Get your patient on Caffeine Citrate - Caffeine Citrate injection (Caffeine Citrate)
Caffeine Citrate - Caffeine Citrate injection prescribing information
INDICATIONS AND USAGE
Caffeine citrate injection and caffeine citrate oral solution are indicated for the short term treatment of apnea of prematurity in infants between 28 and <33 weeks gestational age.
DOSAGE AND ADMINISTRATION
Prior to initiation of caffeine citrate, baseline serum levels of caffeine should be measured in infants previously treated with theophylline, since preterm infants metabolize theophylline to caffeine. Likewise, baseline serum levels of caffeine should be measured in infants born to mothers who consumed caffeine prior to delivery, since caffeine readily crosses the placenta.
The recommended loading dose and maintenance doses of caffeine citrate follow.
| Dose of Caffeine Citrate | Dose of Caffeine Citrate | Route | Frequency | |
| Volume | mg/kg | |||
| Loading Dose | 1 mL/kg | 20 mg/kg | Intravenous using a syringe infusion pump | One Time |
| (over 30 | ||||
| minutes) | ||||
| Maintenance | 0.25 mL/kg | 5 mg/kg | Intravenous | Every |
| Dose | (over 10 | 24 hours beginning 24 hours after the loading dose | ||
| minutes) or | ||||
| Orally |
NOTE THAT THE DOSE OF CAFFEINE BASE IS ONE-HALF THE DOSE WHEN EXPRESSED AS CAFFEINE CITRATE (e.g., 20 mg of caffeine citrate is equivalent to 10 mg of caffeine base).
Serum concentrations of caffeine may need to be monitored periodically throughout treatment to avoid toxicity. Serious toxicity has been associated with serum levels greater than 50 mg/L.
Caffeine citrate injection and caffeine citrate oral solution should be inspected visually for particulate matter and discoloration prior to administration. Vials containing discolored solution or visible particulate matter should be discarded.
Drug Compatibility
To test for drug compatibility with common intravenous solutions or medications, 20 mL of caffeine citrate injection were combined with 20 mL of a solution or medication, with the exception of an Intralipid ® admixture, which was combined as 80 mL/80 mL. The physical appearance of the combined solutions was evaluated for precipitation. The admixtures were mixed for 10 minutes and then assayed for caffeine. The admixtures were then continually mixed for 24 hours, with further sampling for caffeine assays at 2, 4, 8, and 24 hours.
Based on this testing, caffeine citrate injection, 60 mg/3 mL is chemically stable for 24 hours at room temperature when combined with the following test products.
- Dextrose Injection, USP 5%
- 50% Dextrose Injection USP
- Intralipid ® 20% IV Fat Emulsion
- Aminosyn ® 8.5% Crystalline Amino Acid Solution
- Dopamine HCI Injection, USP 40 mg/mL diluted to 0.6 mg/mL with Dextrose Injection, USP 5%
- Calcium Gluconate Injection, USP 10% (0.465 mEq/Ca +2 /mL)
- Heparin Sodium Injection, USP 1000 units/mL diluted to 1 unit/mL with Dextrose Injection, USP 5%
Fentanyl Citrate Injection, USP 50 µg/mL diluted to 10 µg/mL with Dextrose Injection, USP 5%
CONTRAINDICATIONS
Caffeine citrate injection and caffeine citrate oral solution are contraindicated in patients who have demonstrated hypersensitivity to any of its components.
ADVERSE REACTIONS
Overall, the reported number of adverse events in the double-blind period of the controlled trial was similar for the caffeine citrate and placebo groups. The following table shows adverse events that occurred in the double-blind period of the controlled trial and that were more frequent in caffeine citrate treated patients than placebo.
| Adverse Event (AE) | Caffeine Citrate N=46 | Placebo N=39 |
| n (%) | n (%) | |
| BODY AS A WHOLE | ||
| Accidental Injury | 1 (2.2) | 0 (0.0) |
| Feeding Intolerance | 4 (8.7) | 2 (5.1) |
| Sepsis | 2 (4.3) | 0 (0.0) |
| CARDIOVASCULAR SYSTEM | ||
| Hemorrhage | 1 (2.2) | 0 (0.0) |
| DIGESTIVE SYSTEM | ||
| Necrotizing Enterocolitis | 2 (4.3) | 1 (2.6) |
| Gastritis | 1 (2.2) | 0 (0.0) |
| Gastrointestinal Hemorrhage | 1 (2.2) | 0 (0.0) |
| HEMIC AND LYMPHATIC SYSTEM | ||
| Disseminated Intravascular | 1 (2.2) | 0 (0.0) |
| Coagulation | ||
| METABOLIC AND NUTRITIVE DISORDERS | ||
| Acidosis | 1 (2.2) | 0 (0.0) |
| Healing Abnormal | 1 (2.2) | 0 (0.0) |
| NERVOUS SYSTEM | ||
| Cerebral Hemorrhage | 1 (2.2) | 0 (0.0) |
| RESPIRATORY SYSTEM | ||
| Dyspnea | 1 (2.2) | 0 (0.0) |
| Lung Edema | 1 (2.2) | 0 (0.0) |
| SKIN AND APPENDAGES | ||
| Dry Skin | 1 (2.2) | 0 (0.0) |
| Rash | 4 (8.7) | 3 (7.7) |
| Skin Breakdown | 1 (2.2) | 0 (0.0) |
| SPECIAL SENSES | ||
| Retinopathy of Prematurity | 1 (2.2) | 0 (0.0) |
| UROGENITAL SYSTEM | ||
| Kidney Failure | 1 (2.2) | 0 (0.0) |
In addition to the cases above, three cases of necrotizing enterocolitis were diagnosed in patients receiving caffeine citrate during the open-label phase of the study.
Three of the infants who developed necrotizing enterocolitis during the trial died. All had been exposed to caffeine. Two were randomized to caffeine, and one placebo patient was “rescued” with open-label caffeine for uncontrolled apnea.
Adverse events described in the published literature include: central nervous system stimulation (i.e., irritability, restlessness, jitteriness), cardiovascular effects (i.e., tachycardia, increased left ventricular output, and increased stroke volume), gastrointestinal effects (i.e., increased gastric aspirate, gastrointestinal intolerance), alterations in serum glucose (hypoglycemia and hyperglycemia) and renal effects (increased urine flow rate, increased creatinine clearance, and increased sodium and calcium excretion). Published long-term follow-up studies have not shown caffeine to adversely affect neurological development or growth parameters.
Drug Interactions
Cytochrome P450 1A2 (CYP1A2) is known to be the major enzyme involved in the metabolism of caffeine. Therefore, caffeine has the potential to interact with drugs that are substrates for CYP1A2, inhibit CYP1A2, or induce CYP1A2.
Few data exist on drug interactions with caffeine in preterm neonates. Based on adult data, lower doses of caffeine may be needed following coadministration of drugs which are reported to decrease caffeine elimination (e.g., cimetidine and ketoconazole) and higher caffeine doses may be needed following coadministration of drugs that increase caffeine elimination (e.g., phenobarbital and phenytoin).
Caffeine administered concurrently with ketoprofen reduced the urine volume in four healthy volunteers. The clinical significance of this interaction in preterm neonates is not known.
Interconversion between caffeine and theophylline has been reported in preterm neonates. The concurrent use of these drugs is not recommended.
DESCRIPTION
Both caffeine citrate injection for intravenous administration and caffeine citrate oral solution are clear, colorless, sterile, non-pyrogenic, preservative-free, aqueous solutions adjusted to pH 4.7. Each mL contains 20 mg caffeine citrate (equivalent to 10 mg of caffeine base) prepared in solution by the addition of 10 mg caffeine anhydrous, USP to 5 mg citric acid monohydrate, USP, 8.3 mg sodium citrate dihydrate, USP and Water for Injection, USP.
Caffeine, a central nervous system stimulant, is an odorless white crystalline powder or granule, with a bitter taste. It is sparingly soluble in water and ethanol at room temperature. The chemical name of caffeine is 3,7-dihydro-1,3,7-trimethyl-1 H -purine-2,6-dione. In the presence of citric acid it forms caffeine citrate salt in solution. The structural formula and molecular weight of caffeine citrate follows. 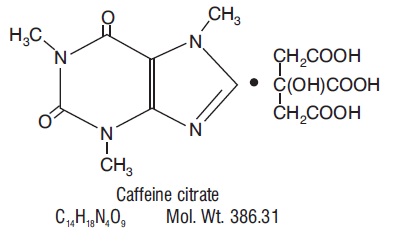
CLINICAL PHARMACOLOGY
Mechanism of Action
Caffeine is structurally related to other methylxanthines, theophylline, and theobromine. It is a bronchial smooth muscle relaxant, a CNS stimulant, a cardiac muscle stimulant, and a diuretic.
Although the mechanism of action of caffeine in apnea of prematurity is not known, several mechanisms have been hypothesized. These include: (1) stimulation of the respiratory center, (2) increased minute ventilation, (3) decreased threshold to hypercapnia, (4) increased response to hypercapnia, (5) increased skeletal muscle tone, (6) decreased diaphragmatic fatigue, (7) increased metabolic rate, and (8) increased oxygen consumption.
Most of these effects have been attributed to antagonism of adenosine receptors, both A 1 and A 2 subtypes, by caffeine, which has been demonstrated in receptor binding assays and observed at concentrations approximating those achieved therapeutically.
Pharmacokinetics
Absorption: After oral administration of 10 mg caffeine base/kg to preterm neonates, the peak plasma level (C max ) for caffeine ranged from 6 to 10 mg/L and the mean time to reach peak concentration (T max ) ranged from 30 minutes to 2 hours. The T max was not affected by formula feeding. The absolute bioavailability, however, was not fully examined in preterm neonates.
Distribution: Caffeine is rapidly distributed into the brain. Caffeine levels in the cerebrospinal fluid of preterm neonates approximate their plasma levels. The mean volume of distribution of caffeine in infants (0.8 to 0.9 L/kg) is slightly higher than that in adults (0.6 L/kg). Plasma protein binding data are not available for neonates or infants. In adults, the mean plasma protein binding in vitro is reported to be approximately 36%.
Metabolism: Hepatic cytochrome P450 1A2 (CYP1A2) is involved in caffeine biotransformation. Caffeine metabolism in preterm neonates is limited due to their immature hepatic enzyme systems.
Interconversion between caffeine and theophylline has been reported in preterm neonates; caffeine levels are approximately 25% of theophylline levels after theophylline administration and approximately 3 to 8% of caffeine administered would be expected to convert to theophylline.
Elimination: In young infants, the elimination of caffeine is much slower than that in adults due to immature hepatic and/or renal function. Mean half-life (T 1/2 ) and fraction excreted unchanged in urine (A e ) of caffeine in infants have been shown to be inversely related to gestational/postconceptual age. In neonates, the T 1/2 is approximately 3-4 days and the A e is approximately 86% (within 6 days). By 9 months of age, the metabolism of caffeine approximates that seen in adults (T 1/2 = 5 hours and A e = 1%).
Special Populations: Studies examining the pharmacokinetics of caffeine in neonates with hepatic or renal insufficiency have not been conducted. Caffeine citrate should be administered with caution in preterm neonates with impaired renal or hepatic function. Serum concentrations of caffeine should be monitored and dose administration of caffeine citrate should be adjusted to avoid toxicity in this population.
Clinical Studies
One multicenter, randomized, double-blind trial compared caffeine citrate to placebo in eighty-five (85) preterm infants (gestational age 28 to <33 weeks) with apnea of prematurity. Apnea of prematurity was defined as having at least 6 apnea episodes of greater than 20 seconds duration in a 24-hour period with no other identifiable cause of apnea. A 1 mL/kg (20 mg/kg caffeine citrate providing 10 mg/kg as caffeine base) loading dose of caffeine citrate was administered intravenously, followed by a 0.25 mL/kg (5 mg/kg caffeine citrate providing 2.5 mg/kg of caffeine base) daily maintenance dose administered either intravenously or orally (generally through a feeding tube). The duration of treatment in this study was limited to 10 to 12 days. The protocol allowed infants to be “rescued” with open-label caffeine citrate treatment if their apnea remained uncontrolled during the double-blind phase of the trial.
The percentage of patients without apnea on day 2 of treatment (24 to 48 hours after the loading dose) was significantly greater with caffeine citrate than placebo. The following table summarizes the clinically relevant endpoints evaluated in this study:
| Caffeine Citrate | Placebo | p-value | |
| Number of patients evaluated Of 85 patients who received drug, 3 were not included in the efficacy analysis because they had <6 apnea episodes/24 hours at baseline. | 45 | 37 | |
| % of patients with zero apnea events on day 2 | 26.7 | 8.1 | 0.03 |
| Apnea rate on day 2 (per 24 hrs.) | 4.9 | 7.2 | 0.134 |
| % of patients with 50% reduction in apnea events from baseline on day 2 | 76 | 57 | 0.07 |
In this 10 to 12 day trial, the mean number of days with zero apnea events was 3 in the caffeine citrate group and 1.2 in the placebo group. The mean number of days with a 50% reduction from baseline in apnea events was 6.8 in the caffeine citrate group and 4.6 in the placebo group.
HOW SUPPLIED
Both caffeine citrate injection and caffeine citrate oral solution are available as clear, colorless, sterile, non-pyrogenic, preservative-free, aqueous solutions in 5 mL colorless glass vials. The vials of caffeine citrate injection are sealed with gray rubber stopper and white flip off aluminum seal printed with “FOR INTRAVENOUS USE ONLY” in red. The vials of caffeine citrate oral solution are sealed with gray rubber stopper and a dark blue matte finish, flip off tear off aluminum seal printed with "FOR ORAL USE ONLY" "FLIP TOP & TEAR" in white.
Both the injection and oral solution vials contain 3 mL solution at a concentration of 20 mg/mL caffeine citrate (60 mg/vial) equivalent to 10 mg/mL caffeine base (30 mg/vial).
Caffeine citrate injection, USP
NDC 47335-289-40: 3 mL vial, individually packaged in a carton.
Caffeine citrate oral solution, USP
NDC 47335-290-44: 3 mL vial (NOT CHILD-RESISTANT), 10 vials per white polypropylene child-resistant container.
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F) [see USP Controlled Room Temperature].
Preservative free. For single use only. Discard unused portion. ATTENTION PHARMACIST: Detach “Instructions for Use” from the package insert and dispense with caffeine citrate oral solution prescription. Distributed by: Sun Pharmaceutical Industries, Inc. Cranbury, NJ 08512 Manufactured by: Sun Pharmaceutical Industries Ltd. Halol-Baroda Highway, Halol-389 350, Gujarat, India. PJPI0274B ISS. 12/2020