Get your patient on Captopril And Hydrochlorothiazide - Captopril And Hydrochlorothiazide tablet (Captopril And Hydrochlorothiazide)
Captopril And Hydrochlorothiazide - Captopril And Hydrochlorothiazide tablet prescribing information
BOXED WARNING
USE IN PREGNANCY When used in pregnancy during the second and third trimesters, ACE Inhibitors can cause injury and even death to the developing fetus. When pregnancy is detected, captopril and hydrochlorothiazide should be discontinued as soon as possible. See WARNINGS: Captopril: Fetal/Neonatal Morbidity and Mortality .
INDICATIONS & USAGE
Captopril and hydrochlorothiazide tablets are indicated for the treatment of hypertension. The blood pressure lowering effects of captopril and thiazides are approximately additive. This fixed combination drug may be used as initial therapy or substituted for previously titrated doses of the individual components. When captopril and hydrochlorothiazide are given together it may not be necessary to administer captopril in divided doses to attain blood pressure control at trough (before the next dose). Also, with such a combination, a daily dose of 15 mg of hydrochlorothiazide may be adequate. Treatment may, therefore, be initiated with captopril and hydrochlorothiazide tablets 25 mg/15 mg once daily. Subsequent titration should be with additional doses of the components (captopril, hydrochlorothiazide) as single agents or as captopril and hydrochlorothiazide tablets 50 mg/15 mg, 25 mg/25 mg, or 50 mg/25 mg (see DOSAGE AND ADMINISTRATION ).
In using captopril and hydrochlorothiazide, consideration should be given to the risk of neutropenia/agranulocytosis (see WARNINGS ).
Captopril and hydrochlorothiazide may be used for patients with normal renal function, in whom the risk is relatively low. In patients with impaired renal function, particularly those with collagen vascular disease, captopril and hydrochlorothiazide should be reserved for hypertensives who have either developed unacceptable side effects on other drugs, or have failed to respond satisfactorily to other drug combinations.
ACE inhibitors (for which adequate data are available) cause a higher rate of angioedema in black than in non-black patients (see WARNINGS: Captopril: Head and Neck Angioedema and Intestinal Angioedema ).
DOSAGE & ADMINISTRATION
DOSAGE MUST BE INDIVIDUALIZED ACCORDING TO PATIENT'S RESPONSE. Captopril and hydrochlorothiazide tablets may be substituted for the previously titrated individual components. Alternatively, therapy may be instituted with a single tablet of captopril and hydrochlorothiazide 25 mg/15 mg taken once daily. For patients insufficiently responsive to the initial dose, additional captopril or hydrochlorothiazide may be added as individual components or by using captopril and hydrochlorothiazide tablets 50 mg/15 mg, 25 mg/25 mg or 50 mg/25 mg, or divided doses may be used. Because the full effect of a given dose may not be attained for 6 to 8 weeks, dosage adjustments should generally be made at 6 week intervals, unless the clinical situation demands more rapid adjustment. In general, daily doses of captopril should not exceed 150 mg and of hydrochlorothiazide should not exceed 50 mg. Captopril and hydrochlorothiazide tablets should be taken one hour before meals. Dosage Adjustment in Renal Impairment: Because captopril and hydrochlorothiazide are excreted primarily by the kidneys, excretion rates are reduced in patients with impaired renal function. These patients will take longer to reach steady-state captopril levels and will reach higher steady-state levels for a given daily dose than patients with normal renal function. Therefore, these patients may respond to smaller or less frequent doses of captopril and hydrochlorothiazide. After the desired therapeutic effect has been achieved, the dose intervals should be increased or the total daily dose reduced until the minimal effective dose is achieved. When concomitant diuretic therapy is required in patients with severe renal impairment, a loop diuretic (e.g., furosemide), rather than a thiazide diuretic is preferred for use with captopril; therefore, for patients with severe renal dysfunction the captopril-hydrochlorothiazide combination tablet is not usually recommended. (See WARNINGS: Captopril: Anaphylactoid Reactions During Membrane Exposure and PRECAUTIONS: Hemodialysis ).
CONTRAINDICATIONS
Captopril : This product is contraindicated in patients who are hypersensitive to captopril or any other angiotensin-converting enzyme inhibitor (e.g., a patient who has experienced angioedema during therapy with any other ACE inhibitor). Hydrochlorothiazide : Hydrochlorothiazide is contraindicated in anuria. It is also contraindicated in patients who have previously demonstrated hypersensitivity to hydrochlorothiazide or other sulfonamide-derived drugs.
ADVERSE REACTIONS
Captopril : Reported incidences are based on clinical trials involving approximately 7000 patients.
Renal : About one of 100 patients developed proteinuria (see WARNINGS ). Each of the following has been reported in approximately 1 to 2 of 1000 patients and are of uncertain relationship to drug use: renal insufficiency, renal failure, nephrotic syndrome, polyuria, oliguria, and urinary frequency.
Hematologic : Neutropenia/agranulocytosis has occurred (see WARNINGS ). Cases of anemia, thrombocytopenia, and pancytopenia have been reported.
Dermatologic : Rash, often with pruritus, and sometimes with fever, arthralgia, and eosinophilia, occurred in about 4 to 7 (depending on renal status and dose) of 100 patients, usually during the first four weeks of therapy. It is usually maculopapular, and rarely urticarial. The rash is usually mild and disappears within a few days of dosage reduction, short-term treatment with an antihistaminic agent, and/or discontinuing therapy; remission may occur even if captopril is continued. Pruritus, without rash, occurs in about 2 of 100 patients. Between 7 and 10 percent of patients with skin rash have shown eosinophilia and/or positive ANA titers. A reversible associated pemphigoid-like lesion, and photosensitivity, have also been reported. Flushing or pallor has been reported in 2 to 5 of 1000 patients.
Cardiovascular : Hypotension may occur; see WARNINGS and PRECAUTIONS (Drug Interactions) for discussion of hypotension with captopril therapy. Tachycardia, chest pain, and palpitations have each been observed in approximately 1 of 100 patients. Angina pectoris, myocardial infarction, Raynaud’s syndrome, and congestive heart failure have each occurred in 2 to 3 of 1000 patients. Dysgeusia : Approximately 2 to 4 (depending on renal status and dose) of 100 patients developed a diminution or loss of taste perception. Taste impairment is reversible and usually self-limited (2 to 3 months) even with continued drug administration. Weight loss may be associated with the loss of taste. Angioedema : Angioedema involving the extremities, face, lips, mucous membranes, tongue, glottis or larynx has been reported in approximately one in 1000 patients. Angioedema involving the upper airways has caused fatal airway obstruction. (See WARNINGS: Captopril: Head and Neck Angioedema and Intestinal Angioedema and PRECAUTIONS: Information for Patients ). Cough : Cough has been reported in 0.5 to 2% of patients treated with captopril in clinical trials (see PRECAUTIONS: General: Captopril: Cough ). The following have been reported in about 0.5 to 2 percent of patients but did not appear at increased frequency compared to placebo or other treatments used in controlled trials: gastric irritation, abdominal pain, nausea, vomiting, diarrhea, anorexia, constipation, aphthous ulcers, peptic ulcer, dizziness, headache, malaise, fatigue, insomnia, dry mouth, dyspnea, alopecia, paresthesias. Other clinical adverse effects reported since the drug was marketed are listed below by body system. In this setting, an incidence or causal relationship cannot be accurately determined. Body as a Whole : Anaphylactoid reactions (see WARNINGS: Captopril: Anaphylactoid and Possibly Related Reactions and PRECAUTIONS: Hemodialysis ). General : asthenia, gynecomastia. Cardiovascular : cardiac arrest, cerebrovascular accident/insufficiency, rhythm disturbances, orthostatic hypotension, syncope. Dermatologic : bullous pemphigus, erythema multiforme (including Stevens-Johnson syndrome), exfoliative dermatitis. Gastrointestinal : pancreatitis, glossitis, dyspepsia. Hematologic : anemia, including aplastic and hemolytic. Hepatobiliary : jaundice, hepatitis, including rare cases of necrosis, cholestasis. Metabolic : symptomatic hyponatremia. Musculoskeletal : myalgia, myasthenia. Nervous/Psychiatric : ataxia, confusion, depression, nervousness, somnolence. Respiratory : bronchospasm, eosinophilic pneumonitis, rhinitis. Special Senses : blurred vision. Urogenital : impotence. As with other ACE inhibitors, a syndrome has been reported which may include: fever, myalgia, arthralgia, interstitial nephritis, vasculitis, rash or other dermatologic manifestations, eosinophilia and an elevated ESR. Fetal/Neonatal Morbidity and Mortality: See WARNINGS: Captopril: Fetal/Neonatal Morbidity and Mortality .
Hydrochlorothiazide : Gastrointestinal System : anorexia, gastric irritation, nausea, vomiting, cramping, diarrhea, constipation, jaundice (intrahepatic cholestatic jaundice), pancreatitis, and sialadenitis. Central Nervous System : dizziness, vertigo, paresthesias, headache, and xanthopsia. Hematologic : leukopenia, agranulocytosis, thrombocytopenia, aplastic anemia, and hemolytic anemia. Cardiovascular : orthostatic hypotension. Hypersensitivity : purpura, photosensitivity, rash, urticaria, necrotizing angiitis (vasculitis; cutaneous vasculitis), fever, respiratory distress including pneumonitis, and anaphylactic reactions. Other : hyperglycemia, glycosuria, hyperuricemia, muscle spasm, weakness, restlessness, and transient blurred vision. Whenever adverse reactions are moderate or severe, thiazide dosage should be reduced or therapy withdrawn. Altered Laboratory Findings: Serum Electrolytes : Hyperkalemia: small increases in serum potassium, especially in patients with renal impairment (see PRECAUTIONS: Captopril ). Hyponatremia : particularly in patients receiving a low sodium diet or concomitant diuretics. BUN/Serum Creatinine : Transient elevations of BUN or serum creatinine especially in volume or salt depleted patients or those with renovascular hypertension may occur. Rapid reduction of longstanding or markedly elevated blood pressure can result in decreases in the glomerular filtration rate and, in turn, lead to increases in BUN or serum creatinine. Hematologic : A positive ANA has been reported. Liver Function Tests : Elevations of liver transaminases, alkaline phosphatase, and serum bilirubin have occurred. Postmarketing Experience : Non-melanoma Skin Cancer : Hydrochlorothiazide is associated with an increased risk of non-melanoma skin cancer. In a study conducted in the Sentinel System, increased risk was predominantly for squamous cell carcinoma (SCC) and in white patients taking large cumulative doses. The increased risk for SCC in the overall population was approximately 1 additional case per 16,000 patients per year, and for white patients taking a cumulative dose of ≥ 50,000 mg the risk increase was approximately 1 additional SCC case for every 6,700 patients per year. To report SUSPECTED ADVERSE REACTIONS, contact Rising Pharma Holdings, Inc. at 1-844- 874-7464 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions:
Captopril : Hypotension—Patients On Diuretic Therapy : Patients on diuretics and especially those in whom diuretic therapy was recently instituted, as well as those on severe dietary salt restrictions or dialysis, may occasionally experience a precipitous reduction of blood pressure usually within the first hour after receiving the initial dose of captopril.
The possibility of hypotensive effects with captopril can be minimized by either discontinuing the diuretic or increasing the salt intake approximately one week prior to initiation of treatment with captopril or initiating therapy with small doses (6.25 or 12.5 mg). Alternatively, provide medical supervision for at least one hour after the initial dose. If hypotension occurs, the patient should be placed in a supine position and, if necessary, receive an intravenous infusion of normal saline. This transient hypotensive response is not a contraindication to further doses which can be given without difficulty once the blood pressure has increased after volume expansion.
Agents Having Vasodilator Activity : Data on the effect of concomitant use of other vasodilators in patients receiving captopril for heart failure are not available; therefore, nitroglycerin or other nitrates (as used for management of angina) or other drugs having vasodilator activity should, if possible, be discontinued before starting captopril. If resumed during captopril therapy, such agents should be administered cautiously, and perhaps at lower dosage.
Agents Causing Renin Release : Captopril’s effect will be augmented by antihypertensive agents that cause renin release. For example, diuretics (e.g., thiazides) may activate the renin-angiotensin-aldosterone system.
Agents Affecting Sympathetic Activity : The sympathetic nervous system may be especially important in supporting blood pressure in patients receiving captopril alone or with diuretics. Therefore, agents affecting sympathetic activity (e.g., ganglionic blocking agents or adrenergic neuron blocking agents) should be used with caution. Beta-adrenergic blocking drugs add some further antihypertensive effect to captopril, but the overall response is less than additive.
Agents Increasing Serum Potassium : Since captopril decreases aldosterone production, elevation of serum potassium may occur. Potassium-sparing diuretics such as spironolactone, triamterene, or amiloride, or potassium supplements, should be given only for documented hypokalemia, and then with caution, since they may lead to a significant increase of serum potassium. Salt substitutes containing potassium should also be used with caution.
Gold : Nitritoid reactions (symptoms include facial flushing, nausea, vomiting and hypotension) have been reported rarely in patients on therapy with injectable gold (sodium aurothiomalate) and concomitant ACE inhibitor therapy including captopril and hydrochlorothiazide tablets.
Inhibitors of Endogenous Prostaglandin Synthesis : It has been reported that indomethacin may reduce the antihypertensive effect of captopril, especially in cases of low renin hypertension. Other non-steroidal anti-inflammatory agents (e.g., aspirin) may also have this effect.
Lithium : Increased serum lithium levels and symptoms of lithium toxicity have been reported in patients receiving concomitant lithium and ACE inhibitor therapy. These drugs should be coadministered with caution and frequent monitoring of serum lithium levels is recommended. If a diuretic is also used, it may increase the risk of lithium toxicity (see PRECAUTIONS: Drug Interactions: Hydrochlorothiazide: Lithium ). Hydrochlorothiazide : When administered concurrently the following drugs may interact with thiazide diuretics:
Alcohol, Barbiturates, or Narcotics : potentiation of orthostatic hypotension may occur.
Amphotericin B, Corticosteroids, or Corticotropin (ACTH) : may intensify electrolyte imbalance, particularly hypokalemia. Monitor potassium levels; use potassium replacements if necessary.
Anticoagulants (Oral) : dosage adjustments of anticoagulant medication may be necessary since hydrochlorothiazide may decrease their effects.
Antigout Medications : dosage adjustments of antigout medication may be necessary since hydrochlorothiazide may raise the level of blood uric acid.
Other Antihypertensive Medications (e.g., Ganglionic or Peripheral Adrenergic Blocking Agents) : dosage adjustments may be necessary since hydrochlorothiazide may potentiate their effects.
Antidiabetic Drugs (Oral Agents and Insulin): since thiazides may elevate blood glucose levels, dosage adjustments of antidiabetic agents may be necessary.
Calcium Salts : Increased serum calcium levels due to decreased excretion may occur. If calcium must be prescribed, monitor serum calcium levels and adjust calcium dosage accordingly.
Cardiac Glycosides : enhanced possibility of digitalis toxicity associated with hypokalemia. Monitor potassium levels (see PRECAUTIONS: Drug Interactions: Captopril ).
Cholestyramine and Colestipol Resins : Absorption of hydrochlorothiazide is impaired in the presence of anionic exchange resins. Single doses of either cholestyramine or colestipol resins bind the hydrochlorothiazide and reduce its absorption from the gastrointestinal tract by up to 85 and 43 percent, respectively.
Diazoxide : enhanced hyperglycemic, hyperuricemic, and antihypertensive effects. Be cognizant of possible interaction; monitor blood glucose and serum uric acid levels.
Lithium : diuretic agents reduce the renal clearance of lithium and increase the risk of lithium toxicity. These drugs should be coadministered with caution and frequent monitoring of serum lithium levels is recommended (see PRECAUTIONS: Drug Interactions: Captopril: Lithium ).
MAO Inhibitors : dosage adjustments of one or both agents may be necessary since hypotensive effects are enhanced.
Nondepolarizing Muscle Relaxants, Preanesthetics and Anesthetics Used in Surgery (e.g., Tubocurarine Chloride and Gallamine Triethiodide) : effects of these agents may be potentiated; dosage adjustments may be required. Monitor and correct any fluid and electrolyte imbalances prior to surgery if feasible.
Non-steroidal Anti-inflammatory Agents : in some patients, the administration of a non-steroidal anti-inflammatory agent can reduce the diuretic, natriuretic, and antihypertensive effect of loop, potassium-sparing or thiazide diuretics. Therefore, when hydrochlorothiazide and non-steroidal anti-inflammatory agents are used concomitantly, the patient should be observed closely to determine if the desired effect of the diuretic is obtained.
Methenamine : possible decreased effectiveness due to alkalinization of the urine.
Pressor Amines (e.g., Norepinephrine) : decreased arterial responsiveness, but not sufficient to preclude effectiveness of the pressor agent for therapeutic use. Use caution in patients taking both medications who undergo surgery. Administer preanesthetic and anesthetic agents in reduced dosage, and if possible, discontinue hydrochlorothiazide therapy one week prior to surgery.
Probenecid or Sulfinpyrazone : increased dosage of these agents may be necessary since hydrochlorothiazide may have hyperuricemic effects.
DESCRIPTION
Captopril and hydrochlorothiazide tablets, USP for oral administration combines two antihypertensive agents: captopril and hydrochlorothiazide. Captopril, the first of a new class of antihypertensive agents, is a specific competitive inhibitor of angiotensin I-converting enzyme (ACE), the enzyme responsible for the conversion of angiotensin I to angiotensin II. Hydrochlorothiazide is a benzothiadiazide (thiazide) diuretic-antihypertensive. Captopril, USP is a white to off-white crystalline powder with characteristic sulfide-like odor; it is freely soluble in water, ethanol, methanol, chloroform, isopropyl alcohol and methylene chloride and soluble in ethyl acetate. It dissolves in dilute solution of alkali hydroxide. Hydrochlorothiazide, USP is a white or almost white, almost odorless crystalline powder. Very slightly soluble in water, sparingly soluble in Ethanol and Methanol, soluble in Acetone. Freely soluble in N,N-Dimethylformamide, in n-Butylamine and in diluted solutions of alkali hydroxides. Practically insoluble in Ether, in Chloroform and in diluted mineral acids. Captopril is designated chemically as 1-[(2S)-3-Mercapto-2-methylpropionyl]-L-proline; Hydrochlorothiazide is 6-Chloro-3,4-dihydro-2 H -1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide. Their structural formulas are:
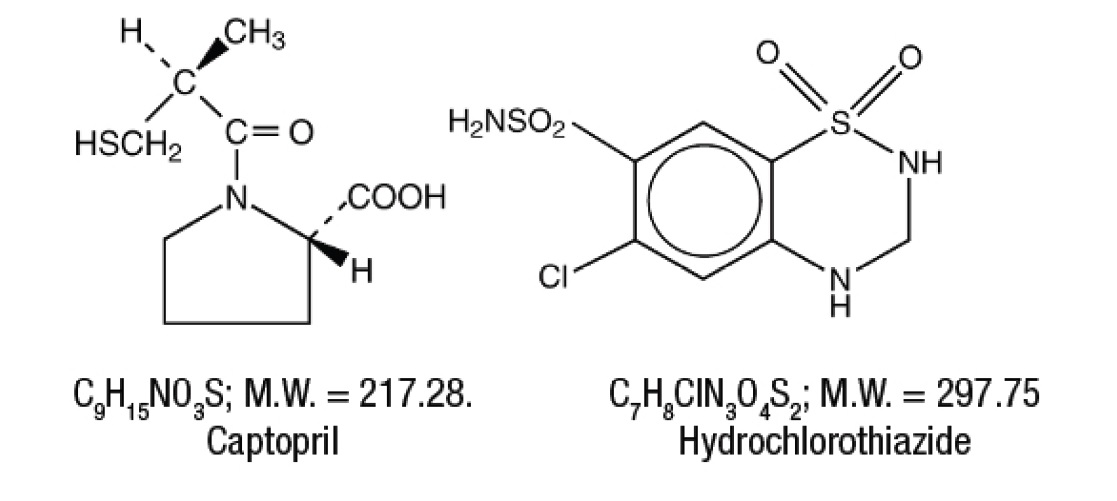
Captopril and hydrochlorothiazide tablets are available for oral administration in four combinations of captopril with hydrochlorothiazide: 25 mg with 15 mg, 25 mg with 25 mg, 50 mg with 15 mg, and 50 mg with 25 mg. In addition, each tablet contains the following inactive ingredients: anhydrous lactose, colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, pregelatinized starch (corn) and sodium lauryl sulfate. The 25 mg/25 mg and 50 mg/25 mg tablets also contain FD&C Yellow No. 6 Aluminum Lake.
CLINICAL PHARMACOLOGY
Captopril : Mechanism of Action : The mechanism of action of captopril has not yet been fully elucidated. Its beneficial effects in hypertension and heart failure appear to result primarily from suppression of the renin-angiotensin-aldosterone system. However, there is no consistent correlation between renin levels and response to the drug. Renin, an enzyme synthesized by the kidneys, is released into the circulation where it acts on a plasma globulin substrate to produce angiotensin I, a relatively inactive decapeptide. Angiotensin I is then converted by angiotensin converting enzyme (ACE) to angiotensin II, a potent endogenous vasoconstrictor substance. Angiotensin II also stimulates aldosterone secretion from the adrenal cortex, thereby contributing to sodium and fluid retention. Captopril prevents the conversion of angiotensin I to angiotensin II by inhibition of ACE, a peptidyldipeptide carboxy hydrolase. This inhibition has been demonstrated in both healthy human subjects and in animals by showing that the elevation of blood pressure caused by exogenously administered angiotensin I was attenuated or abolished by captopril. In animal studies, captopril did not alter the pressor responses to a number of other agents, including angiotensin II and norepinephrine, indicating specificity of action.
ACE is identical to “bradykininase”, and captopril may also interfere with the degradation of the vasodepressor peptide, bradykinin. Increased concentrations of bradykinin or prostaglandin E 2 may also have a role in the therapeutic effect of captopril.
Inhibition of ACE results in decreased plasma angiotensin II and increased plasma renin activity (PRA), the latter resulting from loss of negative feedback on renin release caused by reduction in angiotensin II. The reduction of angiotensin II leads to decreased aldosterone secretion, and, as a result, small increases in serum potassium may occur along with sodium and fluid loss.
The antihypertensive effects persist for a longer period of time than does demonstrable inhibition of circulating ACE. It is not known whether the ACE present in vascular endothelium is inhibited longer than the ACE in circulating blood.
Pharmacokinetics:
After oral administration of therapeutic doses of captopril, rapid absorption occurs with peak blood levels at about one hour. The presence of food in the gastrointestinal tract reduces absorption by about 30 to 40 percent; captopril therefore should be given one hour before meals. Based on carbon-14 labeling, average minimal absorption is approximately 75 percent. In a 24-hour period, over 95 percent of the absorbed dose is eliminated in the urine; 40 to 50 percent is unchanged drug; most of the remainder is the disulfide dimer of captopril and captopril-cysteine disulfide. Approximately 25 to 30 percent of the circulating drug is bound to plasma proteins. The apparent elimination half-life for total radioactivity in blood is probably less than three hours. An accurate determination of half-life of unchanged captopril is not, at present, possible, but it is probably less than two hours. In patients with renal impairment, however, retention of captopril occurs (see DOSAGE AND ADMINISTRATION ).
Pharmacodynamics:
Administration of captopril results in a reduction of peripheral arterial resistance in hypertensive patients with either no change, or an increase, in cardiac output. There is an increase in renal blood flow following administration of captopril and glomerular filtration rate is usually unchanged. In patients with heart failure, significantly decreased peripheral (systemic vascular) resistance and blood pressure (afterload), reduced pulmonary capillary wedge pressure (preload) and pulmonary vascular resistance, increased cardiac output, and increased exercise tolerance time (ETT) have been demonstrated.
Reductions of blood pressure are usually maximal 60 to 90 minutes after oral administration of an individual dose of captopril. The duration of effect is dose related and is extended in the presence of a thiazide-type diuretic. The full effect of a given dose may not be attained for 6 to 8 weeks (see DOSAGE AND ADMINISTRATION ). The blood pressure lowering effects of captopril and thiazide-type diuretics are additive. In contrast, captopril and beta-blockers have a less than additive effect.
Blood pressure is lowered to about the same extent in both standing and supine positions. Orthostatic effects and tachycardia are infrequent but may occur in volume-depleted patients. Abrupt withdrawal of captopril has not been associated with a rapid increase in blood pressure. Studies in rats and cats indicate that captopril does not cross the blood-brain barrier to any significant extent. Hydrochlorothiazide : Thiazides affect the renal tubular mechanism of electrolyte reabsorption. At maximal therapeutic dosage all thiazides are approximately equal in their diuretic potency. Thiazides increase excretion of sodium and chloride in approximately equivalent amounts. Natriuresis causes a secondary loss of potassium and bicarbonate. The mechanism of the antihypertensive effect of thiazides is unknown. Thiazides do not affect normal blood pressure. The mean plasma half-life of hydrochlorothiazide in fasted individuals has been reported to be approximately 2.5 hours. Onset of diuresis occurs in two hours and the peak effect at about four hours. Its action persists for approximately six to twelve hours. Hydrochlorothiazide is eliminated rapidly by the kidney.
HOW SUPPLIED
Captopril and Hydrochlorothiazide Tablets, USP are available containing 25 mg or 50 mg of captopril, USP with 15 mg or 25 mg of hydrochlorothiazide, USP providing the following available combinations: 25 mg/15 mg, 25 mg/25 mg, 50 mg/15 mg or 50 mg/25 mg.
The 25 mg/15 mg tablets are supplied as a white, round, biconvex tablet debossed with ‘R over 27’ on one side of the tablet and quadrisect scored on the other side. They are available as follows: Bottles of 90 tablets - NDC 16571-827-09
The 25 mg/25 mg tablets are supplied as a peach, round, biconvex tablet debossed with ‘R over 28’ on one side of the tablet and quadrisect scored on the other side. They are available as follows: Bottles of 90 tablets - NDC 16571-828-09
The 50 mg/15 mg tablets are supplied as a white, capsule-shaped, biconvex tablet debossed with ‘ R ’ to the left of the partial score and ‘ 29 ’ to the right of the partial score on one side of the tablet and partially scored on the other side. They are available as follows: Bottles of 90 tablets - NDC 16571-829-09
The 50 mg/25 mg tablets are supplied as a peach, capsule-shaped, biconvex tablet debossed with R to the left of the partial score and 30 to the right of the partial score on one side of the tablet and partially scored on the other side. They are available as follows: Bottles of 90 tablets - NDC 16571-830-09
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure. Keep container tightly closed.
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Protect from moisture.
Manufactured for: Rising Pharma Holdings, Inc. East Brunswick, NJ 08816
Issued: 12/2022 200640 PIR83001-00
Captopril And Hydrochlorothiazide - Captopril And Hydrochlorothiazide tablet PubMed™ news
- Journal Article • 2026 MayPost-Approval Pediatric Use of Drugs Granted Waivers from Pediatric Testing.
- Journal Article • 2026 MayACE-inhibitory peptide Leu-Tyr-Ala in skeletal muscle actin exhibits anti-aging effects through the inhibition of acn-1 in Caenorhabditis elegans.
- Journal Article • 2026 MayEvaluation of the antihypertensive and antihyperlipidaemic effects of a pyrimidine derivative in preventing cardiometabolic risk.
- Journal Article • 2026 MayInnovative valsartan-loaded self-nanoemulsifying drug delivery system combat liver inflammation and oxidative stress in streptozotocin-induced diabetic rats.
- Journal Article • 2026 MayPro-inflammatory roles of ultraviolet radiation in cutaneous photocarcinogenesis.