Get your patient on Allernaze (Triamcinolone Acetonide)
Allernaze prescribing information
INDICATIONS AND USAGE:
AllerNaze is indicated for the treatment of the nasal symptoms of seasonal and perennial allergic rhinitis in adults and children 12 years of age or older.
DOSAGE AND ADMINISTRATION:
The recommended starting dose of AllerNaze for most patients is 200 mcg per day given as 2 sprays (approximately 50 mcg/spray) in each nostril once a day. The maximum dose should not exceed 400 mcg per day. If the 400 mcg dose is used, it may be given either as a once a day dosage (4 sprays in each nostril) or divided into two daily doses of two sprays/nostril twice a day.
The nasal spray pump must be primed before AllerNaze is used for the first time. To prime the pump, press down on the shoulder of the white nasal applicator using your forefinger and middle finger while supporting the base of the bottle with your thumb. Press down and release the pump until it sprays 3 times or until a fine mist is observed (see DIRECTIONS FOR USE ).
Some patients may obtain relief of symptoms sooner when started on a 400 mcg per day dose of AllerNaze than with 200 mcg per day. Onset of significant relief of nasal symptoms was seen within two days after starting treatment at 400 mcg once daily. A starting dose of 400 mcg per day may be considered in patients when starting therapy with AllerNaze in cases where a faster onset of relief is desirable. Generally, maximum relief of symptoms may take several days or up to one week to occur.
After symptoms have been brought under control, patients should be titrated to the minimum effective dose to reduce the possibility of adverse effects.
If relief of symptoms is not achieved after 14-21 days of AllerNaze therapy given in an adequate dose, AllerNaze should be discontinued and alternative diagnosis and therapies considered.
AllerNaze is not recommended for use in persons under 12 years of age since its safety and effectiveness have not been established in this age group.
DIRECTION FOR USE:
Illustrated patient instructions for use accompany each package of AllerNaze .
CONTRAINDICATIONS:
AllerNaze is contraindicated in patients with a hypersensitivity to any of its ingredients.
ADVERSE REACTIONS:
In adequate, well-controlled and uncontrolled studies, 1187 patients have received triamcinolone acetonide solution. The adverse reactions summarized below, are based upon seven placebo controlled clinical trials of 2-6 weeks duration in 847 patients with seasonal or perennial allergic rhinitis (504 patients received 200 mcg or 400 mcg per day of triamcinolone acetonide solution and 343 patients received vehicle placebo). Adverse events reported by 2% or more of patients (regardless of relationship to treatment) who received triamcinolone acetonide solution 200 or 400 mcg once daily and that were more common with triamcinolone acetonide solution than with placebo are displayed in the table below. Overall, the incidence and nature of adverse events with triamcinolone acetonide solution 400 mcg was comparable to that seen with triamcinolone acetonide solution 200 mcg and with vehicle placebo.
ADVERSE EVENTS | 200 mcg of triamcinolone acetonide once daily n = 204 | 400 mcg of triamcinolone acetonide once daily n = 300 | Combined (200 and 400 mcg) use of triamcinolone acetonide n = 504 | Vehicle Placebo n = 343 |
| BODY AS A WHOLE | ||||
| Headache | 51.0% | 44.3% | 47.0% | 41.1% |
| Back Pain | 7.8% | 4.7% | 6.0% | 3.5% |
| RESPIRATORY SYSTEM | ||||
| Pharyngitis | 13.7% | 10.3% | 11.7% | 7.9% |
| Asthma | 5.4% | 4.3% | 4.8% | 2.9% |
| Cough Increased | 2.0% | 2.7% | 2.4% | 2.3% |
| DIGESTIVE SYSTEM | ||||
| Dyspepsia | 4.9% | 2.7% | 3.6% | 2.0% |
| Nausea | 2.0% | 3.0% | 2.6% | 0.6% |
| Vomiting | 1.5% | 2.7% | 2.2% | 1.5% |
| SPECIAL SENSES | ||||
| Taste Perversion | 7.8% | 5.0% | 6.2% | 2.9% |
| Conjunctivitis | 4.4% | 1.3% | 2.6% | 1.5% |
| MUSCULOSKELETAL SYSTEM | ||||
| Myalgia | 2.5% | 3.3% | 3.0% | 2.6% |
Adverse events reported by 2% or more of patients who received triamcinolone acetonide solution 200 or 400 mcg once daily and that were more common with placebo than with triamcinolone acetonide solution included: application site reaction (e.g. transient nasal burning and stinging), rhinitis, dysmenorrhea, pain (unspecified) and allergic reaction.
The adverse effects related to the irritation of nasal mucous membranes (i.e. application site reaction) did not usually interfere with treatment. In the controlled and uncontrolled studies, approximately 0.3% of patients discontinued because of irritation of nasal mucous membranes.
DESCRIPTION:
Triamcinolone acetonide, the active ingredient of AllerNaze , is a corticosteroid with the chemical name, 9α-Fluoro-11β,16α, 17, 21-tetrahydroxypregna-1,4-diene-3, 20-dione cyclic 16, 17-acetal with acetone (C 24 H 31 FO 6 ). Its structural formula is:
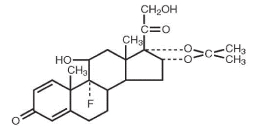
Triamcinolone acetonide, USP, is a white crystalline powder, with a molecular weight of 434.51. It is practically insoluble in water, and sparingly soluble in dehydrated alcohol, in chloroform and in methanol. It has a melting point temperature range between 292° and 294°C.
AllerNaze is a metered-dose manual spray pump in an amber polyethylene terephthalate (PET) bottle with 0.05% w/v triamcinolone acetonide in a solution containing citric acid, edetate disodium, polyethylene glycol 3350, propylene glycol, purified water, sodium citrate, and 0.01% benzalkonium chloride as a preservative. AllerNaze pH is 5.3.
After initial priming (three sprays) of the AllerNaze metered pump delivery system, each spray will deliver 50 mcg of triamcinolone acetonide. If the pump was not used for more than 14 days, reprime with 3 sprays or until a fine mist is observed. Each 15 mL bottle contains 7.5 mg of triamcinolone acetonide to deliver 120 metered sprays. After 120 sprays, the amount of triamcinolone acetonide delivered per spray may not be consistent and the bottle should be discarded.
CLINICAL PHARMACOLOGY:
Triamcinolone acetonide is a more potent derivative of triamcinolone. Triamcinolone acetonide is approximately eight times more potent than prednisone in animal models of inflammation. The clinical significance of this is unclear.
Although the precise mechanism of corticosteroid antiallergic action is unknown, corticosteroids have been shown to have a wide range of effects on multiple cell types (e.g. mast cells, eosinophils, neutrophils, macrophages, and lymphocytes) and mediators (e.g. histamines, eicosanoids, leukotrienes, and cytokines) involved in inflammation.
Pharmacokinetics:
Absorption :
The pharmacokinetics of triamcinolone acetonide solution was evaluated in a single-dose study conducted in 24 patients with perennial allergic rhinitis. Following a single intranasal dose of 400 mcg of triamcinolone acetonide (twice the recommended starting dose of triamcinolone acetonide solution), the mean C max of the drug was 1.12 ng/mL (SD = 0.38) with a median T max of 0.5 hours (range: 0.08 - 1.0).
A pharmacokinetic study to demonstrate dose proportionality was conducted in patients with perennial allergic rhinitis. The C max and AUC of the 200 and 400 mcg doses increased less than proportionally when compared to the 100 mcg dose. Following multiple dosing (100 or 200 or 400 mcg QD for 7 days), there was no evidence of drug accumulation.
Distribution :
The volume of distribution (Vd) reported was 99.5 L (SD = 27.5).
Metabolism :
In animal studies using rats and dogs, three metabolites of triamcinolone acetonide have been identified. They are 6β-hydroxytriamcinolone acetonide, 21-carboxytriamcinolone acetonide and 21-carboxy-6β-hydroxytriamcinolone acetonide. All three metabolites are expected to be substantially less active than the parent compound due to (a) the dependence of anti-inflammatory activity on the presence of a 21-hydroxyl group, (b) the decreased activity observed upon 6-hydroxylation, and (c) the markedly increased water solubility favoring rapid elimination. There appeared to be some quantitative differences in the metabolites among species. No differences were detected in metabolic pattern as a function of route of administration.
Elimination :
After a single intranasal dose of 400 mcg of triamcinolone acetonide (twice the recommended starting dose of triamcinolone acetonide solution), the mean observed elimination half-life was 2.26 hours (SD=0.77). Based upon intravenous dosing of triamcinolone acetonide phosphate ester, the half-life of triamcinolone acetonide was reported to be 88 minutes. The reported clearance was 45.2 L/hour (SD=9.1) for triamcinolone acetonide.
Special Populations
Age :
The effect of age, specifically in geriatric and pediatric patients, on the pharmacokinetics of triamcinolone acetonide has not been studied.
Gender :
Gender did not significantly influence the pharmacokinetics of triamcinolone acetonide solution.
Race :
The effect of race on the pharmacokinetics of triamcinolone acetonide solution has not been studied.
Renal/Hepatic Insufficiency :
No specific pharmacokinetic studies have been conducted in renally or hepatically impaired subjects.
Drug-Drug Interactions :
No specific drug-drug interactions have been investigated.
Pharmacodynamics:
A small (approximately 5 to 7 patients per treatment group), parallel trial was conducted to assess the effect of triamcinolone acetonide solution on the Hypothalamic-Pituitary-Adrenal (HPA) axis. Patients with allergic rhinitis were treated for six weeks with 400 mcg, 800 mcg, or 1600 mcg total daily doses of triamcinolone acetonide solution, 10 mg oral prednisone once daily, or placebo. Adrenal response to a six-hour cosyntropin stimulation test suggests that intranasal triamcinolone acetonide solution 400 mcg/day for six weeks did not measurably affect adrenal activity. Triamcinolone acetonide solution treatment arms using doses of 800 and 1600 mcg/day demonstrated a trend toward dose-related suppression of HPA response. However, this decrease did not reach statistical significance, whereas 10 mg daily oral prednisone did.
CLINICAL TRIALS:
The efficacy of triamcinolone acetonide solution has been evaluated in 746 patients with seasonal or perennial allergic rhinitis who completed 8 controlled clinical trials.
In total, 1187 patients have been treated with triamcinolone acetonide solution in the clinical development program. Three adequate and well controlled multi-center trials involving 541 patients with seasonal allergic rhinitis who received doses of triamcinolone acetonide solution ranging from 50 mcg to 400 mcg once daily were conducted. These trials evaluated the total nasal symptom scores that included stuffiness, rhinorrhea, itching, and sneezing.
The results showed that patients who received ≥ 200 mcg daily of the active drug had statistically significant relief in the total nasal symptom score compared to those receiving placebo.
In one clinical trial that examined efficacy after 2 days of 200 or 400 mcg triamcinolone acetonide solution treatment, only the 400 mcg dose showed statistically significant improvement over placebo in the nasal symptoms of seasonal allergic rhinitis.
HOW SUPPLIED:
Each 15 mL bottle of AllerNaze (NDC 27437-143-01) contains 7.5 mg (0.50 mg/mL) of triamcinolone acetonide, USP and is fitted with a meter pump with white nasal applicator, teal blue dust cover and teal blue locking clip sealed in a foil pouch. The unit delivers 120 metered actuations and comes with a patient's instructions for use leaflet. The bottle should be discarded when the labeled number of actuations has been reached even though the bottle is not completely empty.
Keep out of reach of children.
Store at controlled room temperature: 20°-25°C (68°-77°F). Protect from freezing.
Use AllerNaze within 2 months after opening of the protective foil pouch or before expiration date, whichever comes first.
Rx only
Lupin Pharma Baltimore, MD 21202 Tel: 1-800-399-2561 www.lupinpharmaceuticals.com
AllerNaze TM [AL-er-nāz]
(triamcinolone acetonide, USP)
Nasal Spray