Get your patient on Cefprozil - Cefprozil tablet, Film Coated (Cefprozil)
Cefprozil - Cefprozil tablet, Film Coated prescribing information
INDICATIONS AND USAGE
To reduce the development of drug-resistant bacteria and maintain the effectiveness of cefprozil and other antibacterial drugs, cefprozil should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy. Cefprozil tablets are indicated for the treatment of patients with mild to moderate infections caused by susceptible strains of the designated microorganisms in the conditions listed below: UPPER RESPIRATORY TRACT Pharyngitis/tonsillitis caused by Streptococcus pyogenes . NOTE: The usual drug of choice in the treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever, is penicillin given by the intramuscular route. Cefprozil is generally effective in the eradication of Streptococcus pyogenes from the nasopharynx; however, substantial data establishing the efficacy of cefprozil in the subsequent prevention of rheumatic fever are not available at present. Otitis Media caused by Streptococcus pneumoniae , Haemophilus influenzae (including β-lactamase-producing strains), and Moraxella (Branhamella) catarrhalis (including β-lactamase-producing strains). (See CLINICAL STUDIES .) NOTE: In the treatment of otitis media due to β-lactamase producing organisms, cefprozil had bacteriologic eradication rates somewhat lower than those observed with a product containing a specific β-lactamase inhibitor. In considering the use of cefprozil, lower overall eradication rates should be balanced against the susceptibility patterns of the common microbes in a given geographic area and the increased potential for toxicity with products containing β-lactamase inhibitors. Acute Sinusitis caused by Streptococcus pneumoniae , Haemophilus influenzae (including β-lactamase-producing strains), and Moraxella (Branhamella) catarrhalis (including β-lactamase-producing strains). LOWER RESPIRATORY TRACT Acute Bacterial Exacerbation of Chronic Bronchitis caused by Streptococcus pneumoniae , Haemophilus influenzae (including β-lactamase-producing strains), and Moraxella (Branhamella) catarrhalis (including β-lactamase-producing strains). SKIN AND SKIN STRUCTURE Uncomplicated Skin and Skin-Structure Infections caused by Staphylococcus aureus (including penicillinase-producing strains) and Streptococcus pyogenes . Abscesses usually require surgical drainage.
DOSAGE AND ADMINISTRATION
Cefprozil tablets are administered orally.
| Population/Infection | Dosage (mg) | Duration (days) |
|---|---|---|
| a In the treatment of infections due to Streptococcus pyogenes , cefprozil should be administered for at least 10 days. b Not to exceed recommended adult doses. | ||
| ADULTS (13 years and older) | ||
| UPPER RESPIRATORY TRACT | ||
| Pharyngitis/Tonsillitis | 500 q24h | 10 a |
| Acute Sinusitis (For moderate to severe infections, the higher dose should be used) | 250 q12h or 500 q12h | 10 |
| LOWER RESPIRATORY TRACT | ||
| Acute Bacterial Exacerbation of Chronic Bronchitis | 500 q12h | 10 |
| SKIN AND SKIN STRUCTURE | ||
| Uncomplicated Skin and Skin Structure Infections | 250 q12h or 500 q24h or 500 q12h | 10 |
| CHILDREN (2 years to 12 years) | ||
| UPPER RESPIRATORY TRACT b | ||
| Pharyngitis/Tonsillitis | 7.5 mg/kg q12h | 10 a |
| SKIN AND SKIN STRUCTURE | ||
| Uncomplicated Skin and Skin Structure Infections | 20 mg/kg q24h | 10 |
| INFANTS & CHILDREN (6 months to 12 years) | ||
| UPPER RESPIRATORY TRACT b | ||
| Otitis Media (See INDICATIONS AND USAGE and CLINICAL STUDIES ) | 15 mg/kg q12h | 10 |
| Acute Sinusitis (For moderate to severe infections, the higher dose should be used) | 7.5 mg/kg q12h or 15 mg/kg q12h | 10 |
Renal Impairment
Cefprozil may be administered to patients with impaired renal function. The following dosage schedule should be used.
| Creatinine Clearance (mL/min) | Dosage (mg) | Dosing Interval |
|---|---|---|
| • Cefprozil is in part removed by hemodialysis; therefore, cefprozil should be administered after the completion of hemodialysis. | ||
| 30 to 120 0 to 29• | standard 50% of standard | standard standard |
Hepatic Impairment
No dosage adjustment is necessary for patients with impaired hepatic function.
CONTRAINDICATIONS
Cefprozil tablets are contraindicated in patients with known allergy to the cephalosporin class of antibiotics.
ADVERSE REACTIONS
The adverse reactions to cefprozil are similar to those observed with other orally administered cephalosporins. Cefprozil was usually well tolerated in controlled clinical trials. Approximately 2% of patients discontinued cefprozil therapy due to adverse events. The most common adverse effects observed in patients treated with cefprozil are: Gastrointestinal: Diarrhea (2.9%), nausea (3.5%), vomiting (1%), and abdominal pain (1%). Hepatobiliary: Elevations of AST (SGOT) (2%), ALT (SGPT) (2%), alkaline phosphatase (0.2%), and bilirubin values (<0.1%). As with some penicillins and some other cephalosporin antibiotics, cholestatic jaundice has been reported rarely. Hypersensitivity: Rash (0.9%), urticaria (0.1%). Such reactions have been reported more frequently in children than in adults. Signs and symptoms usually occur a few days after initiation of therapy and subside within a few days after cessation of therapy. CNS: Dizziness (1%), hyperactivity, headache, nervousness, insomnia, confusion, and somnolence have been reported rarely (<1%). All were reversible. Hematopoietic: Decreased leukocyte count (0.2%), eosinophilia (2.3%). Renal: Elevated BUN (0.1%), serum creatinine (0.1%). Other: Diaper rash and superinfection (1.5%), genital pruritus and vaginitis (1.6%). The following adverse events, regardless of established causal relationship to cefprozil tablets, have been rarely reported during postmarketing surveillance: anaphylaxis, angioedema, colitis (including pseudomembranous colitis), erythema multiforme, fever, serum-sickness like reactions, Stevens-Johnson syndrome, and thrombocytopenia.
Cephalosporin class paragraph
In addition to the adverse reactions listed above which have been observed in patients treated with cefprozil, the following adverse reactions and altered laboratory tests have been reported for cephalosporin-class antibiotics: Aplastic anemia, hemolytic anemia, hemorrhage, renal dysfunction, toxic epidermal necrolysis, toxic nephropathy, prolonged prothrombin time, positive Coombs’ test, elevated LDH, pancytopenia, neutropenia, agranulocytosis. Several cephalosporins have been implicated in triggering seizures, particularly in patients with renal impairment, when the dosage was not reduced. (See DOSAGE AND ADMINISTRATION and OVERDOSAGE .) If seizures associated with drug therapy occur, the drug should be discontinued. Anticonvulsant therapy can be given if clinically indicated.
Drug Interactions
Nephrotoxicity has been reported following concomitant administration of aminoglycoside antibiotics and cephalosporin antibiotics. Concomitant administration of probenecid doubled the AUC for cefprozil. The bioavailability of the capsule formulation of cefprozil was not affected when administered 5 minutes following an antacid.
DESCRIPTION
Cefprozil is a semi-synthetic broad-spectrum cephalosporin antibiotic.
Cefprozil is a cis and trans isomeric mixture (≥90% cis). The chemical name for the monohydrate is (6 R ,7 R )-7-[( R )-2-Amino-2-( p -hydroxyphenyl)acetamido]-8-oxo-3-propenyl-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid monohydrate, and the structural formula is: 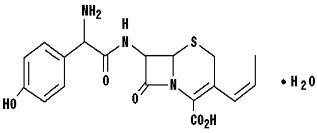
Cefprozil USP is a white to yellowish powder with a molecular formula for the monohydrate of C 18 H 19 N 3 O 5 S•H 2 O and a molecular weight of 407.45.
Cefprozil tablets USP are intended for oral administration.
Cefprozil tablets USP contain cefprozil USP equivalent to 250 mg or 500 mg of anhydrous cefprozil. In addition, each tablet contains the following inactive ingredients: microcrystalline cellulose, sodium starch glycolate, magnesium stearate, hypromellose, polyethylene glycol, polysorbate 80, and titanium dioxide. The 250 mg tablets also contain FD&C Yellow #6 aluminum lake. The tablets are imprinted with edible ink containing shellac glaze, black iron oxide, propylene glycol and ammonium hydroxide.
CLINICAL PHARMACOLOGY
The pharmacokinetic data were derived from the capsule formulation; however, bioequivalence has been demonstrated for the oral solution, capsule, tablet, and suspension formulations under fasting conditions. Following oral administration of cefprozil to fasting subjects, approximately 95% of the dose was absorbed. The average plasma half-life in normal subjects was 1.3 hours, while the steady-state volume of distribution was estimated to be 0.23 L/kg. The total body clearance and renal clearance rates were approximately 3 mL/min/kg and 2.3 mL/min/kg, respectively. Average peak plasma concentrations after administration of 250 mg, 500 mg, or 1 g doses of cefprozil to fasting subjects were approximately 6.1, 10.5, and 18.3 mcg/mL, respectively, and were obtained within 1.5 hours after dosing. Urinary recovery accounted for approximately 60% of the administered dose. (See Table.)
| •Data represent mean values of 12 healthy volunteers. | ||||
| Dosage (mg) | Mean Plasma Cefprozil Concentrations (mcg/mL)• | 8-hour Urinary Excretion (%) | ||
| Peak appx. 1.5 h | 4 h | 8 h | ||
| 250 mg | 6.1 | 1.7 | 0.2 | 60% |
| 500 mg | 10.5 | 3.2 | 0.4 | 62% |
| 1000 mg | 18.3 | 8.4 | 1.0 | 54% |
During the first 4-hour period after drug administration, the average urine concentrations following 250 mg, 500 mg, and 1 g doses were approximately 700 mcg/mL, 1000 mcg/mL, and 2900 mcg/mL, respectively. Administration of cefprozil with food did not affect the extent of absorption (AUC) or the peak plasma concentration (C max ) of cefprozil. However, there was an increase of 0.25 to 0.75 hours in the time to maximum plasma concentration of cefprozil (T max ). The bioavailability of the capsule formulation of cefprozil was not affected when administered 5 minutes following an antacid. Plasma protein binding is approximately 36% and is independent of concentration in the range of 2 mcg/mL to 20 mcg/mL. There was no evidence of accumulation of cefprozil in the plasma in individuals with normal renal function following multiple oral doses of up to 1000 mg every 8 hours for 10 days. In patients with reduced renal function, the plasma half-life may be prolonged up to 5.2 hours depending on the degree of the renal dysfunction. In patients with complete absence of renal function, the plasma half-life of cefprozil has been shown to be as long as 5.9 hours. The half-life is shortened during hemodialysis. Excretion pathways in patients with markedly impaired renal function have not been determined. (See PRECAUTIONS and DOSAGE AND ADMINISTRATION .) In patients with impaired hepatic function, the half-life increases to approximately 2 hours. The magnitude of the changes does not warrant a dosage adjustment for patients with impaired hepatic function. Healthy geriatric volunteers (≥65 years old) who received a single 1 g dose of cefprozil had 35% to 60% higher AUC and 40% lower renal clearance values compared with healthy adult volunteers 20 to 40 years of age. The average AUC in young and elderly female subjects was approximately 15% to 20% higher than in young and elderly male subjects. The magnitude of these age- and gender-related changes in the pharmacokinetics of cefprozil is not sufficient to necessitate dosage adjustments. Adequate data on CSF levels of cefprozil are not available. Comparable pharmacokinetic parameters of cefprozil are observed between pediatric patients (6 months to 12 years) and adults following oral administration of selected matched doses. The maximum concentrations are achieved at 1 to 2 hours after dosing. The plasma elimination half-life is approximately 1.5 hours. In general, the observed plasma concentrations of cefprozil in pediatric patients at the 7.5, 15, and 30 mg/kg doses are similar to those observed within the same time frame in normal adult subjects at the 250, 500, and 1000 mg doses, respectively. The comparative plasma concentrations of cefprozil in pediatric patients and adult subjects at the equivalent dose level are presented in the table below.
| a n=11; b n=5; c n=9; d n=11. | ||||||
| Mean (SD) Plasma Cefprozil Concentrations (mcg/mL) | ||||||
| Population | Dose | 1 h | 2 h | 4 h | 6 h | TT 1/2 (h) |
| children (n=18) | 7.5 mg/kg | 4.70 (1.57) | 3.99 (1.24) | 0.91 (0.30) | 0.23 a (0.13) | 0.94 (0.32) |
| adults (n=12) | 250 mg | 4.82 (2.13) | 4.92 (1.13) | 1.70 b (0.53) | 0.53 (0.17) | 1.28 (0.34) |
| children (n=19) | 15 mg/kg | 10.86 (2.55) | 8.47 (2.03) | 2.75 (1.07) | 0.61 c (0.27) | 1.24 (0.43) |
| adults (n=12) | 500 mg | 8.39 (1.95) | 9.42 (0.98) | 3.18 d (0.76) | 1.00 d (0.24) | 1.29 (0.14) |
| children (n=10) | 30 mg/kg | 16.69 (4.26) | 17.61 (6.39) | 8.66 (2.70) | — | 2.06 (0.21) |
| adults (n=12) | 1000 mg | 11.99 (4.67) | 16.95 (4.07) | 8.36 (4.13) | 2.79 (1.77) | 1.27 (0.12) |
Microbiology
Cefprozil has in vitro activity against a broad range of gram-positive and gram-negative bacteria. The bactericidal action of cefprozil results from inhibition of cell-wall synthesis. Cefprozil has been shown to be active against most strains of the following microorganisms both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
| Aerobic gram-positive microorganisms: | Aerobic gram-negative microorganisms: |
|---|---|
| Staphylococcus aureus (including β-lactamase-producing strains) | Haemophilus influenzae (including β-lactamase-producing strains) |
| NOTE : Cefprozil is inactive against methicillin-resistant staphylococci. | Moraxella (Branhamella) catarrhalis (including β-lactamase-producing strains) |
| Streptococcus pneumoniae | |
| Streptococcus pyogenes |
The following in vitro data are available; however, their clinical significance is unknown. Cefprozil exhibits in vitro minimum inhibitory concentrations (MICs) of 8 mcg/mL or less against most (≥90%) strains of the following microorganisms; however, the safety and effectiveness of cefprozil in treating clinical infections due to these microorganisms have not been established in adequate and well-controlled clinical trials. Aerobic gram-positive microorganisms: Enterococcus durans Staphylococcus warneri Enterococcus faecalis Streptococcus agalactiae Listeria monocytogenes Streptococci (Groups C,D,F, and G) Staphylococcus epidermidis viridans group Streptococci Staphylococcus saprophyticus NOTE: Cefprozil is inactive against Enterococcus faecium . Aerobic gram-negative microorganisms: Citrobacter diversus Proteus mirabilis Escherichia coli Salmonella spp. Klebsiella pneumoniae Shigella spp . Neisseria gonorrhoeae Vibrio spp. (including β-lactamase-producing strains) NOTE: Cefprozil is inactive against most strains of Acinetobacter , Enterobacter , Morganella morganii , Proteus vulgaris , Providencia , Pseudomonas , and Serratia . Anaerobic microorganisms: Prevotella (Bacteroides) melaninogenicus Fusobacterium spp. Clostridium difficile Peptostreptococcus spp. Clostridium perfringens Propionibacterium acnes NOTE: Most strains of the Bacteroides fragilis group are resistant to cefprozil. Susceptibility Testing For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
CLINICAL STUDIES
Study One:
In a controlled clinical study of acute otitis media performed in the United States where significant rates of β-lactamase-producing organisms were found, cefprozil was compared to an oral antimicrobial agent that contained a specific β-lactamase inhibitor. In this study, using very strict evaluability criteria and microbiologic and clinical response criteria at the 10 to 16 days post-therapy follow-up, the following presumptive bacterial eradication/clinical cure outcomes i.e., clinical success) and safety results were obtained:
| EFFICACY: | ||
| Pathogen | % of Cases with Pathogen (n=155) | Outcome |
| S. pneumoniae | 48.4% | cefprozil success rate 5% better than control |
| H. influenzae | 35.5% | cefprozil success rate 17% less than control |
| M. catarrhalis | 13.5% | cefprozil success rate 12% less than control |
| S. pyogenes | 2.6% | cefprozil equivalent to control |
| Overall | 100.0% | cefprozil success rate 5% less than control |
SAFETY: The incidences of adverse events, primarily diarrhea and rash•, were clinically and statistically significantly higher in the control arm versus the cefprozil arm.
| Age Group | Cefprozil | Control |
|---|---|---|
| •The majority of these involved the diaper area in young children. | ||
| 6 months to 2 years | 21% | 41% |
| 3 to 12 years | 10% | 19% |
Study Two:
In a controlled clinical study of acute otitis media performed in Europe, cefprozil was compared to an oral antimicrobial agent that contained a specific β-lactamase inhibitor. As expected in a European population, this study population had a lower incidence of β-lactamase-producing organisms than usually seen in U.S. trials. In this study, using very strict evaluability criteria and microbiologic and clinical response criteria at the 10 to 16 days post-therapy follow-up, the following presumptive bacterial eradication/clinical cure outcomes (i.e., clinical success) were obtained:
| EFFICACY: | ||
| Pathogen | % of Cases with Pathogen (n=47) | Outcome |
| S. pneumoniae | 51.0% | cefprozil equivalent to control |
| H. influenzae | 29.8% | cefprozil equivalent to control |
| M. catarrhalis | 6.4% | cefprozil equivalent to control |
| S. pyogenes | 12.8% | cefprozil equivalent to control |
| Overall | 100.0% | cefprozil equivalent to control |
SAFETY: The incidence of adverse events in the cefprozil arm was comparable to the incidence of adverse events in the control arm (agent that contained a specific β-lactamase inhibitor).
Distributed by: Aurobindo Pharma USA, Inc. 279 Princeton-Hightstown Road East Windsor, NJ 08520
Manufactured by: Aurobindo Pharma Limited Hyderabad-500 032, India Revised: 10/2021
HOW SUPPLIED
Cefprozil Tablets, USP 250 mg are orange colored, biconvex, film-coated capsule-shaped tablets, imprinted “C16” with black ink on one side. Bottles of 30 NDC 65862-068-30 Bottles of 50 NDC 65862-068-50 Bottles of 100 NDC 65862-068-01 Bottles of 500 NDC 65862-068-05 Cefprozil Tablets, USP 500 mg are white, biconvex, film-coated capsule-shaped tablets, imprinted “C17” with black ink on one side. Bottles of 30 NDC 65862-069-30 Bottles of 50 NDC 65862-069-50 Bottles of 100 NDC 65862-069-01 Bottles of 500 NDC 65862-069-05 Store at 20° to 25°C (68° to 77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].