Get your patient on Amyvid - Florbetapir F 18 injection, Solution (Florbetapir F 18)
Amyvid - Florbetapir F 18 injection, Solution prescribing information
INDICATIONS AND USAGE
AMYVID is indicated for positron emission tomography (PET) of the brain to estimate amyloid beta neuritic plaque density in adults with cognitive impairment for:
- Evaluation of Alzheimer's disease (AD) and other causes of cognitive decline
- Selection of patients who are indicated for amyloid beta-directed therapy as described in the prescribing information of the therapeutic products
DOSAGE AND ADMINISTRATION
- The recommended amount of radioactivity is 370 MBq (10 mCi) administered as a single intravenous bolus in a total volume of up to 10 mL. (2.2 )
- Follow the injection with an intravenous flush of approximately 10 mL of 0.9% sodium chloride injection. (2.2 )
- Obtain 10-minute PET images starting approximately 30 minutes to 50 minutes after drug administration. (2.3 )
- See full prescribing information for image interpretation and radiation dosimetry. (2.4 , 2.5 )
Radiation Safety - Drug Handling
Handle AMYVID with appropriate safety measures to minimize radiation exposure during administration [see Warnings and Precautions (5.2 )] . Use waterproof gloves and effective radiation shielding, including syringe shields when handling and administering AMYVID.
Radiopharmaceuticals, including AMYVID, should be used by or under the control of healthcare providers who are qualified by specific training and experience in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radionuclides.
Recommended Dosage and Administration Instructions
Recommended Dosage
The recommended amount of radioactivity of AMYVID is 370 MBq (10 mCi) in a total volume of up to 10 mL, administered as a single intravenous bolus. The maximum mass dose is 50 mcg. Follow the injection with an intravenous flush of approximately 10 mL of 0.9% sodium chloride injection.
Patient Preparation
Instruct patients to hydrate before and after AMYVID administration and to void following AMYVID administration before imaging and frequently thereafter [see Warnings and Precautions (5.2 )] .
Administration
- Use aseptic technique and radiation shielding when withdrawing and administering AMYVID.
- Visually inspect AMYVID for particulate matter and discoloration prior to administration. Do not use AMYVID if it contains particulate matter or if it is discolored.
- Do not dilute AMYVID.
- Assay the dose in a dose calibrator prior to administration.
- Inject AMYVID through a short intravenous catheter (approximately 1.5 inches or less) to minimize the potential for adsorption of the drug to the catheter. Portions of the AMYVID dose may adhere to longer catheters.
- Dispose of unused product in a safe manner in compliance with applicable regulations.
Image Acquisition Instructions
- Position the patient supine with the head positioned to center the brain, including the cerebellum, in the PET scanner field of view. Tape or other flexible head restraints may be employed to reduce head movement.
- Acquire 10-minute PET images starting 30 minutes to 50 minutes after AMYVID administration.
- Image reconstruction should include attenuation correction with resulting transaxial pixel sizes between 2 mm and 3 mm.
Image Display and Interpretation
Image Display
- Display images in the transaxial orientation with access as needed to the sagittal and coronal planes.
- In reviewing the images, include all transaxial slices of the brain using a black-white scale set to the maximum intensity of all the brain pixels.
- Initially locate the brain slice with the highest levels of image contrast (highest signal intensity) and adjust the contrast appropriately.
- Start image interpretation by displaying slices sequentially from the bottom of the brain to the top. Periodically refer to the sagittal and coronal plane image display as needed to better define the signal intensity and to ensure that the entire brain is displayed.
Visual Assessment
AMYVID images should be interpreted only by readers who successfully complete the training program provided by the manufacturer.
Perform image interpretation independently of the patient's clinical features, relying on the recognition of unique image features.
Interpret AMYVID images based upon the distribution of signal intensity within the cerebral cortex by comparing the signal intensity in the cortical gray matter and the adjacent white matter. The signal intensity in the cerebellum does not contribute to the scan interpretation. For example, a positive scan may show retained cerebellar gray-white contrast even when the cortical gray-white contrast is lost.
Some scans may be difficult to interpret due to image noise, atrophy with a thinned cortical ribbon, or image blur. For cases where there is uncertainty as to the location of cortical signal, use co-registered anatomical imaging to improve localization of signal [see Warnings and Precautions (5.1 )] .
Negative AMYVID Scan
Negative scans show more signal in white matter than in adjacent cortical gray matter, creating clear gray-white contrast.
A negative scan indicates sparse to no amyloid beta neuritic plaques. In patients being evaluated for AD and other causes of cognitive decline who have not been treated with amyloid beta-directed therapy, a negative scan is inconsistent with a neuropathological diagnosis of AD at the time of image acquisition and reduces the likelihood that a patient's cognitive impairment is due to AD. A negative scan result does not preclude the accumulation of amyloid beta in the brain in the future.
Positive AMYVID Scan
Positive scans show cortical areas with reduction or loss of the normally distinct gray-white contrast. These scans will have one or more areas with increased cortical gray matter signal which results in reduced (or absent) gray-white contrast. Specifically, a positive scan will have either:
- Two or more brain areas (each larger than a single cortical gyrus) in which there is reduced or absent gray-white contrast. This is the most common appearance of a positive scan. or
- One or more areas in which cortical gray matter signal is intense and clearly exceeds the signal in adjacent white matter.
A positive scan establishes the presence of moderate to frequent amyloid beta neuritic plaques. Neuropathological examination has shown that moderate to frequent amyloid beta neuritic plaques are present in patients with AD, but may also be present in patients with other types of neurologic conditions as well as older people with normal cognition.
Figures 1, 2, and 3 provide examples of negative and positive scans.
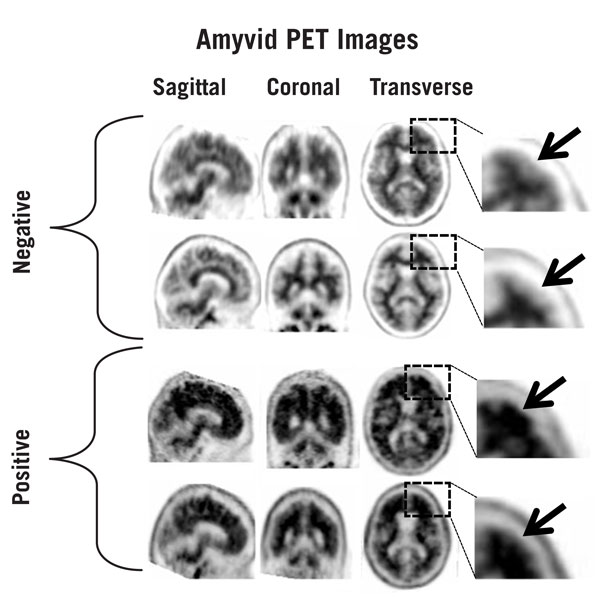
Figure 1: Examples of AMYVID negative scans (top two rows) and positive scans (bottom two rows). Left to right panels show sagittal, coronal, and transverse PET image slices. Final panel to right shows an enlarged picture of the brain area under the box. The top two arrows are pointing to normal preserved gray-white contrast with cortical signal intensity less than the adjacent white matter. The bottom two arrows indicate areas of decreased gray-white contrast with increased cortical signal intensity that is comparable to the intensity in the adjacent white matter.
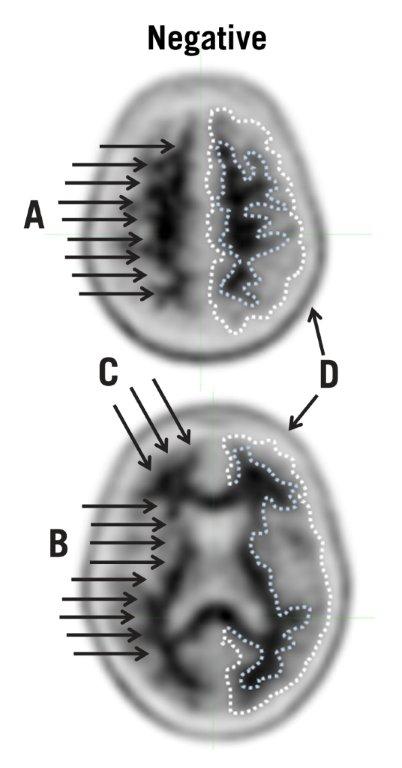 | Figure 2: Typical Negative Scan. The upper (top) and lower (bottom) transverse slices both show good gray-white matter contrast. The dotted lines illustrate the edge of the cortical gray matter (outer line) and the gray-white border (inner line) and highlight the contrast between less intense signal in the gray matter and more intense signal in the white matter. The arrows illustrate the following points: A. White matter tracts can be delineated from the frontal lobe to parietal lobe. B. White matter tracts are clearly identified throughout the occipital / temporal area. C. Scalloped appearance is seen with “fingers” of white matter in the frontal cortex. D. Low levels of signal in scalp or skull are distinguished from gray matter signal by the shape and position. |
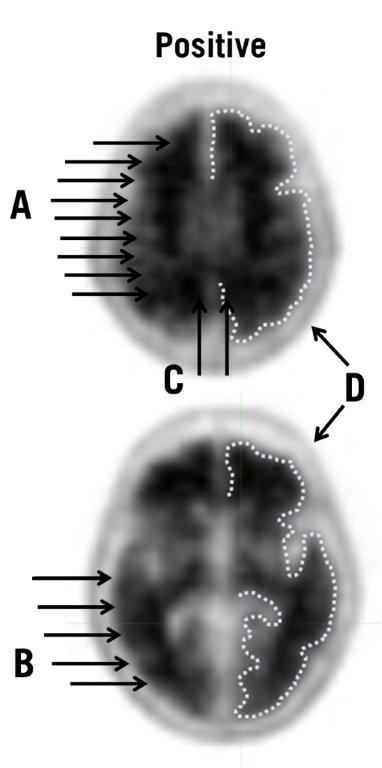 | Figure 3: Typical Positive Scan: The upper (top) and lower (bottom) transverse slices show loss of gray-white matter contrast in multiple brain regions. The dotted lines illustrate the edge of the cortical gray matter. Compared to the images from the negative case in Figure 2, the gray matter signal is similar to white matter signal and the gray-white matter border is more difficult to discern. The arrows show the following points: A. White matter tracts are difficult to fully identify as they travel from frontal to parietal lobe. B. Borders of white matter tracts in occipital / temporal area are lost in places. C. Gray matter in medial parietal cortex (precuneus) has increased signal. D. Low levels of signal in scalp or skull are distinguished from gray matter signal by the shape and position. |
Quantitative Analysis
Quantification of amyloid beta neuritic plaque levels (e.g., Centiloid scale or standardized uptake value ratio (SUVR)) can be used in conjunction with visual assessment and performed with FDA-authorized software indicated for quantification of brain amyloid beta PET scans. Refer to the drug manufacturer's training materials for qualitative and quantitative assessment and software manufacturers' documentation for software operation.
Radiation Dosimetry
Estimated radiation absorbed doses for adults from intravenous injection of AMYVID are shown in Table 1 .
| ORGAN/TISSUE | MEAN ABSORBED DOSE PER UNIT ADMINISTERED ACTIVITY (microGy/MBq) |
| Adrenal | 14 |
| Brain | 10 |
| Breasts | 6 |
| Gallbladder Wall | 143 |
| Heart Wall | 13 |
| Kidneys | 14 |
| Liver | 64 |
| Lower Large Intestine Wall | 28 |
| Lungs | 9 |
| Muscle | 9 |
| Osteogenic Cells | 28 |
| Ovaries | 18 |
| Pancreas | 14 |
| Red Marrow | 14 |
| Skin | 6 |
| Small Intestine | 66 |
| Spleen | 9 |
| Stomach Wall | 12 |
| Testes | 7 |
| Thymus | 7 |
| Thyroid | 7 |
| Upper Large Intestine Wall | 74 |
| Urinary Bladder Wall | 27 |
| Uterus | 16 |
| Total Body | 12 |
| Effective Dose (microSv/MBq) | 19 |
The whole-body effective dose resulting from administration of 370 MBq (10 mCi) of AMYVID to an adult is estimated to be 7 mSv. When PET/CT is performed, exposure to radiation will increase by an amount dependent on the settings used in the CT acquisition.
DOSAGE FORMS AND STRENGTHS
Injection: 500 MBq/mL to 1,900 MBq/mL (13.5 mCi/mL to 51 mCi/mL) of florbetapir F 18 in up to 100 mL volume at end of synthesis (EOS) as a clear, colorless solution in a multiple-dose vial.
USE IN SPECIFIC POPULATIONS
Lactation: Temporarily discontinue breastfeeding. A lactating woman should pump and discard breast milk for 24 hours after AMYVID administration. (8.2 )
Pregnancy
Risk Summary
There are no available data on AMYVID use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes. Animal reproduction studies have not been conducted with florbetapir F 18 to evaluate its effect on female reproduction and embryo-fetal development.
All radiopharmaceuticals, including AMYVID, have the potential to cause fetal harm depending on the stage of fetal development and the magnitude of the radiation dose. If considering AMYVID administration to a pregnant woman, inform the patient about the potential for adverse pregnancy outcomes based on the radiation dose from the drug and the gestational timing of exposure.
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Lactation
Risk Summary
There are no data on the presence of florbetapir F 18 in human milk, the effects on the breastfed infant, or effects on milk production. Lactation studies have not been conducted in animals. Exposure of AMYVID to a breastfed infant can be minimized by temporary discontinuation of breastfeeding ( see Clinical Considerations ). The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for AMYVID and any potential adverse effects on the breastfed child from AMYVID or from the underlying maternal condition.
Clinical Considerations
To decrease radiation exposure to the breastfed infant, advise a lactating woman to pump and discard breast milk for 24 hours (>10 half-lives of radioactive decay for the F 18 isotope) following administration of AMYVID.
Pediatric Use
The safety and effectiveness of AMYVID have not been established in pediatric patients.
Geriatric Use
Of the 496 patients in clinical studies of AMYVID, 307 patients were 65 years of age and older, while 203 patients were 75 years of age and older. No overall differences in safety or effectiveness were observed between patients 65 years of age and older and younger adult patients.
CONTRAINDICATIONS
None.
WARNINGS AND PRECAUTIONS
- Risk of Image Misinterpretation and Other Errors: Image interpretation errors have been observed. (5.1 )
- Radiation Risk: AMYVID contributes to a patient's long-term cumulative radiation exposure. Ensure safe drug handling to protect patients and health care providers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration. (2.1 , 2.2 , 5.2 )
Risk of Image Misinterpretation and Other Errors
Errors may occur in the estimation of brain amyloid beta neuritic plaque density during AMYVID image interpretation [see Clinical Studies (14 )].
The use of clinical information in the interpretation of AMYVID images has not been evaluated and may lead to an inaccurate assessment. Extensive brain atrophy as well as motion artifacts that distort the image may limit the ability to distinguish gray and white matter on an AMYVID scan.
Perform image interpretation independently of the patient's clinical information. For cases where there is uncertainty as to the location of cortical signal, use co-registered anatomical imaging to improve localization of signal [see Dosage and Administration (2.4 )].
Radiation Risk
AMYVID contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk of cancer. Ensure safe drug handling to protect patients and health care providers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration [see Dosage and Administration (2.1 , 2.2 )] .
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥ 0.4%) were headache, musculoskeletal pain, increased blood pressure, nausea, fatigue, injection site reaction, anxiety, back pain, claustrophobia, dizziness, feeling cold, insomnia, and neck pain. (6.1 )
To report SUSPECTED ADVERSE REACTIONS, contact Eli Lilly and Company at 1-800-545-5979 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of AMYVID was evaluated in 555 adult subjects who received AMYVID by intravenous injection in clinical trials. Table 2 shows adverse reactions reported in ≥ 0.4% of subjects from the clinical trials.
a Includes the terms blood pressure increased and hypertension. | |
b Includes the terms injection site hemorrhage, injection site irritation, and injection site pain. | |
c Includes the terms feeling cold and chills. | |
| Adverse Reaction | AMYVID N=555 % |
| Headache | 1.8 |
| Musculoskeletal pain | 0.7 |
| Blood pressure increased a | 0.7 |
| Nausea | 0.7 |
| Fatigue | 0.5 |
| Injection site reaction b | 0.5 |
| Anxiety | 0.4 |
| Back pain | 0.4 |
| Claustrophobia | 0.4 |
| Dizziness | 0.4 |
| Feeling cold c | 0.4 |
| Insomnia | 0.4 |
| Neck pain | 0.4 |
Adverse reactions that occurred in <0.4% of subjects included infusion site rash, dysgeusia, pruritus, urticaria, and flushing.
DESCRIPTION
Drug Characteristics
AMYVID (florbetapir F 18 injection) is a radioactive diagnostic drug for intravenous use.
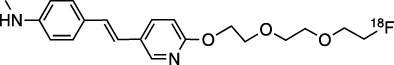
Chemically, florbetapir F 18 is (E)-4-(2-(6-(2-(2-(2[ 18 F] fluoroethoxy)ethoxy)ethoxy)pyridine-3-yl)vinyl)-N-methylbenzamine. The molecular weight is 359 and the structural formula is:
AMYVID is a sterile, non-pyrogenic clear, colorless solution. Each mL contains 0.1 mcg to 19 mcg of florbetapir and 500 MBq to 1,900 MBq (13.5 mCi to 51 mCi) of florbetapir F 18 at EOS and the following inactive ingredients: 4.5 mg sodium ascorbate and 0.1 mL dehydrated alcohol in 0.9% sodium chloride injection. The pH of the solution is between 5.5 and 8.0.
Nuclear Physical Characteristics
Fluorine-18 (F 18) decays by positron (β + ) emission to oxygen-18 and has a physical half-life of 109.8 minutes. The principal photons useful for diagnostic imaging are the coincident pair of 511 keV gamma photons, resulting from the interaction of the emitted positron with an electron (Table 3 ).
| Radiation | Energy Level (keV) | Abundance (%) |
| Positron | 249.8 | 96.9 |
| Gamma | 511 | 193.5 |
The point source air-kerma coefficient for F 18 is 3.74E -17 Gy m 2 /(Bq s); this coefficient was formerly defined as the specific gamma-ray constant of 5.7 R/hr/mCi at 1 cm. The first half-value thickness of lead (Pb) for F 18 gamma rays is approximately 6 mm. The relative reduction of radiation emitted by F 18 that results from various thicknesses of lead shielding is shown in Table 4 . The use of ~8 cm of Pb will decrease the radiation transmission (i.e., exposure) by a factor of about 10,000.
| Shield Thickness cm of Lead (Pb) | Coefficient of Attenuation |
| 0.6 | 0.5 |
| 2 | 0.1 |
| 4 | 0.01 |
| 6 | 0.001 |
| 8 | 0.0001 |
For use in correcting for physical decay of this radionuclide, the fractions remaining at selected intervals after calibration are shown in Table 5 .
| Minutes | Fraction Remaining |
| 0 | 1.00 |
| 15 | 0.909 |
| 30 | 0.826 |
| 60 | 0.683 |
| 110 | 0.500 |
| 220 | 0.250 |
| 440 | 0.060 |
CLINICAL PHARMACOLOGY
Mechanism of Action
Florbetapir F 18 binds to amyloid beta plaques and the F 18 isotope produces a positron signal that is detected by a PET scanner. In in vitro binding studies using postmortem human brain homogenates containing amyloid beta plaques, the dissociation constant (Kd) for florbetapir was 3.7 ± 0.3 nM. The binding of florbetapir F 18 to amyloid beta aggregates was demonstrated in postmortem human brain sections using autoradiographic methods, thioflavin S and traditional silver staining correlation studies as well as immunohistochemistry (monoclonal antibody to amyloid beta) correlation studies. Florbetapir binding to tau protein and a battery of neuroreceptors was not detected in in vitro studies.
Pharmacodynamics
Following intravenous injection, florbetapir F 18 diffuses across the human blood-brain barrier and produces a signal intensity detectable throughout the brain. Subsequently, cerebral perfusion decreases the brain florbetapir F 18 content, with differential retention of the drug in areas that contain amyloid beta aggregates compared to areas that lack the aggregates. The time-activity curves for florbetapir F 18 in the brain of subjects with positive scans show continual signal increases from time zero through 30 minutes post-administration, with stable values thereafter up to at least 90 minutes post-injection. Differences in signal intensity between brain regions showing specific and non-specific florbetapir F 18 uptake form the basis for the image interpretation method [see Dosage and Administration (2.4 )] .
The test-retest distribution of florbetapir F 18 was evaluated in 21 subjects (11 with probable AD and 10 healthy subjects) who underwent two administrations of florbetapir F 18 (followed by PET scans) separated by a time period of 2 to 30 days. Images were shown to maintain signal distribution reproducibility when evaluated qualitatively (by a reader blinded to image time points) as well as quantitatively using an automated assessment of standardized uptake value ratio (SUVR) in pre-specified brain regions. A comparison of a 10-minute versus a 20-minute image acquisition time showed no difference in the mean cortical to cerebellar SUVR results obtained.
Pharmacokinetics
Following intravenous administration of 370 MBq (10 mCi) of AMYVID in healthy subjects, florbetapir F 18 was distributed throughout the body with less than 5% of the injected F 18 radioactivity present in the blood by 20 minutes following administration, and less than 2% present by 45 minutes after administration. The F 18 in circulation during the 30-minute to 90-minute imaging window was principally in the form of polar florbetapir metabolites. Whole body scanning following the intravenous injection showed accumulation of radioactivity in the liver within 4 minutes post-injection, followed by elimination of the radioactivity primarily through the biliary/gastrointestinal tract. Some radioactivity is detected in the bladder. Essentially all radioactivity collected in the urine was present as polar metabolites of florbetapir F 18.
Drug Interaction Studies
Clinical
In patients with probable AD, mean cortical SUVR did not differ between patients taking or not taking donepezil, galantamine, or memantine.
In vitro
Effect of acetylcholinesterase inhibitors: donepezil, galantamine, and tacrine did not alter florbetapir F 18 binding to its target.
Effect of amyloid beta-directed therapy: donanemab and lecanemab did not alter florbetapir F 18 binding to its target.
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies to assess the carcinogenicity or reproductive toxicity potential of florbetapir F 18 have not been conducted.
Mutagenesis
In an in vitro bacterial reverse mutation assay (Ames test), increases in the number of revertant colonies were observed in 2 of the 5 strains exposed to 19 F-AV-45, the non-radioactive form of florbetapir F 18. In a chromosomal aberration in vitro study with cultured human peripheral lymphocytes, 19 F-AV-45 did not increase the percentage of cells with structural aberrations with 3-hour exposure with or without activation; however, 22-hour exposure produced a statistically significant increase in structural aberrations at all tested concentrations. Potential in vivo genotoxicity of 19 F-AV-45 was evaluated in a rat micronucleus study. In this assay, 19 F-AV-45 did not increase the number of micronucleated polychromatic erythrocytes at the highest achievable dose level, 372 mcg/kg/day, when given twice daily for 3 consecutive days.
CLINICAL STUDIES
Evaluation of AD and Other Causes of Cognitive Decline
The effectiveness of AMYVID was evaluated in two single-arm clinical studies (i.e., Studies 1 and 2) in subjects with a range of cognitive function, including some terminally ill subjects who had agreed to participate in a postmortem brain donation program as well as healthy subjects. Subjects underwent an AMYVID injection and scan. The images were interpreted using a clinically applicable binary image interpretation method (negative or positive) by five independent readers who were blinded to all clinical information [see Dosage and Administration (2.3 , 2.4 )] . Image interpretations used co-registration with CT scans when PET scans were performed on dual PET-CT scanners. Before image interpretation, all readers underwent special training on image interpretation: in-person training or electronic media training.
The neuritic plaque density in both studies was determined using an algorithm in which microscopic measures of highest plaque density within a brain region were averaged to produce an estimate of global neuritic plaque density. The global neuritic plaque density was categorized in the same manner as that for a region (Table 6 ), where plaques were counted on slides with modified Bielschowsky silver stained tissue sections. To determine the agreement between the in vivo AMYVID image results and the postmortem amyloid beta neuritic plaque density, AMYVID results (negative or positive) were pre-specified to correspond to specific histopathology-derived plaque density scores, based upon a modification of the Consortium to Establish a Registry for Alzheimer's Disease (CERAD) criteria, which use neuritic plaque counts as a necessary pathological feature of AD.
| Histopathology Categorization | AMYVID PET Read | |
| Neuritic Plaque Counts | CERAD Score | |
| <1 | none | Negative |
| 1 - 5 | sparse | |
| 6 - 19 | moderate | Positive |
| 20+ | frequent | |
Study 1 evaluated performance characteristics (sensitivity and specificity) in terminally ill subjects by comparing premortem AMYVID scans to a postmortem truth standard of amyloid beta neuritic plaque density. A total of 59 subjects underwent autopsy after being dosed with AMYVID and imaged. The mean age was 79 years (range 47 to 103 years), half were females, and most were White (93%). A total of 29 subjects had an AD clinical diagnosis, 13 had another type of dementing disorder, 5 had mild cognitive impairment (MCI), and 12 had no cognitive impairment. The time interval between the AMYVID scan and death was less than one year for 46 subjects and between one and two years for 13 subjects. At autopsy, the global brain neuritic plaque density category (CERAD score as in Table 6 ) was: frequent (n=30); moderate (n=9); sparse (n=5); and none (n=15).
Study 2 evaluated inter-reader and intra-reader reproducibility of image interpretation using images from 59 subjects with a truth standard (same subjects as in Study 1) and 92 subjects without a truth standard (52 subjects with MCI, 20 subjects with AD, and 20 healthy subjects). Intra-reader reproducibility was assessed with images from 33 subjects (22%). Among the 151 subjects, the mean age was 75 years (range 47 to 103 years), half were females, and most were White (93%).
Among all subjects who underwent autopsy within two years (n=59; 39 positive and 20 negative based on histopathology), the sensitivity using the majority interpretation of the readers trained in-person (Study 1) was 92% (95% CI: 78%, 98%) and specificity was 100% (95% CI: 80%, 100%). The median (and range) of correct, false negative, and false positive reads were 55 (45, 56), 3 (2, 12), 1 (0, 2), respectively, for in-person training; and were 51 (46 to 54), 7 (3 to 12), 1 (1 to 2), respectively, for electronic media training. Table 7 shows the inter-reader reproducibility results among readers who underwent electronic media training (Study 2) for various groups of subjects. Inter-reader reproducibility analyses for all images showed an overall Fleiss' kappa statistic of 0.83 (95% CI: 0.78, 0.88); the lower bound of the 95% CI exceeded the pre-specified success criterion of 0.58. Intra-reader reproducibility analyses showed that between the two readings for each of the 33 duplicate scans, one of the five readers had complete agreement for all 33 scans, two readers had discrepant reads for a single scan, one reader had discrepant reads for two scans, and another reader had discrepant reads for three scans.
a Readers who underwent electronic media training. | |||||
b Shown is the median number of scans interpreted as positive across the five readers for each subgroup of subjects listed in the first column. | |||||
| Subject Group by Cognitive Status and Truth Standard (TS) | Positive Scans, n b | Kappa (95% CI) | Percent of Scans with Inter-Reader Agreement | ||
| 3 of 5 readers agreed | 4 of 5 readers agreed | 5 of 5 readers agreed | |||
| All subjects with a TS, n=59 | 33 | 0.75 (0.67, 0.83) | 14 | 10 | 76 |
| All subjects without a TS, n=92 | 33 | 0.88 (0.82, 0.94) | 2 | 11 | 87 |
| AD, n=49 (29 with TS; 20 no TS) | 38 | 0.67 (0.58, 0.76) | 10 | 14 | 76 |
| MCI, n=57 (5 with TS; 52 no TS) | 17 | 0.91 (0.83, 0.99) | 2 | 7 | 91 |
| Other non-AD dementia with TS, n=13 | 7 | 0.52 (0.35, 0.69) | 23 | 23 | 54 |
| Cognitively normal with TS, n=12 | 1 | 0.73 (0.55, 0.87) | 0 | 8 | 92 |
| Healthy subjects without TS, n=20 | 4 | 0.83 (0.69, 0.97) | 5 | 5 | 90 |
Selection of Patients Indicated for Amyloid Beta-Directed Therapy
Refer to the Prescribing Information of amyloid beta-directed therapy for description of clinical trials in which the efficacy of amyloid beta PET for selecting patients has been established.
Brain amyloid beta PET scans have been used to assess reduction of plaque in some clinical trials of amyloid beta-directed therapies as also described in the prescribing information of the therapeutic products.
HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
AMYVID (florbetapir F 18 injection) is a clear, colorless solution supplied in a shielded multiple-dose vial available as:
| Concentration @ EOS | Volume | Vial Size | NDC |
| 500 MBq/mL to 1,900 MBq/mL (13.5 mCi/mL to 51 mCi/mL) | 10 mL to 50 mL | 50 mL | 0002-1200-50 |
| 500 MBq/mL to 1,900 MBq/mL (13.5 mCi/mL to 51 mCi/mL) | 10 mL to 100 mL | 100 mL | 0002-1200-48 |
Storage and Handling
Store AMYVID in the original container with radiation shielding at 25ºC (77°F); excursions permitted to 15ºC to 30ºC (59°F to 86°F) [see USP Controlled Room Temperature].
AMYVID does not contain a preservative.
Do not use after the expiration date and time provided on the container label.
AMYVID multiple-dose vial expires 10 hours after EOS.
Dispose of unused product in accordance with all federal, state, and local laws and institutional requirements.
This preparation is for use by persons under license by the Nuclear Regulatory Commission or the relevant regulatory authority of an Agreement State.
Mechanism of Action
Florbetapir F 18 binds to amyloid beta plaques and the F 18 isotope produces a positron signal that is detected by a PET scanner. In in vitro binding studies using postmortem human brain homogenates containing amyloid beta plaques, the dissociation constant (Kd) for florbetapir was 3.7 ± 0.3 nM. The binding of florbetapir F 18 to amyloid beta aggregates was demonstrated in postmortem human brain sections using autoradiographic methods, thioflavin S and traditional silver staining correlation studies as well as immunohistochemistry (monoclonal antibody to amyloid beta) correlation studies. Florbetapir binding to tau protein and a battery of neuroreceptors was not detected in in vitro studies.