Cefdinir - Cefdinir powder, For Suspension prescribing information
INDICATIONS AND USAGE
To reduce the development of drug-resistant bacteria and maintain the effectiveness of cefdinir for oral suspension and other antibacterial drugs, cefdinir for oral suspension should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
Cefdinir for oral suspension is indicated for the treatment of patients with mild to moderate infections caused by susceptible strains of the designated microorganisms in the conditions listed below.
Adults and Adolescents:
Caused by Haemophilus influenzae (including β-lactamase producing strains), Haemophilus parainfluenzae (including β-lactamase producing strains), Streptococcus pneumoniae (penicillin-susceptible strains only), and Moraxella catarrhalis (including β-lactamase producing strains) (see CLINICAL STUDIES ).
Caused by Haemophilus influenzae (including β-lactamase producing strains), Haemophilus parainfluenzae (including β-lactamase producing strains), Streptococcus pneumoniae (penicillin-susceptible strains only), and Moraxella catarrhalis (including β-lactamase producing strains).
Caused by Haemophilus influenzae (including β-lactamase producing strains), Streptococcus pneumoniae (penicillin-susceptible strains only), and Moraxella catarrhalis (including β-lactamase producing strains).
NOTE : For information on use in pediatric patients, see Pediatric Use and DOSAGE AND ADMINISTRATION .
Caused by Streptococcus pyogenes (see CLINICAL STUDIES ).
NOTE : Cefdinir is effective in the eradication of S. pyogenes from the oropharynx. Cefdinir has not, however, been studied for the prevention of rheumatic fever following S. pyogenes pharyngitis/tonsillitis.
Only intramuscular penicillin has been demonstrated to be effective for the prevention of rheumatic fever.
Caused by Staphylococcus aureus (including β-lactamase producing strains) and Streptococcus pyogenes .
Pediatric Patients:
Caused by Haemophilus influenzae (including β-lactamase producing strains), Streptococcus pneumoniae (penicillin-susceptible strains only), and Moraxella catarrhalis (including β-lactamase producing strains).
Caused by Streptococcus pyogenes (see CLINICAL STUDIES ).
NOTE : Cefdinir is effective in the eradication of S. pyogenes from the oropharynx. Cefdinir has not, however, been studied for the prevention of rheumatic fever following S. pyogenes pharyngitis/tonsillitis.
Only intramuscular penicillin has been demonstrated to be effective for the prevention of rheumatic fever.
Caused by Staphylococcus aureus (including β-lactamase producing strains) and Streptococcus pyogenes .
DOSAGE AND ADMINISTRATION
(see INDICATIONS AND USAGE for Indicated Pathogens)
The recommended dosage and duration of treatment for infections in pediatric patients are described in the following chart; the total daily dose for all infections is 14 mg/kg, up to a maximum dose of 600 mg per day. Once-daily dosing for 10 days is as effective as BID dosing. Once-daily dosing has not been studied in skin infections; therefore, cefdinir for oral suspension should be administered twice daily in this infection. Cefdinir for oral suspension may be administered without regard to meals.
| Header$Type of Infection | Dosage | Duration |
| Acute Bacterial Otitis Media | 7 mg/kg q12h or | 5 to 10 days |
| 14 mg/kg q24h | 10 days | |
| Acute Maxillary Sinusitis | 7 mg/kg q12h or | 10 days |
| 14 mg/kg q24h | 10 days | |
| Pharyngitis/Tonsillitis | 7 mg/kg q12h or | 5 to 10 days |
| 14 mg/kg q24h | 10 days | |
| Uncomplicated Skin and Skin Structure Infections | 7 mg/kg q12h | 10 days |
a Pediatric patients who weight ≥43 kg should receive the maximum daily dose of 600 mg. | ||
| Header$Weight | 125 mg/5 mL | 250 mg/5 mL |
| 9 kg/20 lbs | 2.5 mL q12h or 5 mL q24h | Use 125 mg/5 mL product |
| 18 kg/40 lbs | 5 mL q12h or 10 mL q24h | 2.5 mL q12h or 5 mL q24h |
| 27 kg/60 lbs | 7.5 mL q12h or 15 mL q24h | 3.75 mL q12h or 7.5 mL q24h |
| 36 kg/80 lbs | 10 mL q12h or 20 mL q24h | 5 mL q12h or 10 mL q24h |
| ≥43 kg a /95 lbs | 12 mL q12h or 24 mL q24h | 6 mL q12h or 12 mL q24h |
Patients With Renal Insufficiency:
For adult patients with creatinine clearance <30 mL/min, the dose of cefdinir should be 300 mg given once daily.
Creatinine clearance is difficult to measure in outpatients. However, the following formula may be used to estimate creatinine clearance (CL cr ) in adult patients. For estimates to be valid, serum creatinine levels should reflect steady-state levels of renal function.
(weight) (140 – age)
Males: CL cr = ————————————
(72) (serum creatinine)
Females: CL cr = 0.85 x above value
where creatinine clearance is in mL/min, age is in years, weight is in kilograms, and serum creatinine is in mg/dL. 4
The following formula may be used to estimate creatinine clearance in pediatric patients:
body length or height
CL cr = K x ———————————
serum creatinine
where K = 0.55 for pediatric patients older than 1 year 5 and 0.45 for infants (up to 1 year) 6 .
In the above equation, creatinine clearance is in mL/min/1.73 m 2 , body length or height is in centimeters, and serum creatinine is in mg/dL.
For pediatric patients with a creatinine clearance of <30 mL/min/1.73 m 2 , the dose of cefdinir should be 7 mg/kg (up to 300 mg) given once daily.
Patients on Hemodialysis:
Hemodialysis removes cefdinir from the body. In patients maintained on chronic hemodialysis, the recommended initial dosage regimen is a 300 mg or 7 mg/kg dose every other day. At the conclusion of each hemodialysis session, 300 mg (or 7 mg/kg) should be given. Subsequent doses (300 mg or 7 mg/kg) are then administered every other day.
| Final Concentration | Final Volume(mL) | Amount of Water | Directions |
| 125 mg/5 mL | 60 | 38 mL | Tap bottle to loosen powder, then add water in 2 |
| 100 | 61 mL | portions. Shake well after each aliquot. | |
| 250 mg/5 mL | 60 | 38 mL | Tap bottle to loosen powder, then add water in 2 |
| 100 | 61 mL | portions. Shake well after each aliquot. |
After mixing, the suspension can be stored at 20° to 25°C (68° to 77°F). The container should be kept tightly closed, and the suspension should be shaken well before each administration. The suspension may be used for 10 days, after which any unused portion must be discarded.
CONTRAINDICATIONS
Cefdinir is contraindicated in patients with known allergy to the cephalosporin class of antibiotics.
DESCRIPTION
Cefdinir for oral suspension contain the active ingredient cefdinir, an extended-spectrum, semisynthetic cephalosporin, for oral administration. Chemically, cefdinir is [6R-[6α,7β(Z)]]-7-[[(2-amino-4-thiazolyl)(hydroxyimino)acetyl]amino]-3-ethenyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid. Cefdinir is a white to slightly brownish-yellow solid. It is slightly soluble in dilute hydrochloric acid and sparingly soluble in 0.1 M pH 7.0 phosphate buffer. The molecular formula is C 14 H 13 N 5 O 5 S 2 and the molecular weight is 395.42. Cefdinir has the structural formula shown below:
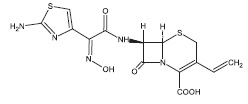
Cefdinir for oral suspension, after reconstitution, contains 125 mg cefdinir per 5 mL or 250 mg cefdinir per 5 mL and the following inactive ingredients: anhydrous citric acid; colloidal silicon dioxide; guar gum; anhydrous sodium citrate; sodium benzoate; strawberry flavour; sucrose; and xanthan gum.
CLINICAL PHARMACOLOGY
Pharmacokinetics and Drug Metabolism:
Oral Bioavailability: Maximal plasma cefdinir concentrations occur 2 to 4 hours postdose following capsule or suspension administration. Plasma cefdinir concentrations increase with dose, but the increases are less than dose-proportional from 300 mg (7 mg/kg) to 600 mg (14 mg/kg). Following administration of suspension to healthy adults, cefdinir bioavailability is 120% relative to capsules. Estimated bioavailability of cefdinir capsules is 21% following administration of a 300 mg capsule dose, and 16% following administration of a 600 mg capsule dose. Estimated absolute bioavailability of cefdinir suspension is 25%. Cefdinir oral suspension of 250 mg/5 mL strength was shown to be bioequivalent to the 125 mg/5 mL strength in healthy adults under fasting conditions.
Effect of Food: The C max and AUC of cefdinir from the capsules are reduced by 16% and 10%, respectively, when given with a high-fat meal. In adults given the 250 mg/5 mL oral suspension with a high-fat meal, the C max and AUC of cefdinir are reduced by 44% and 33%, respectively. The magnitude of these reductions is not likely to be clinically significant because the safety and efficacy studies of oral suspension in pediatric patients were conducted without regard to food intake. Therefore, cefdinir may be taken without regard to food.
Cefdinir Suspension : Cefdinir plasma concentrations and pharmacokinetic parameter values following administration of single 7 and 14 mg/kg oral doses of cefdinir to pediatric subjects (age 6 months-12 years) are presented in the following table:
| Dose | Cmax ( mcg / mL ) | tmax ( hr ) | AUC ( mcg . hr / mL ) |
| 7 mg/kg | 2.30(0.65) | 2.2(0.6) | 8.31(2.50) |
| 14 mg/kg | 3.86(0.62) | 1.8(0.4) | 13.4(2.64) |
Multiple Dosing: Cefdinir does not accumulate in plasma following once- or twice-daily administration to subjects with normal renal function.
The mean volume of distribution (Vd area ) of cefdinir in adult subjects is 0.35 L/kg (±0.29); in pediatric subjects (age 6 months-12 years), cefdinir Vd area is 0.67 L/kg (±0.38). Cefdinir is 60% to 70% bound to plasma proteins in both adult and pediatric subjects; binding is independent of concentration.
Skin Blister: In adult subjects, median (range) maximal blister fluid cefdinir concentrations of 0.65 (0.33-1.1) and 1.1 (0.49-1.9) mcg/mL were observed 4 to 5 hours following administration of 300 and 600 mg doses, respectively. Mean (±SD) blister C max and AUC (0- ∞) values were 48% (±13) and 91% (±18) of corresponding plasma values.
Tonsil Tissue: In adult patients undergoing elective tonsillectomy, respective median tonsil tissue cefdinir concentrations 4 hours after administration of single 300 and 600 mg doses were 0.25 (0.22-0.46) and 0.36 (0.22-0.80) mcg/g. Mean tonsil tissue concentrations were 24% (±8) of corresponding plasma concentrations.
Sinus Tissue: In adult patients undergoing elective maxillary and ethmoid sinus surgery, respective median sinus tissue cefdinir concentrations 4 hours after administration of single 300 and 600 mg doses were <0.12 (<0.12-0.46) and 0.21 (<0.12-2.0) mcg/g. Mean sinus tissue concentrations were 16% (±20) of corresponding plasma concentrations.
Lung Tissue: In adult patients undergoing diagnostic bronchoscopy, respective median bronchial mucosa cefdinir concentrations 4 hours after administration of single 300 and 600 mg doses were 0.78 (<0.06-1.33) and 1.14 (<0.06-1.92) mcg/mL, and were 31% (±18) of corresponding plasma concentrations. Respective median epithelial lining fluid concentrations were 0.29 (<0.3-4.73) and 0.49 (<0.3-0.59) mcg/mL, and were 35% (±83) of corresponding plasma concentrations.
Middle Ear Fluid: In 14 pediatric patients with acute bacterial otitis media, respective median middle ear fluid cefdinir concentrations 3 hours after administration of single 7 and 14 mg/kg doses were 0.21 (<0.09-0.94) and 0.72 (0.14-1.42) mcg/mL. Mean middle ear fluid concentrations were 15% (±15) of corresponding plasma concentrations.
CSF: Data on cefdinir penetration into human cerebrospinal fluid are not available.
Cefdinir is not appreciably metabolized. Activity is primarily due to parent drug. Cefdinir is eliminated principally via renal excretion with a mean plasma elimination half-life (t 1/2 ) of 1.7 (±0.6) hours. In healthy subjects with normal renal function, renal clearance is 2.0 (±1.0) mL/min/kg, and apparent oral clearance is 11.6 (±6.0) and 15.5 (±5.4) mL/min/kg following doses of 300 and 600 mg, respectively. Mean percent of dose recovered unchanged in the urine following 300 and 600 mg doses is 18.4% (±6.4) and 11.6% (±4.6), respectively. Cefdinir clearance is reduced in patients with renal dysfunction (see Special Populations: Patients with Renal Insufficiency ).
Because renal excretion is the predominant pathway of elimination, dosage should be adjusted in patients with markedly compromised renal function or who are undergoing hemodialysis (see DOSAGE AND ADMINISTRATION ).
Patients with Renal Insufficiency: Cefdinir pharmacokinetics were investigated in 21 adult subjects with varying degrees of renal function. Decreases in cefdinir elimination rate, apparent oral clearance (CL/F), and renal clearance were approximately proportional to the reduction in creatinine clearance (CL cr ). As a result, plasma cefdinir concentrations were higher and persisted longer in subjects with renal impairment than in those without renal impairment. In subjects with CL cr between 30 and 60 mL/min, C max and t 1/2 increased by approximately 2-fold and AUC by approximately 3-fold. In subjects with CL cr <30 mL/min, C max increased by approximately 2-fold, t 1/2 by approximately 5-fold, and AUC by approximately 6-fold. Dosage adjustment is recommended in patients with markedly compromised renal function (creatinine clearance <30 mL/min; see DOSAGE AND ADMINISTRATION ).
Hemodialysis: Cefdinir pharmacokinetics were studied in 8 adult subjects undergoing hemodialysis. Dialysis (4 hours duration) removed 63% of cefdinir from the body and reduced apparent elimination t 1/2 from 16 (±3.5) to 3.2 (±1.2) hours. Dosage adjustment is recommended in this patient population (see DOSAGE AND ADMINISTRATION ).
Hepatic Disease: Because cefdinir is predominantly renally eliminated and not appreciably metabolized, studies in patients with hepatic impairment were not conducted. It is not expected that dosage adjustment will be required in this population.
Geriatric Patients: The effect of age on cefdinir pharmacokinetics after a single 300 mg dose was evaluated in 32 subjects 19 to 91 years of age. Systemic exposure to cefdinir was substantially increased in older subjects (N=16), C max by 44% and AUC by 86%. This increase was due to a reduction in cefdinir clearance. The apparent volume of distribution was also reduced, thus no appreciable alterations in apparent elimination t 1/2 were observed (elderly: 2.2 ± 0.6 hours vs young: 1.8 ± 0.4 hours). Since cefdinir clearance has been shown to be primarily related to changes in renal function rather than age, elderly patients do not require dosage adjustment unless they have markedly compromised renal function (creatinine clearance <30 mL/min, see Patients with Renal Insufficiency , above).
Gender and Race: The results of a meta-analysis of clinical pharmacokinetics (N=217) indicated no significant impact of either gender or race on cefdinir pharmacokinetics.
Microbiology:
Mechanism of Action:
As with other cephalosporins, bactericidal activity of cefdinir results from inhibition of cell wall synthesis. Cefdinir is stable in the presence of some, but not all, β-lactamase enzymes. As a result, many organisms resistant to penicillins and some cephalosporins are susceptible to cefdinir.
Mechanism of Resistance:
Resistance to cefdinir is primarily through hydrolysis by some β-lactamases, alteration of penicillin-binding proteins (PBPs) and decreased permeability. Cefdinir is inactive against most strains of Enterobacter spp., Pseudomonas spp., Enterococcus spp., penicillin-resistant streptococci, and methicillin-resistant staphylococci. β-lactamase negative, ampicillin-resistant (BLNAR) H. influenzae strains are typically non-susceptible to cefdinir.
Antimicrobial Activity:
Cefdinir has been shown to be active against most strains of the following microorganisms, both in vitro and in clinical infections as described in INDICATIONS AND USAGE .
Gram-Positive Bacteria:
Staphylococcus aureus (methicillin-susceptible strains only)
Streptococcus pneumoniae (penicillin-susceptible strains only)
Streptococcus pyogenes
Gram-Negative Bacteria:
Haemophilus influenzae
Haemophilus parainfluenzae
Moraxella catarrhalis
The following in vitro data are available, but their clinical significance is unknown.
Cefdinir exhibits in vitro minimum inhibitory concentrations (MICs) of 1 mcg/mL or less against (≥ 90%) strains of the following microorganisms; however, the safety and effectiveness of cefdinir in treating clinical infections due to these microorganisms have not been established in adequate and well-controlled clinical trials.
Gram-Positive Bacteria:
Staphylococcus epidermidis (methicillin-susceptible strains only)
Streptococcus agalactiae
Viridans group streptococci
Gram-Negative Bacteria:
Citrobacter koseri
Escherichia coli
Klebsiella pneumoniae
Proteus mirabilis
Susceptibility Test Methods:
When available, the clinical microbiology laboratory should provide periodic reports that describe the regional/local susceptibility profile of potential nosocomial and community-acquired pathogens. These reports should aid the physician in selecting an antibacterial drug for treatment.
Dilution Techniques:
Quantitative methods are used to determine antimicrobial minimum inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized test method 1 (broth and/or agar). The MIC values should be interpreted according to criteria provided in Table 1.
Diffusion Techniques:
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. The zone size should be determined using a standardized method. 2 The procedure uses paper disks impregnated with 5 mcg cefdinir to test the susceptibility of bacteria. The disk diffusion interpretive criteria are provided in Table 1.
| Minimu m Inhibitory | Z one Diameter | |||||
| Microorganisms Streptococci other than S. pneumoniae that are susceptible to penicillin (MIC ≤ 0.12 mcg/mL), can be considered susceptible to cefdinir. | Concentratio n (mcg/mL) | (mm) | ||||
| S | I | R | S | I | R | |
| H aemophilus influenzae | ≤ 1 | - | - | ≥ 20 | - | - |
| H aemophilus parainfluenzae | ≤ 1 | - | - | ≥ 20 | - | - |
| M oraxella catarrhalis | ≤ 1 | 2 | ≥ 4 | ≥ 20 | 17 to 19 | ≤ 16 |
| Streptococcus pneumoniae S. pneumoniae that are susceptible to penicillin (MIC ≤ 0.06 mcg/mL) can be considered susceptible to cefdinir. Isolates of S. pneumoniae tested against a 1-µg oxacillin disk with oxacillin zone sizes ≥ 20 mm are susceptible to penicillin and can be considered susceptible to cefdinir. Testing of cefdinir against penicillin-intermediate or penicillin-resistant isolates is not recommended. Reliable interpretive criteria for cefdinir are not available. | ≤ 0.5 | 1 | ≥ 2 | - | - | - |
| Streptococcus pyogenes | ≤ 1 | 2 | ≥ 4 | ≥ 20 | 17 to 19 | ≤ 16 |
Susceptibility of staphylococci to cefdinir may be deduced from testing penicillin and either cefoxitin or oxacillin. Staphylococci susceptible to oxacillin (cefoxitin) can be considered susceptible to cefdinir 3 .
A report of "Susceptible" indicates that antimicrobial is likely to inhibit growth of the pathogen if the antimicrobial compound reaches the concentrations at the site of infection necessary to inhibit growth of the pathogen. A report of "Intermediate" indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where a high dosage of drug can be used. This category also provides a buffer zone that prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of "Resistant" indicates that the antimicrobial is not likely to inhibit growth of the pathogen if the antimicrobial compound reaches the concentrations usually achievable at the infection site; other therapy should be selected.
Quality Control:
Standardized susceptibility test procedures require the use of laboratory controls to monitor and ensure the accuracy and precision of supplies and reagents used in the assay, and the techniques of the individual performing the test. 1,2,3 Standard cefdinir powder should provide the following range of MIC values as noted in Table 2. For the diffusion technique using a 5 mcg disk the criteria in Table 2 should be achieved.
| QC Strain | Minimum Inhibitory Concentration (mcg/mL) | Zone Diameter (mm) |
| Escherichia coli ATCC 25922 | 0.12 to 0.5 | 24 to 28 |
| Haemophilus influenzae ATCC 49766 | 0.12 to 0.5 | 24 to 31 |
| Staphylococcus aureus ATCC 25923 | - | 25 to 32 |
| Staphylococcus aureus ATCC 29213 | 0.12 to 0.5 | - |
| Streptococcus pneumoniae ATCC 49619 | 0.03 to 0.25 | 26 to 31 |
CLINICAL STUDIES
Community-Acquired Bacterial Pneumonia:
In a controlled, double-blind study in adults and adolescents conducted in the US, cefdinir BID was compared with cefaclor 500 mg TID. Using strict evaluability and microbiologic/clinical response criteria 6 to 14 days posttherapy, the following clinical cure rates, presumptive microbiologic eradication rates, and statistical outcomes were obtained:
| Cefdinir BID | Cefaclor TID | Outcome | |
| Clinical Cure Rates | 150/187 (80%) | 147/186 (79%) | Cefdinir equivalent to control |
| Eradication Rates | |||
| Overall | 177/195 (91%) | 184/200 (92%) | Cefdinir equivalent to control |
| S . pneumoniae | 31/31 (100%) | 35/35 (100%) | |
| H . influenzae | 55/65 (85%) | 60/72 (83%) | |
| M . catarrhalis | 10/10 (100%) | 11/11 (100%) | |
| H . parainfluenzae | 81/89 (91%) | 78/82 (95%) |
In a second controlled, investigator-blind study in adults and adolescents conducted primarily in Europe, cefdinir BID was compared with amoxicillin/clavulanate 500/125 mg TID. Using strict evaluability and clinical response criteria 6 to 14 days posttherapy, the following clinical cure rates, presumptive microbiologic eradication rates, and statistical outcomes were obtained:
| Cefdinir BID | Amoxicillin / Clavulanate TID | Outcome | |
| Clinical Cure Rates | 83/104 (80%) | 86/97 (89%) | Cefdinir not equivalent to control |
| Eradication Rates | |||
| Overall | 85/96 (89%) | 84/90 (93%) | Cefdinir equivalent to control |
| S . pneumoniae | 42/44 (95%) | 43/44 (98%) | |
| H . influenzae | 26/35 (74%) | 21/26 (81%) | |
| M . catarrhalis | 6/6 (100%) | 8/8 (100%) | |
| H . parainfluenzae | 11/11 (100%) | 12/12 (100%) |
Streptococcal Pharyngitis/Tonsillitis:
In four controlled studies conducted in the United States, cefdinir was compared with 10 days of penicillin in adult, adolescent, and pediatric patients. Two studies (one in adults and adolescents, the other in pediatric patients) compared 10 days of cefdinir QD or BID to penicillin 250 mg or 10 mg/kg QID. Using strict evaluability and microbiologic/clinical response criteria 5 to 10 days posttherapy, the following clinical cure rates, microbiologic eradication rates, and statistical outcomes were obtained:
| Study | Efficacy Parameter | Cefdinir QD | Cefdinir BID | Penicillin QID | Outcome |
| Adults/ Adolescents | Eradication of S . pyogenes | 192/210 (91%) | 199/217 (92%) | 181/217 (83%) | Cefdinir superior to control |
| Clinical Cure Rates | 199/210 (95%) | 209/217 (96%) | 193/217 (89%) | Cefdinir superior to control | |
| Pediatric Patients | Eradication of S . pyogenes | 215/228 (94%) | 214/227 (94%) | 159/227 (70%) | Cefdinir superior to control |
| Clinical Cure Rates | 222/228 (97%) | 218/227 (96%) | 196/227 (86%) | Cefdinir superior to control |
Two studies (one in adults and adolescents, the other in pediatric patients) compared 5 days of cefdinir BID to 10 days of penicillin 250 mg or 10 mg/kg QID. Using strict evaluability and microbiologic/ clinical response criteria 4 to 10 days posttherapy, the following clinical cure rates, microbiologic eradication rates, and statistical outcomes were obtained:
| Study | Efficacy Parameter | Cefdinir BID | Penicillin QID | Outcome |
| Adults/ Adolescents | Eradication of S . pyogenes | 193/218 (89%) | 176/214 (82%) | Cefdinir equivalent to control |
| Clinical Cure Rates | 194/218 (89%) | 181/214 (85%) | Cefdinir equivalent to control | |
| Pediatric Patients | Eradication of S . pyogenes | 176/196 (90%) | 135/193 (70%) | Cefdinir superior to control |
| Clinical Cure Rates | 179/196 (91%) | 173/193 (90%) | Cefdinir equivalent to control |
HOW SUPPLIED
Cefdinir for oral suspension USP, is an off-white to creamish powder formulation that, when reconstituted as directed, contains 125 mg cefdinir/5 mL or 250 mg cefdinir/5 mL. The reconstituted suspension has an off-white to creamish color and strawberry flavor. The powder is available as follows:
125 mg/5 mL:
60 mL bottles NDC 68180-722-04
100 mL bottles NDC 68180-722-05
250 mg/5 mL:
60 mL bottles NDC 68180-723-04
100 mL bottles NDC 68180-723-05
Store dry powder and reconstituted suspension at 20° to 25 ° C (68° to 77 ° F); [See USP Controlled Room Temperature].