Get your patient on Bromocriptine Mesylate - Bromocriptine Mesylate tablet (Bromocriptine Mesylate)
Bromocriptine Mesylate - Bromocriptine Mesylate tablet prescribing information
INDICATIONS AND USAGE
Hyperprolactinemia-Associated Dysfunctions
Bromocriptine mesylate tablets, USP are indicated for the treatment of dysfunctions associated with hyperprolactinemia including amenorrhea with or without galactorrhea, infertility or hypogonadism. Bromocriptine mesylate tablets, USP treatment is indicated in patients with prolactin-secreting adenomas, which may be the basic underlying endocrinopathy contributing to the above clinical presentations. Reduction in tumor size has been demonstrated in both male and female patients with macroadenomas. In cases where adenectomy is elected, a course of bromocriptine mesylate tablets, USP therapy may be used to reduce the tumor mass prior to surgery.
Acromegaly
Bromocriptine mesylate tablets, USP are indicated in the treatment of acromegaly. Bromocriptine mesylate tablets, USP therapy, alone or as adjunctive therapy with pituitary irradiation or surgery, reduces serum growth hormone by 50% or more in approximately one-half of patients treated, although not usually to normal levels.
Since the effects of external pituitary radiation may not become maximal for several years, adjunctive therapy with bromocriptine mesylate tablets, USP offers potential benefit before the effects of irradiation are manifested.
Parkinson's Disease
Bromocriptine mesylate tablets, USP are indicated in the treatment of the signs and symptoms of idiopathic or postencephalitic Parkinson's disease. As adjunctive treatment to levodopa (alone or with a peripheral decarboxylase inhibitor), bromocriptine mesylate tablets, USP therapy may provide additional therapeutic benefits in those patients who are currently maintained on optimal dosages of levodopa, those who are beginning to deteriorate (develop tolerance) to levodopa therapy, and those who are experiencing "end of dose failure" on levodopa therapy. Bromocriptine mesylate tablets, USP therapy may permit a reduction of the maintenance dose of levodopa and, thus may ameliorate the occurrence and/or severity of adverse reactions associated with long-term levodopa therapy such as abnormal involuntary movements (e.g., dyskinesias) and the marked swings in motor function ("on-off" phenomenon). Continued efficacy of bromocriptine mesylate tablets, USP therapy during treatment of more than two years has not been established.
Data are insufficient to evaluate potential benefit from treating newly diagnosed Parkinson's disease with bromocriptine mesylate tablets, USP. Studies have shown, however, significantly more adverse reactions (notably nausea, hallucinations, confusion and hypotension) in bromocriptine mesylate tablets, USP-treated patients than in levodopa/carbidopa-treated patients. Patients unresponsive to levodopa are poor candidates for bromocriptine mesylate tablets, USP therapy.
DOSAGE AND ADMINISTRATION
General
It is recommended that bromocriptine mesylate tablets be taken with food. Patients should be evaluated frequently during dose escalation to determine the lowest dosage that produces a therapeutic response.
Hyperprolactinemic Indications
The initial dosage of bromocriptine mesylate tablets in adults is one-half to one 2.5 mg scored tablet daily. An additional 2.5 mg tablet may be added to the treatment regimen as tolerated every 2 to 7 days until an optimal therapeutic response is achieved. The therapeutic dosage ranged from 2.5 to 15 mg daily in adults studied clinically.
Based on limited data in children of age 11 to 15, (see Pediatric Use ) the initial dose is one-half to one 2.5 mg scored tablet daily. Dosing may need to be increased as tolerated until a therapeutic response is achieved. The therapeutic dosage ranged from 2.5 to 10 mg daily in children with prolactin-secreting pituitary adenomas.
In order to reduce the likelihood of prolonged exposure to bromocriptine mesylate tablets should an unsuspected pregnancy occur, a mechanical contraceptive should be used in conjunction with bromocriptine mesylate tablet therapy until normal ovulatory menstrual cycles have been restored. Contraception may then be discontinued in patients desiring pregnancy.
Thereafter, if menstruation does not occur within 3 days of the expected date, bromocriptine mesylate tablet therapy should be discontinued and a pregnancy test performed.
Acromegaly
Virtually all acromegalic patients receiving therapeutic benefit from bromocriptine mesylate tablets also have reductions in circulating levels of growth hormone. Therefore, periodic assessment of circulating levels of growth hormone will, in most cases, serve as a guide in determining the therapeutic potential of bromocriptine mesylate tablets. If, after a brief trial with bromocriptine mesylate tablet therapy, no significant reduction in growth hormone levels has taken place, careful assessment of the clinical features of the disease should be made, and if no change has occurred, dosage adjustment or discontinuation of therapy should be considered.
The initial recommended dosage is one-half to one 2.5 mg bromocriptine mesylate tablet on retiring (with food) for 3 days. An additional one-half to 1 tablet should be added to the treatment regimen as tolerated every 3 to 7 days until the patient obtains optimal therapeutic benefit. Patients should be reevaluated monthly and the dosage adjusted based on reductions of growth hormone or clinical response. The usual optimal therapeutic dosage range of bromocriptine mesylate tablets varies from 20 to 30 mg/day in most patients. The maximal dosage should not exceed 100 mg/day.
Patients treated with pituitary irradiation should be withdrawn from bromocriptine mesylate tablet therapy on a yearly basis to assess both the clinical effects of radiation on the disease process as well as the effects of bromocriptine mesylate tablet therapy. Usually a 4 to 8 week withdrawal period is adequate for this purpose. Recurrence of the signs/ symptoms or increases in growth hormone indicate the disease process is still active and further courses of bromocriptine mesylate tablets should be considered.
Parkinson's Disease
The basic principle of bromocriptine mesylate tablet therapy is to initiate treatment at a low dosage and, on an individual basis, increase the daily dosage slowly until a maximum therapeutic response is achieved. The dosage of levodopa during this introductory period should be maintained, if possible. The initial dose of bromocriptine mesylate tablets is one-half of a 2.5 mg tablet twice daily with meals. Assessments are advised at two week intervals during dosage titration to ensure that the lowest dosage producing an optimal therapeutic response is not exceeded. If necessary, the dosage may be increased every 14 to 28 days by 2.5 mg/day with meals. Should it be advisable to reduce the dosage of levodopa because of adverse reactions, the daily dosage of bromocriptine mesylate tablets, if increased, should be accomplished gradually in small (2.5 mg) increments.
The safety of bromocriptine mesylate tablets has not been demonstrated in dosages exceeding 100 mg/day.
CONTRAINDICATIONS
Hypersensitivity to bromocriptine or to any of the excipients of bromocriptine mesylate, uncontrolled hypertension and sensitivity to any ergot alkaloids. In patients being treated for hyperprolactinemia, bromocriptine mesylate should be withdrawn when pregnancy is diagnosed (see PRECAUTIONS , Hyperprolactinemic States ). In the event that bromocriptine mesylate is reinstituted to control a rapidly expanding macroadenoma (see PRECAUTIONS , Hyperprolactinemic States ) and a patient experiences a hypertensive disorder of pregnancy, the benefit of continuing bromocriptine mesylate must be weighed against the possible risk of its use during a hypertensive disorder of pregnancy. When bromocriptine mesylate is being used to treat acromegaly, prolactinoma, or Parkinson’s disease in patients who subsequently become pregnant, a decision should be made as to whether the therapy continues to be medically necessary or can be withdrawn. If it is continued, the drug should be withdrawn in those who may experience hypertensive disorders of pregnancy (including eclampsia, preeclampsia, or pregnancy-induced hypertension) unless withdrawal of bromocriptine mesylate is considered to be medically contraindicated.
The drug should not be used during the postpartum period in women with a history of coronary artery disease and other severe cardiovascular conditions unless withdrawal is considered medically contraindicated. If the drug is used in the postpartum period, the patient should be observed with caution.
ADVERSE REACTIONS
Adverse Reactions from Clinical Trials
Hyperprolactinemic Indications
The incidence of adverse effects is quite high (69%) but these are generally mild to moderate in degree. Therapy was discontinued in approximately 5% of patients because of adverse effects. These in decreasing order of frequency are: nausea (49%), headache (19%), dizziness (17%), fatigue (7%), light-headedness (5%), vomiting (5%), abdominal cramps (4%), nasal congestion (3%), constipation (3%), diarrhea (3%), and drowsiness (3%).
A slight hypotensive effect may accompany bromocriptine mesylate treatment. The occurrence of adverse reactions may be lessened by temporarily reducing dosage to 1.25 mg two or three times daily. A few cases of cerebrospinal fluid rhinorrhea have been reported in patients receiving bromocriptine mesylate for treatment of large prolactinomas. This has occurred rarely, usually only in patients who have received previous transsphenoidal surgery, pituitary radiation, or both, and who were receiving bromocriptine mesylate for tumor recurrence. It may also occur in previously untreated patients whose tumor extends into the sphenoid sinus.
Acromegaly
The most frequent adverse reactions encountered in acromegalic patients treated with bromocriptine mesylate were: nausea (18%), constipation (14%), postural/orthostatic hypotension (6%), anorexia (4%), dry mouth/nasal stuffiness (4%), indigestion/dyspepsia (4%), digital vasospasm (3%), drowsiness/ tiredness (3%) and vomiting (2%).
Less frequent adverse reactions (less than 2%) were: gastrointestinal bleeding, dizziness, exacerbation of Raynaud's syndrome, headache and syncope. Rarely (less than 1 %) hair loss, alcohol potentiation, faintness, lightheadedness, arrhythmia, ventricular tachycardia, decreased sleep requirement, visual hallucinations, lassitude, shortness of breath, bradycardia, vertigo, paresthesia, sluggishness, vasovagal attack, delusional psychosis, paranoia, insomnia, heavy headedness, reduced tolerance to cold, tingling of ears, facial pallor and muscle cramps have been reported.
Parkinson's Disease
In clinical trials in which bromocriptine mesylate was administered with concomitant reduction in the dose of levodopa/carbidopa, the most common newly appearing adverse reactions were: nausea, abnormal involuntary movements, hallucinations, confusion, “on-off” phenomenon, dizziness, drowsiness, faintness/ fainting, vomiting, asthenia, abdominal discomfort, visual disturbance, ataxia, insomnia, depression, hypotension, shortness of breath, constipation, and vertigo.
Less common adverse reactions which may be encountered include: anorexia, anxiety, blepharospasm, dry mouth, dysphagia, edema of the feet and ankles, erythromelalgia, epileptiform seizure, fatigue, headache, lethargy, mottling of skin, nasal stuffiness, nervousness, nightmares, paresthesia, skin rash, urinary frequency, urinary incontinence, urinary retention, and rarely, signs and symptoms of ergotism such as tingling of fingers, cold feet, numbness, muscle cramps of feet and legs or exacerbation of Raynaud's syndrome.
Abnormalities in laboratory tests may include elevations in blood urea nitrogen, SGOT, SGPT, GGPT, CPK, alkaline phosphatase and uric acid, which are usually transient and not of clinical significance.
Adverse Reactions from Postmarketing Experience
The following adverse reactions have been reported during postapproval use of bromocriptine mesylate (All Indications Combined). Because adverse reactions from spontaneous reports are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Psychiatric Disorders: Confusion, psychomotor agitation/excitation, hallucinations, psychotic disorders, insomnia, libido increase, hypersexuality, and impulse control/compulsive behaviors (including gambling, spending, and other intense urges).
Nervous System Disorders: Headache, drowsiness, dizziness, dyskinaesia, somnolence, paraesthesia, excess daytime somnolence, sudden onset of sleep.
Eye Disorders: Visual disturbance, vision blurred.
Ear and Labyrinth Disorders: Tinnitus.
Cardiac Disorders: Pericardial effusion, constrictive pericarditis, tachycardia, bradycardia, arrhythmia, cardiac valve fibrosis.
Vascular Disorders: Hypotension, orthostatic hypotension (very rarely leading to syncope), reversible pallor of fingers and toes induced by cold (especially in patients with history of Raynaud's phenomenon)
Respiratory, Thoracic and Mediastinal Disorders: Nasal congestion, pleural effusion, pleural fibrosis, pleurisy, pulmonary fibrosis, dyspnoea.
Gastrointestinal Disorders: Nausea, constipation, vomiting, dry mouth, diarrhea, abdominal pain, retroperitoneal fibrosis, gastrointestinal ulcer, gastrointestinal hemorrhage.
Skin and Subcutaneous Tissue Disorders: Allergic skin reactions, hair loss.
Musculoskeletal and Connective Tissue Disorders: Leg cramps.
General Disorders and Administration Site Conditions: Fatigue, peripheral oedema, a syndrome resembling Neuroleptic Malignant Syndrome on abrupt withdrawal of bromocriptine mesylate, withdrawal symptoms (including apathy, anxiety, depression, fatigue, insomnia, sweating, and pain) with taper or after discontinuation (see PRECAUTIONS ).
Adverse Events Observed in Other Conditions
Postpartum Patients (see above WARNINGS )
In postpartum studies with bromocriptine mesylate, 23 percent of postpartum patients treated had at least 1 side effect, but they were generally mild to moderate in degree. Therapy was discontinued in approximately 3% of patients. The most frequently occurring adverse reactions were: headache (10%), dizziness (8%), nausea (7%), vomiting (3%), fatigue (1.0%), syncope (0.7%), diarrhea (0.4%) and cramps (0.4%). Decreases in blood pressure (≥ 20 mm Hg systolic and ≥10 mm Hg diastolic) occurred in 28% of patients at least once during the first 3 postpartum days; these were usually of a transient nature. Reports of fainting in the puerperium may possibly be related to this effect. In postmarketing experience in the U.S., serious adverse reactions reported include 72 cases of seizures (including 4 cases of status epilepticus), 30 cases of stroke, and 9 cases of myocardial infarction among postpartum patients. Seizure cases were not necessarily accompanied by the development of hypertension. An unremitting and often progressively severe headache, sometimes accompanied by visual disturbance, often preceded by hours to days many cases of seizure and/or stroke. Most patients had shown no evidence of any of the hypertensive disorders of pregnancy including eclampsia, preeclampsia or pregnancy-induced hypertension. One stroke case was associated with sagittal sinus thrombosis, and another was associated with cerebral and cerebellar vasculitis. One case of myocardial infarction was associated with unexplained disseminated intravascular coagulation and a second occurred in conjunction with use of another ergot alkaloid. The relationship of these adverse reactions to bromocriptine mesylate administration has not been established.
In rare cases serious adverse events, including hypertension, myocardial infarction, seizures, stroke, or psychic disorders have been reported in postpartum women treated with bromocriptine mesylate. In some patients the development of seizures or stroke was preceded by severe headache and/or transient visual disturbances. Although the causal relationship of these events to the drug is uncertain, periodic monitoring of blood pressure is advisable in postpartum women receiving bromocriptine mesylate. If hypertension, severe, progressive, or unremitting headache (with or without visual disturbances), or evidence of CNS toxicity develop, the administration of bromocriptine mesylate should be discontinued and the patient should be evaluated promptly.
Particular caution is required in patients who have recently been treated or are on concomitant therapy with drugs that can alter blood pressure, e.g., vasoconstrictors such as sympathomimetics or ergot alkaloids including ergometrine or methylergometrine and their concomitant use in the puerperium is not recommended.
To report SUSPECTED ADVERSE REACTIONS, contact Sandoz Inc. at 1-800-525-8747 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
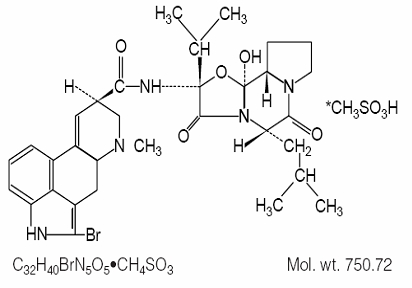
DESCRIPTION
Bromocriptine mesylate is an ergot derivative with potent dopamine receptor agonist activity.
Bromocriptine mesylate is chemically designated as Ergotaman-3´,6´,18-trione, 2-bromo-12´-hydroxy-2´-(1-methylethyl)-5´-(2-methylpropyl)-, (5’α)-monomethanesulfonate (salt).
The structural formula is:
2.5 mg Tablets
Each tablet for oral administration contains bromocriptine mesylate equivalent to 2.5 mg bromocriptine. In addition, each tablet contains the following inactive ingredients: colloidal silicon dioxide, corn starch, disodium edetate, lactose monohydrate, magnesium stearate, maleic acid, and povidone.
CLINICAL PHARMACOLOGY
Bromocriptine mesylate is a dopamine receptor agonist, which activates post-synaptic dopamine receptors. The dopaminergic neurons in the tuberoinfundibular process modulate the secretion of prolactin from the anterior pituitary by secreting a prolactin inhibitory factor (thought to be dopamine); in the corpus striatum the dopaminergic neurons are involved in the control of motor function. Clinically, bromocriptine mesylate significantly reduces plasma levels of prolactin in patients with physiologically elevated prolactin as well as in patients with hyperprolactinemia. The inhibition of physiological lactation as well as galactorrhea in pathological hyperprolactinemic states is obtained at dose levels that do not affect secretion of other tropic hormones from the anterior pituitary. Experiments have demonstrated that bromocriptine induces long-lasting stereotyped behavior in rodents and turning behavior in rats having unilateral lesions in the substantia nigra. These actions, characteristic of those produced by dopamine, are inhibited by dopamine antagonists and suggest a direct action of bromocriptine on striatal dopamine receptors.
Bromocriptine mesylate is a nonhormonal, nonestrogenic agent that inhibits the secretion of prolactin in humans, with little or no effect on other pituitary hormones, except in patients with acromegaly, where it lowers elevated blood levels of growth hormone in the majority of patients.
Bromocriptine mesylate produces its therapeutic effect in the treatment of Parkinson’s disease, a clinical condition characterized by a progressive deficiency in dopamine synthesis in the substantia nigra, by directly stimulating the dopamine receptors in the corpus striatum. In contrast, levodopa exerts its therapeutic effect only after conversion to dopamine by the neurons of the substantia nigra, which are known to be numerically diminished in this patient population.
Pharmacokinetics
Absorption
Following single dose administration of bromocriptine mesylate tablets, 2 x 2.5 mg to 5 healthy volunteers under fasted conditions, the mean peak plasma levels of bromocriptine, time to reach peak plasma concentrations and elimination half-life were 465 pg/mL ± 226, 2.5 hrs ± 2 and 4.85 hr, respectively. 1 Linear relationship was found between single doses of bromocriptine mesylate and C max and AUC in the dose range of 1 to 7.5 mg. 2 The pharmacokinetics of bromocriptine metabolites have not been reported.
Food did not significantly affect the systemic exposure of bromocriptine following administration of bromocriptine mesylate tablets, 2.5 mg. 3 It is recommended that bromocriptine mesylate be taken with food because of the high percentage of subjects who vomit upon receiving bromocriptine under fasting conditions.
Following bromocriptine mesylate, 5 mg administered twice daily for 14 days, the bromocriptine C max and AUC at steady-state were 628 ± 375 pg/mL and 2377 ± 1186 pg•hr/mL, respectively. 4
Distribution
In vitro experiments showed that bromocriptine was 90%-96% bound to serum albumin.
Metabolism
Bromocriptine undergoes extensive first-pass biotransformation, reflected by complex metabolite profiles and by almost complete absence of parent drug in urine and feces.
In vitro studies using human liver microsomes showed that bromocriptine has a high affinity for CYP3A and hydroxylations at the proline ring of the cyclopeptide moiety constituted a main metabolic pathway. 5 Inhibitors and/or potent substrates for CYP3A4 might therefore inhibit the clearance of bromocriptine and lead to increased levels (see PRECAUTIONS , Drug Interactions section). The participation of other major CYP enzymes such as 2D6, 2C8, and 2C19 on the metabolism of bromocriptine has not been evaluated. Bromocriptine is also an inhibitor of CYP3A4 with a calculated IC50 value of 1.69 μM. 6 Given the low therapeutic concentrations of bromocriptine in patients (C max =0.82 nM), a significant alteration of the metabolism of a second drug whose clearance is mediated by CYP3A4 should not be expected. The potential effect of bromocriptine and its metabolites to act as inducers of CYP enzymes has not been reported.
Excretion
About 82% and 5.6 % of the radioactive dose orally administered was recovered in feces and urine, respectively. Bromolysergic acid and bromoisolysergic acid accounted for half of the radioactivity in urine. 5
_______________________
1 Nelson, M. et. al. (1990). Pharmacokinetic evaluation of erythromycin and caffeine administered with bromocriptine. Clin Pharmacol Ther; 47(6):694-7.
2 Schran, H.F., Bhuta, S.I., Schwartz, et al. (1980). The pharmacokinetics of bromocriptine in man. In: Golstein, M. Calne, D.B.,et. Al (eds). Ergot compound and brain function: Neuroendocrine and neuropsychiatric aspects, pp. 125-139, New York, Rave Press.
3 Kopitar, Z., Vrhovac, B., Povsic, L., Plavsic, F., Francetic, I., Urbancic, J. (1991). The effect of food and metoclopramide on the pharmacokinetics and side effects of bromocriptine. Eur J Drug Metab Pharmacokinet; 16(3):177-81
4 Flogstad, A.K., Halse, J., Grass, P., Abisch, E., Djoseland, O., Kutz, K., Bodd, E., and Jervell, J., (1994). A comparison of octreotide, bromocriptine, or a combination of both drugs in acromegaly. Journal of Clinical Endocrinology & Metabolism; Vol 79, 461-465
5 Peyronneau MA, Delaforge M, Riviere R et al. 1994. High affinity of ergopeptides for CYP P450 3A. Importance of their peptide moiety for P450 recognition and hydroxylation of bromocriptine. Eur J Biochem 223:947-56.
6 Wynalda, M.A., Wienkers, L.C. (1997). Assessment of potential interactions between dopamine receptor agonists and various human cytochrome P450 enzymes using a simple in vitro inhibition screen. Drug Metab Dispos; 25:1211-14.
Specific Populations
Effect of Renal Impairment
The effect of renal function on the pharmacokinetics of bromocriptine has not been evaluated.
Since parent drug and metabolites are almost completely excreted via metabolism, and only 6% eliminated via the kidney, renal impairment may not have a significant impact on the PK of bromocriptine and its metabolites (see PRECAUTIONS , General ).
Effect of Hepatic Impairment
The effect of liver impairment on the PK of bromocriptine and its metabolites has not been evaluated. Since bromocriptine is mainly eliminated by metabolism, liver impairment may increase the plasma levels of bromocriptine, therefore, caution may be necessary (see PRECAUTIONS , General ).
The effect of age, race, and gender on the pharmacokinetics of bromocriptine and its metabolites has not been evaluated.
Clinical Studies
In about 75% of cases of amenorrhea and galactorrhea, bromocriptine mesylate therapy suppresses the galactorrhea completely, or almost completely, and reinitiates normal ovulatory menstrual cycles.
Menses are usually reinitiated prior to complete suppression of galactorrhea; the time for this on average is 6 to 8 weeks. However, some patients respond within a few days, and others may take up to 8 months.
Galactorrhea may take longer to control depending on the degree of stimulation of the mammary tissue prior to therapy. At least a 75% reduction in secretion is usually observed after 8 to 12 weeks. Some patients may fail to respond even after 12 months of therapy.
In many acromegalic patients, bromocriptine mesylate produces a prompt and sustained reduction in circulating levels of serum growth hormone.
Clinical Studies
In about 75% of cases of amenorrhea and galactorrhea, bromocriptine mesylate therapy suppresses the galactorrhea completely, or almost completely, and reinitiates normal ovulatory menstrual cycles.
Menses are usually reinitiated prior to complete suppression of galactorrhea; the time for this on average is 6 to 8 weeks. However, some patients respond within a few days, and others may take up to 8 months.
Galactorrhea may take longer to control depending on the degree of stimulation of the mammary tissue prior to therapy. At least a 75% reduction in secretion is usually observed after 8 to 12 weeks. Some patients may fail to respond even after 12 months of therapy.
In many acromegalic patients, bromocriptine mesylate produces a prompt and sustained reduction in circulating levels of serum growth hormone.
HOW SUPPLIED
Bromocriptine mesylate tablets, USP are available as follows:
2.5 mg white to yellowish white round tablets, scored on one side with debossed code BCT 2 1/2 on reverse side.
NDC 0781-5325-31 bottle of 30 tablets
NDC 0781-5325-01 bottle of 100 tablets
Store and Dispense
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature]. Protect from moisture. Dispense in a tight, light-resistant container.
Manufactured in Slovenia
by Novartis d.o.o.
for Sandoz Inc., Princeton, NJ 08540
Rev. June 2024