Amantadine Hydrochloride - Amantadine Hydrochloride solution prescribing information
INDICATIONS AND USAGE
Amantadine hydrochloride oral solution USP is indicated for the prophylaxis and treatment of signs and symptoms of infection caused by various strains of influenza A virus. Amantadine hydrochloride is also indicated in the treatment of parkinsonism and drug-induced extrapyramidal reactions.
Influenza A Prophylaxis
Amantadine hydrochloride is indicated for chemoprophylaxis against signs and symptoms of influenza A virus infection. Because amantadine hydrochloride does not completely prevent the host immune response to influenza A infection, individuals who take this drug may still develop immune responses to natural disease or vaccination and may be protected when later exposed to antigenically related viruses. Following vaccination during an influenza A outbreak, amantadine hydrochloride prophylaxis should be considered for the 2- to 4-week time period required to develop an antibody response.
Influenza A Treatment
Amantadine hydrochloride is also indicated in the treatment of uncomplicated respiratory tract illness caused by influenza A virus strains especially when administered early in the course of illness. There are no well-controlled clinical studies demonstrating that treatment with amantadine hydrochloride will avoid the development of influenza A virus pneumonitis or other complications in high risk patients.
There is no clinical evidence indicating that amantadine hydrochloride is effective in the prophylaxis or treatment of viral respiratory tract illnesses other than those caused by influenza A virus strains.
The following points should be considered before initiating treatment or prophylaxis with amantadine hydrochloride:
- Amantadine hydrochloride is not a substitute for early vaccination on an annual basis as recommended by the Centers for Disease Control and Prevention Advisory Committee on Immunization Practices.
- Influenza viruses change over time. Emergence of resistance mutations could decrease drug effectiveness. Other factors (for example, changes in viral virulence) might also diminish clinical benefit of antiviral drugs. Prescribers should consider available information on influenza drug susceptibility patterns and treatment effects when deciding whether to use amantadine hydrochloride.
Parkinson's Disease/Syndrome
Amantadine hydrochloride is indicated in the treatment of idiopathic Parkinson's disease (Paralysis Agitans), postencephalitic parkinsonism, and symptomatic parkinsonism which may follow injury to the nervous system by carbon monoxide intoxication. It is indicated in those elderly patients believed to develop parkinsonism in association with cerebral arteriosclerosis. In the treatment of Parkinson's disease, amantadine hydrochloride is less effective than levodopa, (-)-3-(3,4-dihydroxyphenyl)-L-alanine, and its efficacy in comparison with the anticholinergic antiparkinson drugs has not yet been established.
Drug-Induced Extrapyramidal Reactions
Amantadine hydrochloride is indicated in the treatment of drug-induced extrapyramidal reactions. Although anticholinergic-type side effects have been noted with amantadine hydrochloride when used in patients with drug-induced extrapyramidal reactions, there is lower incidence of these side effects than that observed with the anticholinergic antiparkinson drugs.
DOSAGE AND ADMINISTRATION
The dose of amantadine hydrochloride may need reduction in patients with congestive heart failure, peripheral edema, orthostatic hypotension, or impaired renal function. ( see Dosage for Impaired Renal Function ).
Dosage for Prophylaxis and Treatment of Uncomplicated Influenza A Virus Illness
Adult
The adult daily dosage of amantadine hydrochloride is 200 mg (four teaspoonfuls of oral solution) as a single daily dose. The daily dosage may be split into two teaspoonfuls, (100 mg) of oral solution twice a day. If central nervous system effects develop in once-a-day dosage, a split dosage schedule may reduce such complaints. In persons 65 years of age or older, the daily dosage of amantadine hydrochloride is 100 mg.
A 100 mg daily dose has also been shown in experimental challenge studies to be effective as prophylaxis in healthy adults who are not at high risk for influenza-related complications. However, it has not been demonstrated that a 100 mg daily dose is as effective as a 200 mg daily dose for prophylaxis, nor has the 100 mg daily dose been studied in the treatment of acute influenza illness. In recent clinical trials, the incidence of central nervous system (CNS) side effects associated with the 100 mg daily dose was at or near the level of placebo. The 100 mg dose is recommended for persons who have demonstrated intolerance to 200 mg of amantadine hydrochloride daily because of CNS or other toxicities.
Pediatric Patients
1 yr. to 9 yrs. of age
The total daily dose should be calculated on the basis of 2 to 4 mg/lb/day (4.4 to 8.8 mg/kg/day), but not to exceed 150 mg per day.
9 yrs. to 12 yrs. of age
The total daily dose is 200 mg given as two teaspoonfuls of oral solution twice a day. The 100 mg daily dose has not been studied in this pediatric population. Therefore, there are no data which demonstrate that this dose is as effective as or is safer than the 200 mg daily dose in this patient population.
Prophylactic dosing should be started in anticipation of an influenza A outbreak and before or after contact with individuals with influenza A virus respiratory tract illness.
Amantadine hydrochloride should be continued daily for at least 10 days following a known exposure. If amantadine hydrochloride is used chemoprophylactically in conjunction with inactivated influenza A virus vaccine until protective antibody responses develop, then it should be administered for 2 to 4 weeks after the vaccine has been given. When inactivated influenza A virus vaccine is unavailable or contraindicated, amantadine hydrochloride should be administered for the duration of known influenza A in the community because of repeated and unknown exposure.
Treatment of influenza A virus illness should be started as soon as possible, preferably within 24 to 48 hours after onset of signs and symptoms, and should be continued for 24 to 48 hours after the disappearance of signs and symptoms.
Dosage for Parkinsonism
Adult
The usual dose of amantadine hydrochloride is 100 mg twice a day when used alone. Amantadine hydrochloride has an onset of action usually within 48 hours.
The initial dose of amantadine hydrochloride is 100 mg daily for patients with serious associated medical illnesses or who are receiving high doses of other antiparkinson drugs. After one to several weeks at 100 mg once daily, the dose may be increased to 100 mg twice daily, if necessary.
Occasionally, patients whose responses are not optimal with amantadine hydrochloride at 200 mg daily may benefit from an increase up to 400 mg daily in divided doses. However, such patients should be supervised closely by their physicians.
Patients initially deriving benefit from amantadine hydrochloride not uncommonly experience a fall-off of effectiveness after a few months. Benefit may be regained by increasing the dose to 300 mg daily. Alternatively, temporary discontinuation of amantadine hydrochloride for several weeks, followed by reinitiation of the drug, may result in regaining benefit in some patients. A decision to use other antiparkinson drugs may be necessary.
Dosage for Concomitant Therapy
Some patients who do not respond to anticholinergic antiparkinson drugs may respond to amantadine hydrochloride. When amantadine hydrochloride or anticholinergic antiparkinson drugs are each used with marginal benefit, concomitant use may produce additional benefit.
When amantadine hydrochloride and levodopa are initiated concurrently, the patient can exhibit rapid therapeutic benefits. Amantadine hydrochloride should be held constant at 100 mg daily or twice daily while the daily dose of levodopa is gradually increased to optimal benefit.
When amantadine hydrochloride is added to optimal well-tolerated doses of levodopa, additional benefit may result, including smoothing out the fluctuations in improvement which sometimes occur in patients on levodopa alone. Patients who require a reduction in their usual dose of levodopa because of development of side effects may possibly regain lost benefit with the addition of amantadine hydrochloride.
Dosage for Drug-induced Extrapyramidal Reactions
Adult
The usual dose of amantadine hydrochloride is 100 mg twice a day. Occasionally, patients whose responses are not optimal with amantadine hydrochloride at 200 mg daily may benefit from an increase up to 300 mg daily in divided doses.
Dosage for Impaired Renal Function
Depending upon creatinine clearance, the following dosage adjustments are recommended:
| CREATININE CLEARANCE (mL/min/1.73m 2 ) | AMANTADINE HYDROCHLORIDE DOSAGE |
|---|---|
| 30 to 50 | 200 mg 1 st day and 100 mg each day thereafter |
| 15 to 29 | 200 mg 1 st day followed by 100 mg on alternate days |
| <15 | 200 mg every 7 days |
The recommended dosage for patients on hemodialysis is 200 mg every 7 days.
CONTRAINDICATIONS
Amantadine hydrochloride oral solution USP is contraindicated in patients with known hypersensitivity to amantadine hydrochloride or any of the other ingredients in the drug.
ADVERSE REACTIONS
The adverse reactions reported most frequently at the recommended dose of amantadine hydrochloride (5 to 10%) are: nausea, dizziness (lightheadedness), and insomnia.
Less frequently (1 to 5%) reported adverse reactions are: depression, anxiety and irritability, hallucinations, confusion, anorexia, dry mouth, constipation, ataxia, livedo reticularis, peripheral edema, orthostatic hypotension, headache, somnolence, nervousness, dream abnormality, agitation, dry nose, diarrhea and fatigue.
Infrequently (0.1 to 1%) occurring adverse reactions are: congestive heart failure, psychosis, urinary retention, dyspnea, skin rash, vomiting, weakness, slurred speech, euphoria, thinking abnormality, amnesia, hyperkinesia, hypertension, decreased libido, and visual disturbance, including punctate subepithelial or other corneal opacity, corneal edema, decreased visual acuity, sensitivity to light, and optic nerve palsy.
Rare (less than 0.1%) occurring adverse reactions are: instances of convulsion, leukopenia, neutropenia, eczematoid dermatitis, oculogyric episodes, suicidal attempt, suicide, and suicidal ideation ( see WARNINGS ).
Other adverse reactions reported during postmarketing experience with amantadine hydrochloride usage include:
Nervous System/Psychiatric
coma, stupor, delirium, hypokinesia, hypertonia, delusions, aggressive behavior, paranoid reaction, manic reaction, involuntary muscle contractions, gait abnormalities, paresthesia, EEG changes, and tremor. Abrupt discontinuation may also precipitate delirium, agitation, delusions, hallucinations, paranoid reaction, stupor, anxiety, depression and slurred speech;
Cardiovascular
cardiac arrest, arrhythmias including malignant arrhythmias, hypotension, and tachycardia;
Respiratory
acute respiratory failure, pulmonary edema, and tachypnea;
Gastrointestinal
dysphagia;
Hematologic
leukocytosis and agranulocytosis;
Special Senses
Keratitis, mydriasis, and corneal edema;
Skin and Appendages
pruritus and diaphoresis;
Miscellaneous
neuroleptic malignant syndrome ( see WARNINGS ), allergic reactions including anaphylactic reactions, edema and fever.
Laboratory Tests
elevated: CPK, BUN, serum creatinine, alkaline phosphatase, LDH, bilirubin, GGT, SGOT, and SGPT.
To report SUSPECTED ADVERSE REACTIONS, contact Pharmaceutical Associates, Inc. at 1-800-845-8210 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Careful observation is required when amantadine hydrochloride is administered concurrently with central nervous system stimulants.
Agents with anticholinergic properties may potentiate the anticholinergic-like side effects of amantadine hydrochloride.
Coadministration of thioridazine has been reported to worsen the tremor in elderly patients with Parkinson's disease, however, it is not known if other phenothiazines produce a similar response.
Coadministration of 1 Dyazide (triamterene/hydrochlorothiazide) resulted in a higher plasma amantadine hydrochloride concentration in a 61-year old man receiving amantadine hydrochloride 100 mg TID for Parkinson's disease. 1 It is not known which of the components of 1 Dyazide contributed to the observation or if related drugs produce a similar response.
Coadministration of quinine or quinidine with amantadine hydrochloride was shown to reduce the renal clearance of amantadine hydrochloride by about 30%.
The concurrent use of amantadine hydrochloride with live attenuated influenza vaccine (LAIV) intranasal has not been evaluated. However, because of the potential for interference between these products, LAIV should not be administered within 2 weeks before or 48 hours after administration of amantadine hydrochloride unless medically indicated. The concern about possible interference arises from the potential for antiviral drugs to inhibit replication of live vaccine virus. Trivalent inactivated influenza vaccine can be administered at any time relative to use of amantadine hydrochloride.
DESCRIPTION
Amantadine Hydrochloride Oral Solution USP is designated generically as amantadine hydrochloride and chemically as 1-adamantanamine hydrochloride.
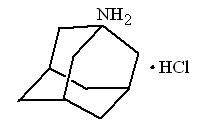
Amantadine hydrochloride is a stable white or nearly white crystalline powder, freely soluble in water and soluble in alcohol and in chloroform.
Amantadine hydrochloride has pharmacological actions as both an anti-Parkinson and an antiviral drug.
Amantadine Hydrochloride Oral Solution USP contains 50 mg of amantadine hydrochloride per 5 mL and has the following inactive ingredients: artificial raspberry flavor, citric acid, methylparaben, propylene glycol, propylparaben, purified water, sorbitol solution. May contain sodium hydroxide to adjust pH to approximately 2.3.
CLINICAL PHARMACOLOGY
Pharmacodynamics
Mechanisms of Action: Antiviral
The mechanism by which amantadine hydrochloride exerts its antiviral activity is not clearly understood. It appears to mainly prevent the release of infectious viral nucleic acid into the host cell by interfering with the function of the transmembrane domain of the viral M2 protein. In certain cases, amantadine hydrochloride is also known to prevent virus assembly during virus replication. It does not appear to interfere with the immunogenicity of inactivated influenza A virus vaccine.
Antiviral Activity
Amantadine hydrochloride inhibits the replication of influenza A virus isolates from each of the subtypes, i.e., H1N1, H2N2 and H3N2. It has very little or no activity against influenza B virus isolates. A quantitative relationship between the in vitro susceptibility of influenza A virus to amantadine hydrochloride and the clinical response to therapy has not been established in man. Sensitivity test results, expressed as the concentration of amantadine hydrochloride required to inhibit by 50% the growth of virus (ED50) in tissue culture vary greatly (from 0.1 µg/mL to 25.0 µg/mL) depending upon the assay protocol used, size of virus inoculum, isolates of influenza A virus strains tested, and the cell type used. Host cells in tissue culture readily tolerated amantadine hydrochloride up to a concentration of 100 µg/mL.
Drug Resistance
Influenza A variants with reduced in vitro sensitivity to amantadine hydrochloride have been isolated from epidemic strains in areas where adamantane derivatives are being used. Influenza viruses with reduced in vitro sensitivity have been shown to be transmissible and to cause typical influenza illness. The quantitative relationship between the in vitro sensitivity of influenza A variants to amantadine hydrochloride and the clinical response to therapy has not been established.
Mechanism of Action: Parkinson's Disease
The mechanism of action of amantadine hydrochloride in the treatment of Parkinson's disease and drug-induced extrapyramidal reactions is not known. Data from earlier animal studies suggest that amantadine hydrochloride may have direct and indirect effects on dopamine neurons. More recent studies have demonstrated that amantadine hydrochloride is a weak, non-competitive NMDA receptor antagonist (K i = 10µM). Although amantadine hydrochloride has not been shown to possess direct anticholinergic activity in animal studies, clinically, it exhibits anticholinergic-like side effects such as dry mouth, urinary retention, and constipation.
Pharmacokinetics
Amantadine hydrochloride is well absorbed orally. Maximum plasma concentrations are directly related to dose for doses up to 200 mg/day. Doses above 200 mg/day may result in a greater than proportional increase in maximum plasma concentrations. It is primarily excreted unchanged in the urine by glomerular filtration and tubular secretion. Eight metabolites of amantadine hydrochloride have been identified in human urine. One metabolite, an N-acetylated compound, was quantified in human urine and accounted for 5 to 15% of the administered dose. Plasma acetylamantadine accounted for up to 80% of the concurrent amantadine hydrochloride plasma concentration in 5 of 12 healthy volunteers following the ingestion of a 200 mg dose of amantadine hydrochloride. Acetylamantadine was not detected in the plasma of the remaining seven volunteers. The contribution of this metabolite to efficacy or toxicity is not known.
There appears to be a relationship between plasma amantadine hydrochloride concentrations and toxicity. As concentration increases, toxicity seems to be more prevalent; however, absolute values of amantadine hydrochloride concentrations associated with adverse effects have not been fully defined.
Amantadine hydrochloride pharmacokinetics were determined in 24 normal adult male volunteers after the oral administration of a single amantadine hydrochloride 100 mg soft gel capsule. The mean ± SD maximum plasma concentration was 0.22 ± 0.03 µg/mL (range: 0.18 to 0.32 µg/mL). The time to peak concentration was 3.3 ± 1.5 hours (range: 1.5 to 8.0 hours). The apparent oral clearance was 0.28 ± 0.11 L/hr/kg (range: 0.14 to 0.62 L/hr/kg). The half-life was 17 ± 4 hours (range: 10 to 25 hours). Across other studies, amantadine hydrochloride plasma half-life has averaged 16 ± 6 hours (range: 9 to 31 hours) in 19 healthy volunteers.
After oral administration of a single dose of 100 mg amantadine hydrochloride oral solution to five healthy volunteers, the mean ± SD maximum plasma concentration C max was 0.24 ± 0.04 µg/mL and ranged from 0.18 to 0.28 µg/mL. After 15 days of amantadine hydrochloride 100 mg b.i.d., the C max was 0.47 ± 0.11 µg/mL in four of the five volunteers. The administration of amantadine hydrochloride tablets as a 200 mg single dose to 6 healthy subjects resulted in a Cmax of 0.51 ± 0.14 µg/mL. Across studies, the time to C max (T max ) averaged about 2 to 4 hours.
Plasma amantadine hydrochloride clearance ranged from 0.2 to 0.3 L/hr/kg after the administration of 5 mg to 25 mg intravenous doses of amantadine hydrochloride to 15 healthy volunteers.
In six healthy volunteers, the ratio of amantadine hydrochloride renal clearance to apparent oral plasma clearance was 0.79 ± 0.17 (mean ± SD).
The volume of distribution determined after the intravenous administration of amantadine hydrochloride to 15 healthy subjects was 3 to 8 L/kg, suggesting tissue binding. Amantadine hydrochloride, after single oral 200 mg doses to 6 healthy young subjects and to 6 healthy elderly subjects has been found in nasal mucus at mean ± SD concentrations of 0.15 ± 0.16, 0.28 ± 0.26, and 0.39 ± 0.34 µg/g at 1, 4, and 8 hours after dosing, respectively. These concentrations represented 31 ± 33%, 59 ± 61%, and 95 ± 86% of the corresponding plasma amantadine hydrochloride concentrations. Amantadine hydrochloride is approximately 67% bound to plasma proteins over a concentration range of 0.1 to 2.0 µg/mL. Following the administration of amantadine hydrochloride 100 mg as a single dose, the mean ± SD red blood cell to plasma ratio ranged from 2.7 ± 0.5 in 6 healthy subjects to 1.4 ± 0.2 in 8 patients with renal insufficiency.
The apparent oral plasma clearance of amantadine hydrochloride is reduced and the plasma half-life and plasma concentrations are increased in healthy elderly individuals age 60 and older. After single dose administration of 25 to 75 mg to 7 healthy, elderly male volunteers, the apparent plasma clearance of amantadine hydrochloride was 0.10 ± 0.04 L/hr/kg (range 0.06 to 0.17 L/hr/kg) and the half-life was 29 ± 7 hours (range 20 to 41 hours). Whether these changes are due to decline in renal function or other age related factors is not known.
In a study of young healthy subjects (n=20), mean renal clearance of amantadine hydrochloride, normalized for body mass index, was 1.5 fold higher in males compared to females (p<0.032).
Compared with otherwise healthy adult individuals, the clearance of amantadine hydrochloride is significantly reduced in adult patients with renal insufficiency. The elimination half-life increases two to three fold or greater when creatinine clearance is less than 40 mL/min/1.73 m 2 and averages eight days in patients on chronic maintenance hemodialysis. Amantadine hydrochloride is removed in negligible amounts by hemodialysis.
The pH of the urine has been reported to influence the excretion rate of amantadine hydrochloride. Since the excretion rate of amantadine hydrochloride increases rapidly when the urine is acidic, the administration of urine acidifying drugs may increase the elimination of the drug from the body.
HOW SUPPLIED
Amantadine Hydrochloride Oral Solution USP 50 mg/5 mL is colorless with a raspberry flavor and is supplied in the following oral dosage forms:
NDC 0121-0646-16: 16 fl oz (473 mL) bottle
NDC 0121-0646-10: 10 mL unit dose cup. Case contains 100 unit dose cups of 10 mL packaged in 10 trays of 10 unit dose cups each.
Dispense in a tight container as defined in the USP, with a child-resistant closure (as required).
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
•Dyazide is a registered trademark of GlaxoSmithKline.
Mechanisms of Action: Antiviral
The mechanism by which amantadine hydrochloride exerts its antiviral activity is not clearly understood. It appears to mainly prevent the release of infectious viral nucleic acid into the host cell by interfering with the function of the transmembrane domain of the viral M2 protein. In certain cases, amantadine hydrochloride is also known to prevent virus assembly during virus replication. It does not appear to interfere with the immunogenicity of inactivated influenza A virus vaccine.
Antiviral Activity
Amantadine hydrochloride inhibits the replication of influenza A virus isolates from each of the subtypes, i.e., H1N1, H2N2 and H3N2. It has very little or no activity against influenza B virus isolates. A quantitative relationship between the in vitro susceptibility of influenza A virus to amantadine hydrochloride and the clinical response to therapy has not been established in man. Sensitivity test results, expressed as the concentration of amantadine hydrochloride required to inhibit by 50% the growth of virus (ED50) in tissue culture vary greatly (from 0.1 µg/mL to 25.0 µg/mL) depending upon the assay protocol used, size of virus inoculum, isolates of influenza A virus strains tested, and the cell type used. Host cells in tissue culture readily tolerated amantadine hydrochloride up to a concentration of 100 µg/mL.
Drug Resistance
Influenza A variants with reduced in vitro sensitivity to amantadine hydrochloride have been isolated from epidemic strains in areas where adamantane derivatives are being used. Influenza viruses with reduced in vitro sensitivity have been shown to be transmissible and to cause typical influenza illness. The quantitative relationship between the in vitro sensitivity of influenza A variants to amantadine hydrochloride and the clinical response to therapy has not been established.