Baclofen - Baclofen solution prescribing information
INDICATIONS AND USAGE
Baclofen oral solution is indicated for the treatment of spasticity resulting from multiple sclerosis, particularly for the relief of flexor spasms and concomitant pain, clonus, and muscular rigidity.
Baclofen oral solution may also be of some value in patients with spinal cord injuries and other spinal cord diseases.
Limitations of Use Baclofen oral solution is not indicated in the treatment of skeletal muscle spasm resulting from rheumatic disorders.
DOSAGE AND ADMINISTRATION
Recommended Dosage
Initiate baclofen oral solution with a low dosage, preferably in divided doses, administered orally. The following gradually increasing dosage regimen is suggested, but should be adjusted based on clinical response and tolerability:
5 mL (5 mg) three times a day for three days
10 mL (10 mg) three times a day for three days
15 mL (15 mg) three times a day for three days
20 mL (20 mg) three times a day for three days
Additional increases may be necessary up to the maximum recommended dosage of 80 mg daily (20 mg four times a day).
2.2 Discontinuation of Baclofen Oral Solution
When discontinuing baclofen oral solution, reduce the dosage slowly and avoid abrupt withdrawn from the drug to help minimize the risk of adverse reactions [see Warnings and Precautions (5.1 )].
3 DOSAGE FORMS AND STRENGTHS
Oral Solution: 5 mg/5 mL baclofen,USP as a clear, colorless solution with a grape flavor.
USE IN SPECIFIC POPULATIONS
Pregnancy
Risk Summary There are no adequate data on the developmental risk associated with the use of baclofen in pregnant women. Oral administration of baclofen to pregnant rats resulted in an increased incidence of fetal structural abnormalities at a dose which was also associated with maternal toxicity. The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations Fetal/Neonatal adverse reactions Baclofen oral solution may increase the risk of late-onset neonatal withdrawal symptoms [see Warnings and Precautions (5.2 )] .
Data
Animal Data Baclofen given orally has been shown to increase the incidence of omphaloceles (ventral hernias) in fetuses of rats given approximately 13 times on a mg/kg basis, or 3 times on a mg/m 2 basis, the maximum oral dose recommended for human use; this dose also caused reductions in food intake and weight gain in the dams. This abnormality was not seen in mice or rabbits.
Lactation
Risk Summary At recommended oral doses, baclofen is present in human milk. There are no human data on the effects of baclofen on milk production. There are no adequate data on the effects of baclofen on the breastfed infant. Withdrawal symptoms can occur in breastfed infants when maternal administration of baclofen is stopped, or when breastfeeding is stopped [see Warnings and Precautions (5.2 )] .
The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for baclofen and any potential adverse effects on the breastfed infant from baclofen or from the underlying maternal condition.
Pediatric Use
Safety and effectiveness in pediatric patients below the age of 12 have not been established.
Geriatric Use
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Renal Impairment
Because baclofen is primarily excreted unchanged through the kidneys, baclofen should be given with caution to patients with renal impairment, and it may be necessary to reduce the dosage.
4 CONTRAINDICTIONS
Baclofen oral solution is contraindicated in patients with hypersensitivity to baclofen.
WARNINGS AND PRECAUTIONS
Adverse Reactions from Abrupt Withdrawal of Baclofen oral solution
Abrupt discontinuation of baclofen, regardless of the cause, has resulted in adverse reactions that include hallucinations, seizures, high fever, altered mental status, exaggerated rebound spasticity, and muscle rigidity, that in rare cases has advanced to rhabdomyolysis, multiple organ-system failure, and death. Therefore, reduce the dosage slowly when baclofen oral solution is discontinued, unless the clinical situation justifies a rapid withdrawal.
Neonatal Withdrawal Symptoms
Withdrawal symptoms in neonates whose mothers were treated with oral baclofen throughout pregnancy have been reported starting hours to days after delivery. The symptoms of withdrawal in these infants have included increased muscle tone, tremor, jitteriness, and seizure. If the potential benefit justifies the potential risk to the fetus and baclofen is continued during pregnancy, gradually reduce the dosage and discontinue baclofen before delivery. If slow withdrawal is not feasible, advise the parents or caregivers of the exposed neonate of the potential for neonatal withdrawal.
Drowsiness and Sedation
Drowsiness and sedation have been reported in up to 63% of patients taking baclofen, the active ingredient in baclofen oral solution [see Adverse Reactions (6.1 )] . Patients should avoid operation of automobiles or other dangerous machinery and activities made hazardous by decreased alertness when starting baclofen or increasing the dose until they know how the drug affects them. Advise patients that the central nervous system depressant effects of baclofen may be additive to those of alcohol and other CNS depressants.
Poor Tolerability in Stroke Patients
Baclofen should be used with caution in patients who have had a stroke. Baclofen has not significantly benefited patients with stroke. These patients have also shown poor tolerability to the drug.
Exacerbation of Psychotic Disorders, Schizophrenia, or Confusional States
Baclofen should be used with caution in patients suffering from psychotic disorders, schizophrenia, or confusional states. If treated with baclofen, these patients should be kept under careful surveillance because exacerbations of these conditions have been observed with oral baclofen administration.
Exacerbation of Autonomic Dysreflexia
Baclofen should be used with caution in patients with a history of autonomic dysreflexia. The presence of nociceptive stimuli or abrupt withdrawal of baclofen may cause an autonomic dysreflexic episode.
Exacerbation of Epilepsy
Baclofen should be used with caution in patients with epilepsy. Deterioration in seizure control has been reported in patients taking baclofen.
Posture and Balance Effects
Baclofen should be used with caution in patients where spasticity is utilized to sustain upright posture and balance in locomotion or whenever spasticity is utilized to obtain increased function.
Ovarian Cysts
A dose-related increase in incidence of ovarian cysts was observed in female rats treated chronically with oral baclofen. Ovarian cysts have been found by palpation in about 4% of the multiple sclerosis patients who were treated with oral baclofen for up to one year. In most cases, these cysts disappeared spontaneously while patients continued to receive the drug. Ovarian cysts are estimated to occur spontaneously in approximately 1% to 5% of the normal female population.
ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Adverse Reactions from Abrupt Withdrawal of Baclofen [see Warnings and Precautions (5.1 )]
- Neonatal Withdrawal Symptoms [see Warnings and Precautions (5.2 )]
- Drowsiness and Sedation [see Warnings and Precautions (5.3 )]
- Poor Tolerability in Stroke Patients [see Warnings and Precautions (5.4 )]
- Exacerbation of Psychotic Disorders, Schizophrenia, or Confusional States [see Warnings and Precautions (5.5 )]
- Exacerbation of Autonomic Dysreflexia [see Warnings and Precautions (5.6 )]
- Exacerbation of Epilepsy [see Warnings and Precautions (5.7 )]
- Posture and Balance Effects [see Warnings and Precautions (5.8 )]
- Ovarian Cysts [see Warnings and Precautions (5.9 )]
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reaction is transient drowsiness. In one controlled study of 175 patients, transient drowsiness was observed in 63% of those receiving baclofen compared to 36% of those in the placebo group. Other common adverse reactions (up to 15%) are dizziness and weakness. Adverse reactions with a frequency of ≥1% are listed in Table 1.
ADVERSE REACTION | PERCENT |
Drowsiness | 10-63% |
Dizziness | 5-15% |
Weakness | 5-15% |
Nausea | 4-12% |
Confusion | 1-11% |
Hypotension | 0-9% |
Headache | 4-8% |
Insomnia | 2-7% |
Constipation | 2-6% |
Urinary Frequency | 2-6% |
Fatigue | 2-4% |
The following adverse reactions not included in Table 1, classified by body system, were also reported:
Neuropsychiatric: euphoria, excitement, depression, hallucinations, paresthesia, muscle pain, tinnitus, slurred speech, coordination disorder, tremor, rigidity, dystonia, ataxia, blurred vision, nystagmus, strabismus, miosis, mydriasis, diplopia, dysarthria, epileptic seizure
Cardiovascular: dyspnea, palpitation, chest pain, syncope
Gastrointestinal: dry mouth, anorexia, taste disorder, abdominal pain, vomiting, diarrhea, and positive test for occult blood in stool
Genitourinary: enuresis, urinary retention, dysuria, impotence, inability to ejaculate, nocturia, hematuria
Other: rash, pruritus, ankle edema, excessive perspiration, weight gain, nasal congestion
The following laboratory tests have been found to be abnormal in patients receiving baclofen: increased SGOT, elevated alkaline phosphatase, and elevation of blood sugar.
DRUG INTERACTIONS
CNS Depressants and Alcohol
Baclofen can cause CNS depression, including drowsiness and sedation, which may be additive when used concomitantly with other CNS depressants or alcohol [see Warnings and Precautions (5.3 )].
DESCRIPTION
Baclofen is a gamma-aminobutyric acid (GABA-ergic) agonist available as 5 mg/5 mL solution for oral administration. Its chemical name is 4-amino-3-(4-chlorophenyl)-butanoic acid, and its structural formula is:
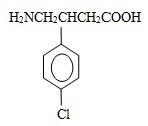
Molecular formula is C 10 H 12 CINO 2 .
Molecular Weight is 213.66.
Baclofen, USP is a white to off-white, odorless or practically odorless crystalline powder. It is slightly soluble in water, very slightly soluble in methanol, and insoluble in chloroform.
The inactive ingredients of baclofen oral solution are: carboxymethyl cellulose sodium, glycerin, grape flavor NAT & ART, methylparaben, propylparaben, purified water, and sucralose. May also contain sodium hydroxide or hydrochloric acid for pH adjustment.
CLINICAL PHARMACOLOGY
Mechanism of Action
The precise mechanism of action of baclofen is not fully understood. Baclofen inhibits both monosynaptic and polysynaptic reflexes at the spinal level, possibly by decreasing excitatory neurotransmitter release from afferent terminals, although actions at supraspinal sites may also occur and contribute to its clinical effect. Baclofen is a structural analog of the inhibitory neurotransmitter gamma- aminobutyric acid (GABA), and may exert its effects by stimulation of the GABA B receptor subtype.
Pharmacodynamics
Pharmacokinetics
A pharmacokinetic study in heathy adult male subjects under fasting conditions at 20 mg dose level demonstrated similar bioavailability for baclofen oral solution and oral tablets. The peak plasma concentrations were achieved in about 0.75 hours from oral solution and the apparent elimination half-life is about 5.7 hours. Baclofen is excreted primarily by the kidney in unchanged form, and there is relatively large intersubject variation in absorption and/or elimination.
NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No increase in tumors was seen in rats receiving baclofen orally for two years at approximately 30 to 60 times on a mg/kg basis, or 10 to 20 times on a mg/m 2 basis, the maximum oral dose recommended for human use.
Mutagenesis
Genetic toxicology assays have not been conducted for baclofen.
Impairment of Fertility
Studies to evaluate the effects of baclofen on fertility have not been conducted.
CLINICAL STUDIES
The efficacy of baclofen is based upon a bioavailability study in healthy adults comparing baclofen oral tablets to baclofen oral solution [see Clinical Pharmacology (12.3 )].
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Baclofen Oral Solution contains 5 mg/5 mL baclofen, USP. It is a clear, colorless solution with a grape flavor and is supplied in bottles of 473 mL, NDC 72888-103-24.
Storage and Handling
Store at 20º to 25ºC (68º to 77ºF); excursions permitted to 15° to 30°C (59° to 86°F) [See USP Controlled Room Temperature]. Product can also be stored at 2°C to 8°C (36°F to 46°F).
Dispense in a tight, light-resistant container with a child-resistant closure.