Ceretec, a Tc99m complex of exametazime is formed.
Administration is by intravenous injection for diagnostic use.
Physical Characteristics
Technetium Tc99m decays by isomeric transition with a physical half-life of 6.03 hours. (1) Photons that are useful for imaging studies are listed in Table 1.
| Radiation | Mean %/ Disintegration | Mean Energy (keV) |
|---|---|---|
| (1) Dillman, L.T. and Von der Lage, F.C. Radionuclide decay schemes and nuclear parameters for use in radiation-dose estimation. MIRD Phamphlet No. 10, p. 62, 1975. | ||
| Gamma 2 | 87.87 | 140.5 |
External Radiation
The specific gamma ray constant for technetium Tc99m is 206 microCoulomb kg-1/37 MBq-h, (0.8 R/millicurie-h) at 1 cm. The first half-value thickness of lead (Pb) for technetium Tc99m is 0.2 mm. A range of values for the relative attenuation of the radiation emitted by this radionuclide that results from interposition of various thicknesses of Pb is shown in Table 2. For example, the use of a 2.7 mm thickness of Pb will decrease the external radiation exposure by a factor of 1,000.
| Shield Thickness (Pb) mm | Coefficient of Attenuation |
|---|---|
| 0.2 | 0.5 |
| 0.95 | 10 –1 |
| 1.8 | 10 –2 |
| 2.7 | 10 –3 |
| 3.6 | 10 –4 |
| 4.5 | 10 –5 |
To correct for physical decay of this radionuclide, the fractions that remain at selected intervals relative to the time of calibration are shown in Table 3.
| Hours | Fraction Remaining | Hours | Fraction Remaining |
|---|---|---|---|
| 0 Calibration time (time of preparation) | 1.000 | 7 | 0.447 |
| 1 | 0.891 | 8 | 0.399 |
| 2 | 0.795 | 9 | 0.355 |
| 3 | 0.708 | 10 | 0.317 |
| 4 | 0.631 | 11 | 0.282 |
| 5 | 0.563 | 12 | 0.252 |
| 6 | 0.502 | 24 | 0.063 |
CLINICAL PHARMACOLOGY
General
When technetium Tc99m pertechnetate is added to exametazime in the presence of stannous reductant, a lipophilic technetium Tc99m complex is formed. This lipophilic complex is the active moiety. It converts at approximately 12%/hour to less lipophilic species. When the secondary complex is separated from the lipophilic species, it is unable to cross the blood-brain-barrier. The useful life of the reconstituted agent is limited to 30 minutes.
Pharmacokinetics
Studies in normal volunteers have shown that the technetium Tc99m complex of the RR,SS(d,l) diastereoisomer of exametazime is rapidly cleared from the blood after intravenous injection. Uptake in the brain reaches a maximum of 3.5-7.0% of the injected dose within one minute of injection. Up to 15% of the activity is eliminated from the brain by 2 minutes post injection, after which little activity is lost for the following 24 hours except by physical decay of technetium Tc99m. The activity not associated with the brain is widely distributed throughout the body, particularly in muscle and soft tissue. About 30% of the injected dose is found in the gastrointestinal tract immediately after injection and about 50% of this is excreted through the intestinal tract over 48 hours. Also, about 40% of the injected dose is excreted through the kidneys and urine over the 48 hours after injection.
Leukocyte
The lipophilic Tc99m exametazime complex is taken up by leukocytes, and selectively retained in neutrophils. Label elution rate is up to 10% in the first hour.
Pharmacodynamics
Tc99m-labeled leukocyte: Based upon in vivo recovery and very low lung and liver uptake, the labeled leukocytes are still functional. Following reinjection of the Tc99m labeled leukocytes the circulating granulocyte activity as a percentage of labeled granulocyte activity at 40 minutes after injection gave a mean of 37% (range 10-47%).
During the first hour following injection of Tc99m labeled leukocytes, activity is seen in the lungs, liver, spleen, blood pool, bone marrow and the bladder. The kidneys (parenchyma and/or renal pelvis) and gall bladder may also be visualized. Over the first 1-6 hours, the Tc99m is visualized in the bowel. At 24 hours post-injection substantial colonic activity is seen. The normal areas visualized in earlier scans are still visible.
CLINICAL TRIALS
Two clinical trials were performed in a total of 88 patients who had suspected intra-abdominal infection or inflammation. Subjects received both Tc99m labeled leukocytes and In-111 labeled leukocytes. Images were obtained at 2 and 30 minutes and at 2 and 4 hours and 24 hours. In two other clinical trials, in a total of 127 patients with suspected abdominal inflammation or infection received Tc99m labeled leukocytes. Imaging was at 24 hours in one study and at 1, 3 and 24 hours in the other. In all 4 studies, images were blindly evaluated and the findings were confirmed by surgery, biopsy or other clinical data.
Based on the above 4 studies, between 2 to 4 hours Tc99m labeled leukocytes had 95-100% sensitivity and 62-85% specificity with similar numbers of false positive and false negative findings. The value of the 24 hour Tc99m labeled leukocyte images is inconsistent. In all studies the false positive and false negatives relate to the bowel background, the location of the site of infection/inflammation and whether or not it is contiguous with the bowel. The 24 hour films should be interpreted with great caution because of a high bowel background; false negatives were noted in both Tc99m and In-111 labeled leukocytes.
Other studies suggest that the interpretation of the images could be affected by the presence of tumors, infarction and peritonitis, etc. Liver abscess may be missed because of the bowel background. Caution should be exercised in making the final diagnosis.
HOW SUPPLIED
The kit comprises 5 individual vials of sterile, non-pyrogenic, freeze-dried mixture of exametazime, stannous chloride dihydrate and sodium chloride, 5 radiation labels, 5 radiochemical purity worksheets, 5 labeling efficiency worksheets, 1 leukocyte labeling schematic and 1 package insert.
NDC 17156-022-05
Storage
Store the kit at 15°-25°C (59°-77°F).
Store the formulated drug for up to 30 minutes at 20°-25°C (68°-77°F) using appropriate radiation shielding.
This reagent kit is approved for use by persons licensed by the Illinois Emergency Management Agency pursuant to 32 Ill. Code Adm. Section, Section 330.260(a) and 335.4010 or under equivalent licenses of the U.S. Nuclear Regulatory Commission, or an Agreement State.