Get your patient on Bicillin Cr - Penicillin G Benzathine And Penicillin G Procaine injection, Suspension (Penicillin G Benzathine And Penicillin G Procaine)
Bicillin Cr - Penicillin G Benzathine And Penicillin G Procaine injection, Suspension prescribing information
WARNING: NOT FOR INTRAVENOUS USE. DO NOT INJECT INTRAVENOUSLY OR ADMIX WITH OTHER INTRAVENOUS SOLUTIONS. THERE HAVE BEEN REPORTS OF INADVERTENT INTRAVENOUS ADMINISTRATION OF PENICILLIN G BENZATHINE WHICH HAS BEEN ASSOCIATED WITH CARDIORESPIRATORY ARREST AND DEATH. Prior to administration of this drug, carefully read the WARNINGS , ADVERSE REACTIONS , and DOSAGE AND ADMINISTRATION sections of the labeling.
INDICATIONS AND USAGE
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Bicillin C-R and other antibacterial drugs, Bicillin C-R should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
This drug is indicated in the treatment of moderately severe infections due to penicillin-G-susceptible microorganisms that are susceptible to serum levels common to this particular dosage form. Therapy should be guided by bacteriological studies (including susceptibility testing) and by clinical response.
Bicillin C-R is indicated in the treatment of the following in adults and pediatric patients:
Moderately severe to severe infections of the upper-respiratory tract, scarlet fever, erysipelas, and skin and soft-tissue infections due to susceptible streptococci.
NOTE: Streptococci in Groups A, C, G, H, L, and M are very sensitive to penicillin G. Other groups, including Group D (enterococci), are resistant. Penicillin G sodium or potassium is recommended for streptococcal infections with bacteremia.
Moderately severe pneumonia and otitis media due to susceptible Streptococcus pneumoniae .
NOTE: Severe pneumonia, empyema, bacteremia, pericarditis, meningitis, peritonitis, and arthritis of pneumococcal etiology are better treated with penicillin G sodium or potassium during the acute stage.
When high, sustained serum levels are required, penicillin G sodium or potassium, either IM or IV, should be used. This drug should not be used in the treatment of venereal diseases, including syphilis, gonorrhea, yaws, bejel, and pinta.
DOSAGE AND ADMINISTRATION
Streptococcal Infections Group A
Infections of the upper-respiratory tract, skin and soft-tissue infections, scarlet fever, and erysipelas.
The following doses are recommended:
Adults and pediatric patients over 60 lbs. in weight: 2,400,000 units.
Pediatric patients from 30 to 60 lbs.: 900,000 units to 1,200,000 units.
Pediatric patients under 30 lbs.: 600,000 units.
NOTE: Treatment with the recommended dosage is usually given at a single session using multiple IM sites when indicated. An alternative dosage schedule may be used, giving one-half (1/2) the total dose on day 1 and one-half (1/2) on day 3. This will also insure the penicillinemia required over a 10-day period; however, this alternate schedule should be used only when the physician can be assured of the patient's cooperation.
Pneumococcal Infections (except pneumococcal meningitis)
600,000 units in pediatric patients and 1,200,000 units in adults, repeated every 2 or 3 days until the temperature is normal for 48 hours. Other forms of penicillin may be necessary for severe cases.
Method of Administration
Bicillin C-R is intended for Intramuscular Injection ONLY. Do not inject into or near an artery or nerve, or intravenously or admix with other intravenous solutions. (See WARNINGS section).
Administer by DEEP INTRAMUSCULAR INJECTION in the upper, outer quadrant of the buttock (dorsogluteal) or the ventrogluteal site. In neonates, infants and small children, the midlateral aspect of the thigh may be preferable. Administration in the anterolateral thigh is not recommended due to the adverse effects observed (see WARNINGS section), and vascularity of this region. When doses are repeated, vary the injection site.
Because of the high concentration of suspended material in this product, the needle may be blocked if the injection is not made at a slow, steady rate.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit.
CONTRAINDICATIONS
A previous hypersensitivity reaction to any penicillin or to procaine is a contraindication.
ADVERSE REACTIONS
As with other penicillins, untoward reactions of the sensitivity phenomena are likely to occur, particularly in individuals who have previously demonstrated hypersensitivity to penicillins or in those with a history of allergy, asthma, hay fever, or urticaria.
The following adverse reactions have been reported with Bicillin C-R during post-marketing experience:
Skin and Appendages: Stevens-Johnson syndrome (SJS) and drug reaction with eosinophilia and systemic symptoms (DRESS) (See WARNINGS .)
The following have been reported with parenteral penicillin G (the active moiety in Bicillin C-R):
General: Hypersensitivity reactions including the following: skin eruptions (maculopapular to exfoliative dermatitis), urticaria, laryngeal edema, fever, eosinophilia; other serum sickness-like reactions (including chills, fever, edema, arthralgia, and prostration); and anaphylaxis including shock and death: severe cutaneous adverse reactions (SCAR), such as toxic epidermal necrolysis (TEN) and acute generalized exanthematous pustulosis (AGEP) (See WARNINGS .) Note: Urticaria, other skin rashes, and serum sickness-like reactions may be controlled with antihistamines and, if necessary, systemic corticosteroids. Whenever such reactions occur, penicillin G should be discontinued unless, in the opinion of the physician, the condition being treated is life-threatening and amenable only to therapy with penicillin G. Serious anaphylactic reactions require immediate emergency treatment with epinephrine. Oxygen, intravenous steroids, and airway management, including intubation, should also be administered as indicated.
Gastrointestinal: Pseudomembranous colitis. Onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment. (See WARNINGS section.)
Hematologic: Hemolytic anemia, leukopenia, thrombocytopenia.
Neurologic: Neuropathy.
Urogenital: Nephropathy.
The following adverse events have been temporally associated with parenteral administrations of penicillin G benzathine (a component of Bicillin C-R):
Body as a Whole: Hypersensitivity reactions including allergic vasculitis, pruritis, fatigue, asthenia, and pain; aggravation of existing disorder; headache, Nicolau syndrome.
Cardiovascular: Cardiac arrest; hypotension; tachycardia; palpitations; pulmonary hypertension; pulmonary embolism; vasodilation; vasovagal reaction; cerebrovascular accident; syncope.
Gastrointestinal: Nausea, vomiting; blood in stool; intestinal necrosis.
Hemic and Lymphatic: Lymphadenopathy.
Immune System Disorders: Acute myocardial ischemia with or without myocardial infarction may occur as part of an allergic reaction (Kounis syndrome).
Injection Site: Injection site reactions including pain, inflammation, lump, abscess, necrosis, edema, hemorrhage, cellulitis, hypersensitivity, atrophy, ecchymosis, and skin ulcer. Neurovascular reactions including warmth, vasospasm, pallor, mottling, gangrene, numbness of the extremities, cyanosis of the extremities, and neurovascular damage.
Metabolic: Elevated BUN, creatinine, and SGOT.
Musculoskeletal: Joint disorder, periostitis; exacerbation of arthritis; myoglobinuria; rhabdomyolysis.
Nervous System: Nervousness; tremors; dizziness; somnolence; confusion; anxiety; euphoria; transverse myelitis; seizures; coma. A syndrome manifested by a variety of CNS symptoms such as severe agitation with confusion, visual and auditory hallucinations, and a fear of impending death (Hoigne's syndrome), has been reported after administration of penicillin G procaine and, less commonly, after injection of the combination of penicillin G benzathine and penicillin G procaine. Other symptoms associated with this syndrome, such as psychosis, seizures, dizziness, tinnitus, cyanosis, palpitations, tachycardia, and/or abnormal perception in taste, also may occur.
Respiratory: Hypoxia; apnea; dyspnea.
Skin: Diaphoresis.
Special Senses: Blurred vision; blindness.
Urogenital: Neurogenic bladder; hematuria; proteinuria; renal failure; impotence; priapism.
Drug Interactions
Tetracycline, a bacteriostatic antibiotic, may antagonize the bactericidal effect of penicillin, and concurrent use of these drugs should be avoided.
Concurrent administration of penicillin and probenecid increases and prolongs serum penicillin levels by decreasing the apparent volume of distribution and slowing the rate of excretion by competitively inhibiting renal tubular secretion of penicillin.
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
| Class | Examples |
|---|---|
Nitrates/Nitrites | nitroglycerin, nitroprusside, nitric oxide, nitrous oxide |
Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
Antineoplastic agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
Antimalarials | chloroquine, primaquine |
Anticonvulsants | phenobarbital, sodium valproate |
Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
DESCRIPTION
Bicillin C-R (penicillin G benzathine and penicillin G procaine injectable suspension) contains equal amounts of the benzathine and procaine salts of penicillin G. It is available for deep intramuscular injection.
Penicillin G benzathine is prepared by the reaction of dibenzylethylene diamine with two molecules of penicillin G. It is chemically designated as (2 S ,5 R ,6 R )-3,3-Dimethyl-7-oxo-6-(2-phenylacetamido)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid compound with N,N' -dibenzylethylenediamine (2:1), tetrahydrate. It occurs as a white, crystalline powder and is very slightly soluble in water and sparingly soluble in alcohol. Its chemical structure is as follows:
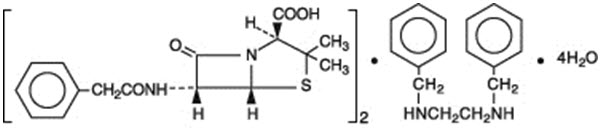
Molecular Formula (C 16 H 18 N 2 O 4 S) 2 ∙ C 16 H 20 N 2 ∙ 4H 2 O | Molecular Wt. 981.19 |
Penicillin G procaine, (2 S ,5 R ,6 R )-3,3-Dimethyl-7-oxo-6-(2-phenylacetamido)-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid compound with 2-(diethylamino)ethyl p-aminobenzoate (1:1) monohydrate, is an equimolar salt of procaine and penicillin G. It occurs as white crystals or a white, microcrystalline powder and is slightly soluble in water. Its chemical structure is as follows:
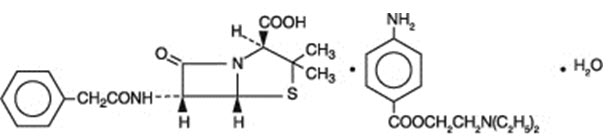
Molecular Formula C 16 H 18 N 2 O 4 S ∙ C 13 H 20 N 2 O 2 ∙ H 2 O | Molecular Wt. 588.72 |
Each disposable syringe (2 mL size) contains the equivalent of 1,200,000 units of penicillin G comprising: the equivalent of 600,000 units of penicillin G as the benzathine salt and the equivalent of 600,000 units of penicillin G as the procaine salt in a stabilized aqueous suspension with sodium citrate buffer; and as w/v, approximately 0.5% lecithin, 0.55% carboxymethylcellulose, 0.55% povidone, 0.1% methylparaben, and 0.01% propylparaben.
Bicillin C-R injectable suspension in the disposable-syringe formulation is viscous and opaque. Read CONTRAINDICATIONS , WARNINGS , PRECAUTIONS , and DOSAGE AND ADMINISTRATION sections prior to use.
CLINICAL PHARMACOLOGY
General
Penicillin G benzathine and penicillin G procaine have a low solubility and, thus, the drugs are slowly released from intramuscular injection sites. The drugs are hydrolyzed to penicillin G. This combination of hydrolysis and slow absorption results in blood serum levels much lower but more prolonged than other parenteral penicillins.
Intramuscular administration of 600,000 units of Bicillin C-R in adults usually produces peak blood levels of 1.0 to 1.3 units per mL within 3 hours; this level falls to an average concentration of 0.32 units per mL at 12 hours, 0.19 units per mL at 24 hours, and 0.03 units per mL at seven days.
Intramuscular administration of 1,200,000 units of Bicillin C-R in adults usually produces peak blood levels of 2.1 to 2.6 units per mL within 3 hours; this level falls to an average concentration of 0.75 units per mL at 12 hours, 0.28 units per mL at 24 hours, and 0.04 units per mL at seven days.
Approximately 60% of penicillin G is bound to serum protein. The drug is distributed throughout the body tissues in widely varying amounts. Highest levels are found in the kidneys with lesser amounts in the liver, skin, and intestines. Penicillin G penetrates into all other tissues and the spinal fluid to a lesser degree. With normal kidney function, the drug is excreted rapidly by tubular excretion. In neonates and young infants and in individuals with impaired kidney function, excretion is considerably delayed.
Microbiology
Mechanism of Action
Penicillin G exerts a bactericidal action against penicillin-susceptible microorganisms during the stage of active multiplication. It acts through the inhibition of biosynthesis of cell-wall peptidoglycan, rendering the cell wall osmotically unstable resulting in death of the bacterium.
Resistance
Penicillin is not active against penicillinase-producing bacteria, or against organisms resistant to beta-lactams because of alterations in the penicillin-binding proteins. Resistance to penicillin G has not been reported in Streptococcus pyogenes .
Penicillin has been shown to be active against most isolates of the following bacteria, both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
Gram-positive bacteria
Beta-hemolytic streptococci (groups A, B, C, G, H, L and M).
Streptococcus pneumoniae (penicillin-susceptible isolates only).
Susceptibility Testing
For specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC .
HOW SUPPLIED
Bicillin C-R (penicillin G benzathine and penicillin G procaine injectable suspension) is supplied in packages of 10 disposable syringes as follows:
2 mL size, containing 1,200,000 units per syringe (21 gauge, thin-wall 1-inch needle for pediatric use), NDC 60793-601-10.
2 mL size, containing 1,200,000 units per syringe (21 gauge, thin-wall 1-1/2-inch needle), NDC 60793-600-10.
Store in a refrigerator, 2º to 8º (36º to 46ºF).
Keep from freezing.
Bicillin Cr - Penicillin G Benzathine And Penicillin G Procaine injection, Suspension PubMed™ news
- Journal Article • 2026 MayIs one all we really need? Reply to Lorenz et al regarding shorter duration of benzathine penicillin G therapy for late or unknown duration syphilis.
- Journal Article • 2026 MayDetection of Antibiotic Residues in Milk and Public Awareness Level Assessment in Selected Districts of the Sidama Region, Ethiopia.
- Journal Article • 2026 MayImplementation of Nursing Protocols for Penicillin-allergy Assessment in an STI Clinic.
- Journal Article • 2026 MayAtypical ocular syphilis presenting as anterior uveitis: clinical response to oral doxycycline in a non-adherent patient.
- Journal Article • 2026 MayTrends of antimicrobial resistance in bloodstream infections at a large traditional Chinese medicine hospital in China: a 10-year surveillance study (2015-2024).