Brompheniramine Maleate, Pseudoephedrine Hydrochloride, - Brompheniramine Maleate, Pseudoephedrine Hydrochloride, syrup prescribing information
INDICATIONS AND USAGE
For relief of coughs and upper respiratory symptoms, including nasal congestion, associated with allergy or the common cold.
DOSAGE AND ADMINISTRATION
Adults and pediatric patients 12 years of age and over: 10 mL (2 teaspoonfuls) every 4 hours. Children 6 to under 12 years of age: 5 mL (1 teaspoonful) every 4 hours. Children 2 to under 6 years of age: 2.5 mL (½ teaspoonful) every 4 hours. Infants 6 months to under 2 years of age: Dosage to be established by a physician.
Do not exceed 6 doses during a 24-hour period.
CONTRAINDICATIONS
Hypersensitivity to any of the ingredients. Do not use in the newborn, in premature infants, in nursing mothers, or in patients with severe hypertension or severe coronary artery disease. Do not use dextromethorphan in patients receiving monoamine oxidase (MAOI) inhibitors (see PRECAUTIONS - Drug Interactions).
Antihistamines should not be used to treat lower respiratory tract conditions including asthma.
ADVERSE REACTIONS
The most frequent adverse reactions to brompheniramine maleate, pseudoephedrine hydrochloride, and dextromethorphan hydrobromide syrup are: sedation; dryness of mouth, nose and throat; thickening of bronchial secretions; dizziness. Other adverse reactions may include:
Dermatologic: Urticaria, drug rash, photosensitivity, pruritus.
Cardiovascular System : Hypotension, hypertension, cardiac arrhythmias, palpitation.
CNS: Disturbed coordination, tremor, irritability, insomnia, visual disturbances, weakness, nervousness, convulsions, headache, euphoria, and dysphoria.
G.U. System: Urinary frequency, difficult urination.
G.I. System: Epigastric discomfort, anorexia, nausea, vomiting, diarrhea, constipation.
Respiratory System: Tightness of chest and wheezing, shortness of breath.
Hematologic System: Hemolytic anemia, thrombocytopenia, agranulocytosis.
Drug Interactions
Monoamine oxidase (MAO) inhibitors Hyperpyrexia, hypotension, and death have been reported coincident with the coadministration of MAO inhibitors and products containing dextromethorphan. In addition, MAO inhibitors prolong and intensify the anticholinergic (drying) effects of antihistamines and may enhance the effect of pseudoephedrine. Concomitant administration of brompheniramine maleate, pseudoephedrine hydrochloride, and dextromethorphan hydrobromide syrup and MAO inhibitors should be avoided (see CONTRAINDICATIONS).
Central nervous system (CNS) depressants Antihistamines have additive effects with alcohol and other CNS depressants (hypnotics, sedatives, tranquilizers, antianxiety agents, etc.)
Antihypertensive drugs Sympathomimetic may reduce the effects of antihypertensive drugs.
DESCRIPTION
Brompheniramine maleate, pseudoephedrine hydrochloride, and dextromethorphan hydrobromide syrup, 2 mg/30 mg/10 mg per 5 mL is a clear, light pink syrup with a butterscotch flavour.
Each 5 mL (1 teaspoonful) contains: Brompheniramine Maleate, USP 2 mg Pseudoephedrine Hydrochloride, USP 30 mg Dextromethorphan Hydrobromide, USP 10 mg Alcohol 0.95% v/v In a palatable, aromatic vehicle.
Inactive Ingredients: artificial butterscotch flavor, citric acid anhydrous, alcohol, FD&C Red No. 40, glycerin, sucrose, methylparaben, propylene glycol, purified water and sodium benzoate. It may contain 10% citric acid solution or 10% sodium citrate solution for pH adjustment. The pH range is between 4.0 and 6.0.
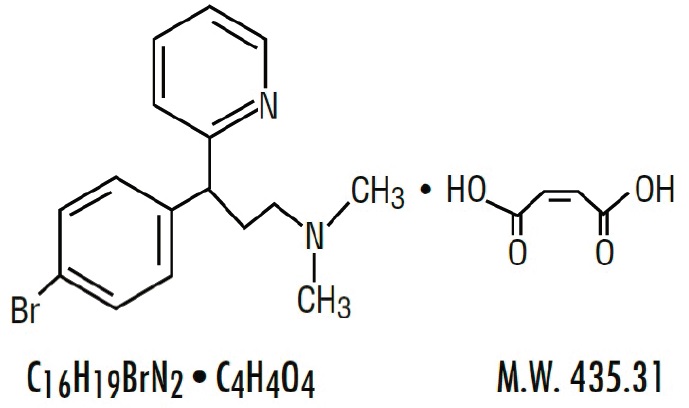
| Brompheniramine Maleate, USP(±)-2- p -Bromo-α-2-(dimethylamino)ethylbenzylpyridine maleate (1:1) |
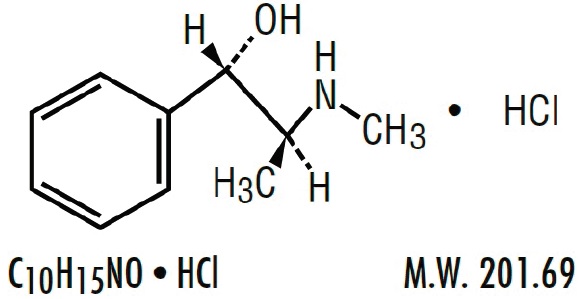
| Pseudoephedrine Hydrochloride, USP (+)-Pseudoephedrine hydrochloride |
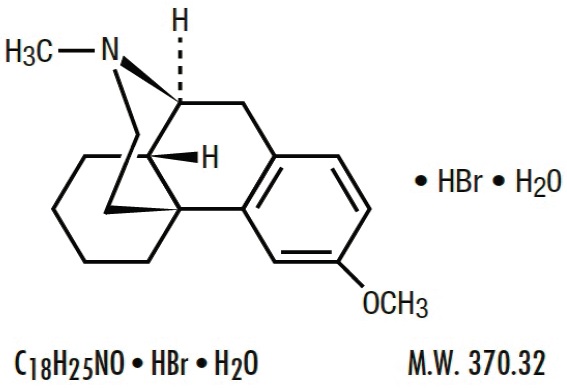
| Dextromethorphan Hydrobromide, USP 3-Methoxy-17-methyl-9α, 13α, 14α-morphinan hydrobromide monohydrate |
Antihistamine/Nasal Decongestant/Antitussive syrup for oral administration.
CLINICAL PHARMACOLOGY
Brompheniramine maleate is a histamine antagonist, specifically an H1-receptor-blocking agent belonging to the alkylamine class of antihistamines. Antihistamines appear to compete with histamine for receptor sites on effector cells. Brompheniramine also has anticholinergic (drying) and sedative effects. Among the antihistaminic effects, it antagonizes the allergic response (vasodilation, increased vascular permeability, increased mucus secretion) of nasal tissue. Brompheniramine is well absorbed from the gastrointestinal tract, with peak plasma concentration after single, oral dose of 4 mg reached in 5 hours; urinary excretion is the major route of elimination, mostly as products of biodegradation; the liver is assumed to be the main site of metabolic transformation.
Pseudoephedrine acts on sympathetic nerve endings and also on smooth muscle, making it useful as a nasal decongestant. The nasal decongestant effect is mediated by the action of pseudoephedrine on α- sympathetic receptors, producing vasoconstriction of the dilated nasal arterioles. Following oral administration, effects are noted within 30 minutes with peak activity occurring at approximately one hour.
Dextromethorphan acts centrally to elevate the threshold for coughing. It has no analgesic or addictive properties. The onset of antitussive action occurs in 15 to 30 minutes after administration and is of long duration.
HOW SUPPLIED
Brompheniramine maleate, pseudoephedrine hydrochloride, and dextromethorphan hydrobromide syrup is a clear, light pink-colored, butterscotch-flavored syrup containing in each 5 mL (1 teaspoonful) brompheniramine maleate 2 mg, pseudoephedrine hydrochloride 30 mg and dextromethorphan hydrobromide 10 mg, available in the following sizes:
4 fl oz (118 mL) NDC 71930-026-18 1 pint (473 mL) NDC 71930-026-43
RECOMMENDED STORAGE Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
KEEP TIGHTLY CLOSED
Dispense in a tight, light-resistant container as defined in the USP
Rx Only
Manufactured by: WES Pharma Inc Westminster, MD – 21157 USA
Distributed by: Eywa Pharma Inc., 2 Research Way, Floor 3, Princeton, NJ 08540
REV. 04/20