Get your patient on Baclofen - Baclofen tablet (Baclofen)
Baclofen - Baclofen tablet prescribing information
INDICATIONS AND USAGE
Baclofen tablets are useful for the alleviation of signs and symptoms of spasticity resulting from multiple sclerosis, particularly for the relief of flexor spasms and concomitant pain, clonus, and muscular rigidity.
Patients should have reversible spasticity so that baclofen treatment will aid in restoring residual function. Baclofen tablets may also be of some value in patients with spinal cord injuries and other spinal cord diseases.
Baclofen tablets are not indicated in the treatment of skeletal muscle spasm resulting from rheumatic disorders.
The efficacy of baclofen in stroke, cerebral palsy, and Parkinson’s disease has not been established and, therefore, it is not recommended for these conditions.
DOSAGE AND ADMINISTRATION
The determination of optimal dosage requires individual titration. Start therapy at a low dosage and increase gradually until optimum effect is achieved (usually between 40 to 80 mg daily).
The following dosage titration schedule is suggested:
5 mg t.i.d. for 3 days
10 mg t.i.d. for 3 days
15 mg t.i.d. for 3 days
20 mg t.i.d. for 3 days
Thereafter additional increases may be necessary but the total daily dose should not exceed a maximum of 80 mg daily (20 mg q.i.d.).
The lowest dose compatible with an optimal response is recommended. If benefits are not evident after a reasonable trial period, patients should be slowly withdrawn from the drug (see WARNINGS , Abrupt Drug Withdrawal).
CONTRAINDICATIONS
Hypersensitivity to baclofen.
ADVERSE REACTIONS
The most common is transient drowsiness (10 to 63%). In one controlled study of 175 patients, transient drowsiness was observed in 63% of those receiving baclofen compared to 36% of those in the placebo group. Other common adverse reactions are dizziness (5 to 15%), weakness (5 to 15%) and fatigue (2 to 4%).
Others reported:
Neuropsychiatric : Confusion (1 to 11%), headache (4 to 8%), insomnia (2 to 7%); and, rarely, euphoria, excitement, depression, hallucinations, paresthesia, muscle pain, tinnitus, slurred speech, coordination disorder, tremor, rigidity, dystonia, ataxia, blurred vision, nystagmus, strabismus, miosis, mydriasis, diplopia, dysarthria, epileptic seizure.
Cardiovascular : Hypotension (0 to 9%). Rare instances of dyspnea, palpitation, chest pain, syncope.
Gastrointestinal : Nausea (4 to 12%), constipation (2 to 6%); and rarely, dry mouth, anorexia, taste disorder, abdominal pain, vomiting, diarrhea, and positive test for occult blood in stool.
Genitourinary : Urinary frequency (2 to 6%); and rarely, enuresis, urinary retention, dysuria, impotence, inability to ejaculate, nocturia, hematuria.
Other : Instances of rash, pruritus, ankle edema, excessive perspiration, weight gain, nasal congestion.
Some of the CNS and genitourinary symptoms may be related to the underlying disease rather than to drug therapy. The following laboratory tests have been found to be abnormal in a few patients receiving baclofen: increased SGOT, elevated alkaline phosphatase, and elevation of blood sugar.
To report SUSPECTED ADVERSE REACTIONS, contact Amneal Pharmaceuticals at 1-877-835-5472 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DESCRIPTION
Baclofen, USP is a muscle relaxant and antispastic.
Its chemical name is 4-amino-3-(4-chlorophenyl)-butanoic acid. The structural formula is:
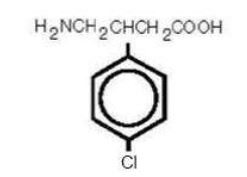
C 10 H 12 ClNO 2 M.W. 213.66
Baclofen, USP is a white to off-white, odorless, or practically odorless crystalline powder.
It is slightly soluble in water, very slightly soluble in methanol and insoluble in chloroform.
Each tablet, for oral administration, contains 5 mg, 10 mg or 20 mg baclofen, USP. In addition, each tablet contains the following inactive ingredients: colloidal silicon dioxide, dibasic calcium phosphate dihydrate, magnesium stearate, microcrystalline cellulose, sodium starch glycolate.
CLINICAL PHARMACOLOGY
The precise mechanism of action of baclofen is not fully known. Baclofen is capable of inhibiting both monosynaptic and polysynaptic reflexes at the spinal level, possibly by hyperpolarization of afferent terminals, although actions at supraspinal sites may also occur and contribute to its clinical effect. Although baclofen is an analog of the putative inhibitory neurotransmitter gamma-aminobutyric acid (GABA), there is no conclusive evidence that actions on GABA systems are involved in the production of its clinical effects. In studies with animals baclofen has been shown to have general CNS depressant properties as indicated by the production of sedation with tolerance, somnolence, ataxia, and respiratory and cardiovascular depression. Baclofen is rapidly and extensively absorbed and eliminated. Absorption may be dose dependent, being reduced with increasing doses. Baclofen is excreted primarily by the kidney in unchanged form and there is relatively large intersubject variation in absorption and/or elimination.
HOW SUPPLIED
Baclofen Tablets USP, 5 mg are available as a white, oval shaped, uncoated tablets, debossed with “C19” on one side and plain on other side.
Bottle of 100: NDC 0115-1011-01
Baclofen Tablets USP, 10 mg are available as an off-white, to beige, round, flat-faced tablet with a beveled edge debossed with a “C21” on top of bisect on one side and plain on the other side.
Bottles of 30: NDC 0115-1010-08
Bottles of 100: NDC 0115-1010-01
Bottles of 500: NDC 0115-1010-02
Bottles of 1000: NDC 0115-1010-03
Baclofen Tablets USP, 20 mg are available as an off-white, to beige, round, flat-faced tablet with a beveled edge debossed with a “C22” on top of bisect on one side and plain on the other side.
Bottles of 30: NDC 0115-1012-08
Bottles of 100: NDC 0115-1012-01
Bottles of 500: NDC 0115-1012-02
Bottles of 1000: NDC 0115-1012-03
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Dispense in a tightly-closed, light-resistant container as defined in the USP using a child-resistant closure.
Keep this and all medications out of the reach of children.
Manufactured by: Amneal Pharmaceuticals Pvt. Ltd. Oral Solid Dosage Unit Ahmedabad 382213, INDIA
Distributed by: Amneal Pharmaceuticals LLC Bridgewater, NJ 08807
Rev.04-2021-02