Get your patient on Betadine - Povidone - Iodine solution (Povidone-Iodine)
Betadine - Povidone - Iodine solution prescribing information
INDICATIONS AND USAGE: BETADINE• 5% Sterile Ophthalmic Prep Solution for the eye is indicated for prepping of the periocular region (lids, brow, and cheek) and irrigation of the ocular surface (cornea, conjunctiva, and palpebral fornices).
DOSAGE AND ADMINISTRATION: While the inner surface and contents of the immediate container (i.e. bottle) are sterile, the outer surface of the bottle is not sterile. The use of the bottle in a sterile field should be avoided. BETADINE• 5% Sterile Ophthalmic Prep Solution is used as follows:
BETADINE• 5% Sterile Ophthalmic Prep Solution is used as follows:
- Make sure container is intact before use. To open, COMPLETELY TWIST OFF TAB, do not pull off.
- Gently pour entire contents of bottle into a sterile prep cup. Saturate sterile cotton-tipped applicator to prep lashes and lid margins using one or more applicators per lid; repeat once.
- Saturate sterile prep sponge or other suitable material to prep lids, brow and cheek in a circular ever-expanding fashion until the entire field is covered; repeat prep three (3) times.
- While separating the lids, irrigate the cornea, conjunctiva and palpebral fornices with BETADINE• 5% Sterile Ophthalmic Prep Solution using a sterile bulb syringe.
- After the BETADINE• 5% Sterile Ophthalmic Prep Solution has been left in contact for two minutes, sterile saline solution in a bulb syringe should be used to flush the residual prep solution from the cornea, conjunctiva, and the palpebral fornices.
CONTRAINDICATIONS: Do not use on individuals known to be sensitive to iodine, or other components of this product.
ADVERSE REACTIONS: Local sensitivity has been exhibited by some individuals to povidone-iodine ophthalmic solution.
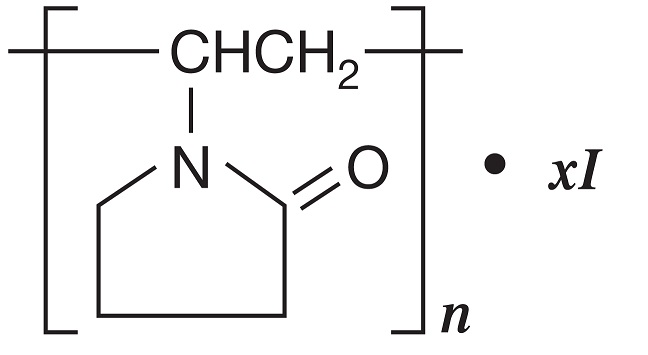
DESCRIPTION: Povidone-Iodine is a broad-spectrum microbicide with the chemical formulas: 2-pyrrolidinone, 1- ethenyl-, homopolymer, compound with iodine; 1-vinyl-2-pyrrolidinone polymer, compound with iodine. The structural formula is as follows:
BETADINE• 5% Sterile Ophthalmic Prep Solution contains 5% povidone-iodine (0.5% available iodine) as a sterile dark brown solution stabilized by glycerin. Inactive Ingredients: purified water, citric acid, glycerin, nonoxynol-9, sodium chloride, sodium hydroxide, and dibasic sodium phosphate.
CLINICAL PHARMACOLOGY: A placebo-controlled study in 38 normal volunteers yielded data for 36 subjects who showed a mean log10 reduction of 3.05 log10 units in total aerobes at 10 minutes following prepping the skin with BETADINE• 5% Sterile Ophthalmic Prep Solution compared with reduction of 1.58 log10 units after prepping with vehicle free of the iodine complex. This placebo-controlled study indicates a mean log10 reduction by the iodine complex compared with the control solution of 1.47 log10 units at 10 minutes and 1.79 log10 units at 45 minutes. The base-line mean aerobic bacterial count was 7,586 organisms/cm2.
HOW SUPPLIED: BETADINE• 5% Sterile Ophthalmic Prep Solution is packaged under sterile conditions, and supplied in 1 fl oz (30 mL) form sealed blue HDPE bottles. Twenty-four (24) bottles are packed in each shipper.
NDC 0065-0411-30 Store at 15°C to 25°C (59°F-77°F). Rx Only Single-use only
@2021 Alcon Inc.
Alcon Manufactured for: Alcon Laboratories, Inc. 6201 South Freeway Fort Worth, TX 76134
Manufactured by: Woodstock Sterile Solutions, Inc. Woodstock, IL 60098
•BETADINE is a registered trademark of Purdue Products L.P.
STW-AC6024-642R01