Get your patient on Butalbital And Acetaminophen - Butalbital And Acetaminophen tablet (Butalbital And Acetaminophen)
Butalbital And Acetaminophen - Butalbital And Acetaminophen tablet prescribing information
WARNING
Hepatotoxicity
Acetaminophen has been associated with cases of acute liver failure, at times resulting in liver transplant and death. Most of the cases of liver injury are associated with the use of acetaminophen at Doses that exceed 4000 milligrams per day, and often involve more than one acetaminophen-containing product.
INDICATIONS AND USAGE
Butalbital and acetaminophen tablets are indicated for the relief of the symptom complex of tension (or muscle contraction) headache.
Evidence supporting the efficacy and safety of this combination product in the treatment of multiple recurrent headaches is unavailable. Caution in this regard is required because butalbital is habit-forming and potentially abusable.
DOSAGE AND ADMINISTRATION
50 mg/325 mg: One to two tablets every four hours as needed. Total daily dosage should not exceed six tablets.
50 mg/300 mg : One or two tablets every four hours. Total daily dosage should not exceed six tablets.
Extended and repeated use of this product is not recommended because of the potential for physical dependence.
CONTRAINDICATIONS
This product is contraindicated under the following conditions:
- Hypersensitivity or intolerance to any component of this product.
- Patients with porphyria
ADVERSE REACTIONS
Frequently Observed: The most frequently reported adverse reactions are drowsiness, lightheadedness, dizziness, sedation, shortness of breath, nausea, vomiting, abdominal pain, and intoxicated feeling.
Infrequently Observed: All adverse events tabulated below are classified as infrequent.
Central Nervous System: headache, shaky feeling, tingling, agitation, fainting, fatigue, heavy eyelids, high energy, hot spells, numbness, sluggishness, seizure. Mental confusion, excitement or depression can also occur due to intolerance, particularly in elderly or debilitated patients, or due to overdosage of butalbital.
Autonomic Nervous System: dry mouth, hyperhidrosis.
Gastrointestinal: difficulty swallowing, heartburn, flatulence, constipation.
Cardiovascular: tachycardia.
Musculoskeletal: leg pain, muscle fatigue.
Genitourinary: diuresis.
Miscellaneous: pruritus, fever, earache, nasal congestion, tinnitus, euphoria, allergic reactions.
Several cases of dermatological reactions, including toxic epidermal necrolysis and erythema multiforme, have been reported.
The following adverse drug events may be borne in mind as potential effects of the components of this product. Potential effects of high dosage are listed in the OVERDOSAGE section.
Acetaminophen: allergic reactions, rash, thrombocytopenia, agranulocytosis.
DESCRIPTION
Each Butalbital and acetaminophen tablets, 50 mg/ 325 mg for oral administration contains:
•Butalbital, USP………...….. 50 mg
Acetaminophen, USP..……. 325 mg
Each Butalbital and acetaminophen tablets, 50 mg/ 300 mg for oral administration contains:
•Butalbital, USP………...….. 50 mg
Acetaminophen, USP..……. 300 mg
• Habit forming ingredient
In addition, each tablet contains the following inactive ingredients: Microcrystalline Cellulose, Silicified Microcrystalline Cellulose, Croscarmellose Sodium, Colloidal Silicon Dioxide, Magnesium Stearate, Pregelatinized Starch, Povidone, Crospovidone and Stearic Acid.
Butalbital (5-allyl-5-isobutylbarbituric acid), a slightly bitter, white, odorless, crystalline powder, is a short to intermediate-acting barbiturate. It has the following structural formula:
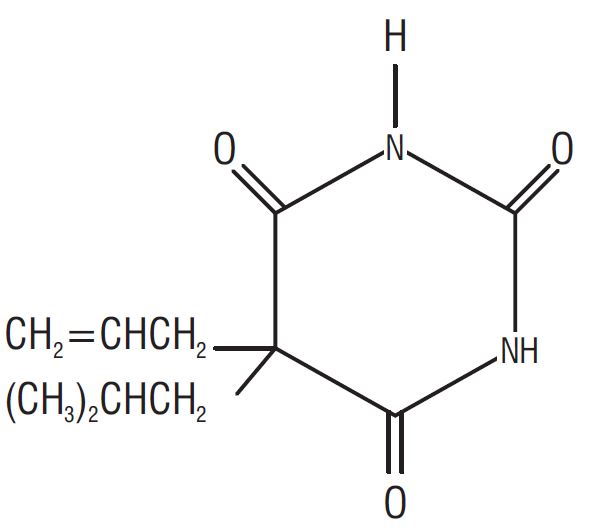
C 11 H 16 N 2 O 3 M.W.=224.26
Acetaminophen (4'-hydroxyacetanilide), a slightly bitter, white, odorless, crystalline powder, is a non- opiate, non-salicylate analgesic and antipyretic. It has the following structural formula:
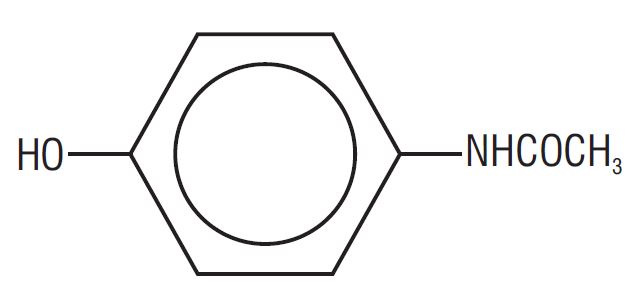
C 8 H 9 NO 2 M.W.=151.16
CLINICAL PHARMACOLOGY
This combination drug product is intended as a treatment for tension headache.
It consists of a fixed combination of butalbital and acetaminophen. The role each component plays in the relief of the complex of symptoms known as tension headache is incompletely understood.
Pharmacokinetics
The behavior of the individual components is described below.
Butalbital : Butalbital is well absorbed from the gastrointestinal tract and is expected to distribute to most tissues in the body. Barbiturates in general may appear in breast milk and readily cross the placental barrier. They are bound to plasma and tissue proteins to a varying degree and binding increases directly as a function of lipid solubility.
Elimination of butalbital is primarily via the kidney (59% to 88% of the dose) as unchanged drug or metabolites. The plasma half-life is about 35 hours. Urinary excretion products include parent drug (about 3.6% of the dose), 5-isobutyl-5-(2,3-dihydroxypropyl) barbituric acid (about 24% of the dose), 5-allyl-5(3-hydroxy-2-methyl-1-propyl) barbituric acid (about 4.8% of the dose), products with the barbituric acid ring hydrolyzed with excretion of urea (about 14% of the dose), as well as unidentified materials. Of the material excreted in the urine, 32% is conjugated. The in vitro plasma protein binding of butalbital is 45% over the concentration range of 0.5 to 20 mcg/mL. This falls within the range of plasma protein binding (20% to 45%) reported with other barbiturates such as phenobarbital, pentobarbital, and secobarbital sodium. The plasma-to-blood concentration ratio was almost unity indicating that there is no preferential; distribution of butalbital into either plasma or blood cells (See OVERDOSAGE for toxicity information).
Acetaminophen : Acetaminophen is rapidly absorbed from the gastrointestinal tract and is distributed throughout most body tissues. The plasma half-life is 1.25 to 3 hours, but may be increased by liver damage and following overdosage. Elimination of acetaminophen is principally by liver metabolism (conjugation) and subsequent renal excretion of metabolites. Approximately 85% of an oral dose appears in the urine within 24 hours of administration, most as the glucuronide conjugate, with small amounts of other conjugates and unchanged drug. (See OVERDOSAGE for toxicity information).
HOW SUPPLIED
Butalbital and Acetaminophen tablets, 50 mg / 325 mg: White to off-white, Capsule shaped bi-convex tablets, debossed "A34" on one side and plain on the other side, in bottles of 100 tablets with child-resistance closure (NDC 35573-456-02).
Butalbital and Acetaminophen tablets, 50 mg / 300 mg: White to off-white, round bi-convex tablets, debossed 'A32' on one side and plain on the other side, in bottles of 100 tablets with child-resistance closure (NDC 35573-455-02).
Storage:
Protect from light and moisture. Store butalbital and acetaminophen tablets at 25°C (77°F); excursions permitted to 15°C-30°C (59°F-86°F).
Dispense in a tight, light-resistant container with a child-resistant closure.
Rx only
Manufactured for:
Burel Pharmaceuticals, LLC
Mason, OH 45040 USA
Code: L7059/00
Rev: 09/2022