Get your patient on Bromfenac Ophthalmic Solution - Bromfenac Sodium solution/ Drops (Bromfenac Sodium)
Bromfenac Ophthalmic Solution - Bromfenac Sodium solution/ Drops prescribing information
INDICATIONS AND USAGE
Bromfenac Ophthalmic Solution 0.07% is indicated for the treatment of postoperative inflammation and reduction of ocular pain in patients who have undergone cataract surgery.
DOSAGE AND ADMINISTRATION
Instill one drop into the affected eye once daily beginning 1 day prior to surgery, continued on the day of surgery, and through the first 14 days postsurgery. (2.1 )
Recommended Dosage
Apply one drop to the affected eye once daily beginning 1 day prior to cataract surgery, continued on the day of surgery, and through the first 14 days of the postoperative period.
Use with Other Topical Ophthalmic Medications
Bromfenac Ophthalmic Solution may be administered in conjunction with other topical ophthalmic medications such as alpha-agonists, beta-blockers, carbonic anhydrase inhibitors, cycloplegics, and mydriatics. Drops should be administered at least 5 minutes apart.
DOSAGE FORMS AND STRENGTHS
Ophthalmic solution: bromfenac 0.07%
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary
There are no available data on Bromfenac Ophthalmic Solution use in pregnant women to evaluate a drug-associated risk of major birth defects, miscarriage or other adverse maternal or fetal outcomes.
The systemic exposure to bromfenac following topical ocular administration is low [see Clinical Pharmacology (12.3 )]. Consequently, the systemic exposure of a pregnant woman to bromfenac is expected to be minimal following topical ocular administration.
However, because of the known effects of prostaglandin biosynthesis-inhibiting drugs on the fetal cardiovascular system (closure of ductus arteriosus), the use of Bromfenac Ophthalmic Solution during late pregnancy should be avoided.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Premature closure of the ductus arteriosus in the fetus has occurred with third trimester use of oral and injectable NSAIDs. Measurable maternal and fetal plasma drug levels are available with oral and injectable routes of NSAID administration. The maternal plasma level of Bromfenac Ophthalmic Solution following ocular administration is unknown [see Clinical Pharmacology (12.3 )].
Data
Animal Data
Embryo-fetal lethality and maternal toxicity were produced in rats and rabbits treated with bromfenac during the period of organogenesis at oral doses up to 0.9 mg/kg/day and 7.5 mg/kg/day, respectively. These doses corresponded to a Cmax 90- and 150- times the predicted Cmax at the recommended human ophthalmic dose (RHOD), respectively. In rats, bromfenac treatment caused delayed parturition at 0.3 mg/kg/day (30 times the predicted human Cmax at the RHOD), and caused dystocia, increased neonatal mortality, and reduced postnatal growth at 0.9 mg/kg/day (90 times the predicted human Cmax at the RHOD).
Lactation
There are no data on the presence of bromfenac in human milk, the effects on the breastfed infant, or the effects on milk production.
The systemic exposure of a breastfeeding woman to bromfenac is expected to be minimal following topical ocular administration, however, the possibility of harm to the breastfed infant cannot be ruled out.
The developmental and health benefits of breastfeeding should be considered, along with the mother’s clinical need for Bromfenac Ophthalmic Solution, and any potential adverse effects on the breastfed infant from Bromfenac Ophthalmic Solution or from the underlying maternal conditions.
Pediatric Use
The safety and effectiveness of Bromfenac Ophthalmic Solution have not been established in pediatric patients.
Geriatric Use
No overall differences in safety or effectiveness of Bromfenac Ophthalmic Solution have been observed between patients 70 years of age and older and younger adult patients.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
Sulfite Allergic Reactions
Bromfenac Ophthalmic Solution contains sodium sulfite, a sulfite that may cause allergic-type reactions including anaphylactic symptoms and life-threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in non-asthmatic people.
Slow or Delayed Healing
All topical nonsteroidal anti-inflammatory drugs (NSAIDs), including bromfenac, may slow or delay healing. Topical corticosteroids are also known to slow or delay healing. Concomitant use of topical NSAIDs and topical steroids may increase the potential for healing problems.
Potential for Cross-Sensitivity
There is the potential for cross-sensitivity to acetylsalicylic acid, phenylacetic acid derivatives, and other NSAIDs, including bromfenac. Therefore, caution should be used when treating individuals who have previously exhibited sensitivities to these drugs.
Increased Bleeding Time
With some NSAIDs, including bromfenac, there exists the potential for increased bleeding time due to interference with platelet aggregation. There have been reports that ocularly applied NSAIDs may cause increased bleeding of ocular tissues (including hyphemas) in conjunction with ocular surgery.
It is recommended that Bromfenac Ophthalmic Solution be used with caution in patients with known bleeding tendencies or who are receiving other medications which may prolong bleeding time.
Keratitis and Corneal Reactions
Use of topical NSAIDs, including bromfenac, may result in keratitis. In some susceptible patients, continued use of topical NSAIDs may result in epithelial breakdown, corneal thinning, corneal erosion, corneal ulceration or corneal perforation. These events may be sight threatening. Patients with evidence of corneal epithelial breakdown should immediately discontinue use of topical NSAIDs, including bromfenac, and should be closely monitored for corneal health.
Postmarketing experience with topical NSAIDs suggests that patients with complicated ocular surgeries, corneal denervation, corneal epithelial defects, diabetes mellitus, ocular surface diseases (e.g., dry eye syndrome), rheumatoid arthritis, or repeat ocular surgeries within a short period of time may be at increased risk for corneal adverse events which may become sight threatening. Topical NSAIDs should be used with caution in these patients.
Postmarketing experience with topical NSAIDs also suggests that use more than 24 hours prior to surgery or use beyond 14 days postsurgery may increase patient risk for the occurrence and severity of corneal adverse events.
Risk of Contamination
Do not touch dropper tip to the eye, eyelids, or to any surface, as this may contaminate the contents. Replace the bottle cap after using.
Contact Lens Wear
Bromfenac Ophthalmic Solution should not be instilled while wearing contact lenses. Remove contact lenses prior to instillation of Bromfenac Ophthalmic Solution. The preservative in Bromfenac Ophthalmic Solution, benzalkonium chloride, may be absorbed by soft contact lenses. Lenses may be reinserted after 10 minutes following administration of Bromfenac Ophthalmic Solution.
ADVERSE REACTIONS
The most commonly reported adverse reactions in 3% to 8% of patients were anterior chamber inflammation, foreign body sensation, eye pain, photophobia, and blurred vision. (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at, 1-800-553-5340 or FDA, at 1-800-FDA-1088 or www.fda.gov/medwatch.
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most commonly reported adverse reactions following use of Bromfenac Ophthalmic Solution following cataract surgery include: anterior chamber inflammation, foreign body sensation, eye pain, photophobia, and vision blurred. These reactions were reported in 3% to 8% of patients.
DESCRIPTION
Bromfenac Ophthalmic Solution 0.07% is a sterile, nonsteroidal anti-inflammatory drug (NSAID) for topical ophthalmic use. Each mL of Bromfenac Ophthalmic Solution contains 0.805 mg bromfenac sodium sesquihydrate (equivalent to 0.7 mg bromfenac free acid). The USAN name for bromfenac sodium sesquihydrate is bromfenac sodium. Bromfenac sodium is designated chemically as sodium [2-amino-3-(4-bromobenzoyl) phenyl] acetate sesquihydrate, with an empirical formula of C 15 H 11 BrNNaO 3 • 1½H 2 O. The chemical structure for bromfenac sodium sesquihydrate is:
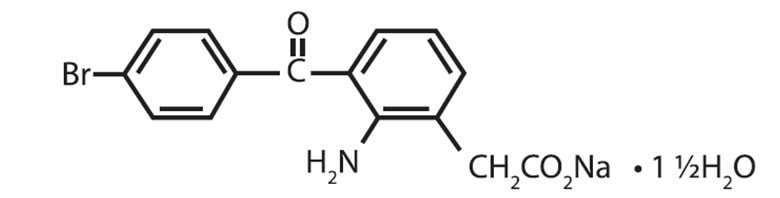
Bromfenac sodium is a yellow to orange crystalline powder. The molecular weight of bromfenac sodium is 383.17. Bromfenac Ophthalmic Solution is supplied as a sterile aqueous 0.07% solution, with a pH of 7.8. The osmolality of Bromfenac Ophthalmic Solution is approximately 300 mOsmol/kg.
Each mL of Bromfenac Ophthalmic Solution contains:
Active: Each mL contains bromfenac sodium sesquihydrate 0.0805%, which is equivalent to bromfenac free acid 0.07%.
Inactives: boric acid, edetate disodium, povidone, sodium borate, sodium sulfite, tyloxapol, sodium hydroxide to adjust pH, and water for injection, USP.
Preservative: benzalkonium chloride 0.005%
CLINICAL PHARMACOLOGY
Mechanism of Action
Bromfenac is a nonsteroidal anti-inflammatory drug (NSAID) that has anti-inflammatory activity. The mechanism of its action is thought to be due to its ability to block prostaglandin synthesis by inhibiting cyclooxygenase (COX) 1 and 2. Prostaglandins have been shown in many animal models to be mediators of certain kinds of intraocular inflammation. In studies performed in animal eyes, prostaglandins have been shown to produce disruption of the blood-aqueous humor barrier, vasodilation, increased vascular permeability, leukocytosis, and increased intraocular pressure.
Pharmacokinetics
The plasma concentration of bromfenac following ocular administration of Bromfenac Ophthalmic Solution 0.07% in humans is unknown. Based on the maximum proposed dose of one drop to each eye (0.035 mg) and PK information from other routes of administration, the systemic concentration of bromfenac is estimated to be below the limit of quantification (50 ng/mL) at steady-state in humans.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Long-term carcinogenicity studies in rats and mice given oral doses of bromfenac up to 0.6 mg/kg/day (systemic exposure 30 times the systemic exposure predicted from RHOD assuming the human systemic concentration is at the limit of quantification) and 5 mg/kg/day (340 times the predicted human systemic exposure), respectively, revealed no significant increases in tumor incidence.
Mutagenesis
Bromfenac did not show mutagenic potential in various mutagenicity studies, including the reverse mutation, chromosomal aberration, and micronucleus tests.
Impairment of Fertility
Bromfenac did not impair fertility when administered orally to male and female rats at doses up to 0.9 mg/kg/day and 0.3 mg/kg/day, respectively (systemic exposure 90 and 30 times the predicted human exposure, respectively).
CLINICAL STUDIES
Ocular Inflammation and Pain
Bromfenac 0.07% QD for the treatment of postoperative inflammation and reduction of ocular pain was evaluated in two multi-center, randomized, double-masked, parallel-group, and placebo (vehicle)-controlled studies. Patients undergoing cataract surgery self-administered bromfenac 0.07% or vehicle once daily, beginning 1 day prior to surgery, continuing on the morning of surgery and for 14 days after surgery. Complete clearance of ocular inflammation (0 cell and no flare) was assessed on Days 1, 3, 8, and 15 postsurgery using slit lamp biomicroscopy. The pain score was self-reported. The primary efficacy endpoint was the proportion of subjects who had complete clearance of ocular inflammation by Day 15. In the intent-to-treat analyses from both assessments, complete clearance at Day 8 and Day 15, bromfenac 0.07% was superior to vehicle as shown in the following table.
Proportion of Subjects with Cleared Ocular Inflammation (0 cell and no flare) | ||||
Study | Visit | Bromfenac 0.07% | Vehicle | Difference (%) (Asymptotic 95% CI) |
Study 1 | At Day 8 | 27/112 (24.1%) | 7/108 (6.5%) | 17.6 (8.4, 26.8) |
At Day 15 | 51/112 (45.5%) | 14/108 (13.0%) | 32.5 (21.4, 43.8) | |
Study 2 | At Day 8 | 33/110 (30.0%) | 14/110 (12.7%) | 17.3 (6.7, 27.9) |
At Day 15 | 50/110 (45.5%) | 30/110 (27.3%) | 18.2 (5.7, 30.7) | |
Proportion of Subjects Who Were Pain Free | ||||
Study | Visit | Bromfenac 0.07% | Vehicle | Difference (%) (Asymptotic 95% CI) |
Study 1 | At Day 1 | 91/112 (81.3%) | 47/108 (43.5%) | 37.7 (25.9, 49.6) |
Study 2 | At Day 1 | 84/110 (76.4%) | 61/110 (55.5%) | 20.9 (8.7, 33.1) |
HOW SUPPLIED/STORAGE AND HANDLING
Bromfenac Ophthalmic Solution 0.07% is supplied in a white LDPE plastic squeeze bottle with a 15 mm LDPE white dropper tip and 15 mm polypropylene gray cap as follows:
- NDC 75907-093-03 3 mL in a 7.5 mL bottle
Storage
Store at 15ºC to 25ºC (59ºF to 77ºF). After opening, Bromfenac Ophthalmic Solution can be used until the expiration date on the bottle.
Mechanism of Action
Bromfenac is a nonsteroidal anti-inflammatory drug (NSAID) that has anti-inflammatory activity. The mechanism of its action is thought to be due to its ability to block prostaglandin synthesis by inhibiting cyclooxygenase (COX) 1 and 2. Prostaglandins have been shown in many animal models to be mediators of certain kinds of intraocular inflammation. In studies performed in animal eyes, prostaglandins have been shown to produce disruption of the blood-aqueous humor barrier, vasodilation, increased vascular permeability, leukocytosis, and increased intraocular pressure.