Get your patient on Albuterol Sulfate - Albuterol Sulfate tablet (Albuterol Sulfate)
Albuterol Sulfate - Albuterol Sulfate tablet prescribing information
INDICATIONS AND USAGE
Albuterol tablets are indicated for the relief of bronchospasm in adults and children 6 years of age and older with reversible obstructive airway disease.
DOSAGE AND ADMINISTRATION
The following dosages of albuterol tablets are expressed in terms of albuterol base.
Usual Dosage
Adults and Children Over 12 Years of Age
The usual starting dosage for adults and children 12 years and older is 2 or 4 mg three or four times a day.
Children 6 to 12 Years of Age
The usual starting dosage for children 6 to 12 years of age is 2 mg three or four times a day.
Dosage Adjustment
Adults and Children Over 12 Years of Age
For adults and children 12 years and older, a dosage above 4 mg four times a day should be used only when the patient fails to respond. If a favorable response does not occur with the 4 mg initial dosage, it should be cautiously increased stepwise up to a maximum of 8 mg four times a day as tolerated.
Children 6 to 12 Years of Age Who Fail to Respond to the Initial Starting Dosage of 2 mg Four Times a Day
For children from 6 to 12 years of age who fail to respond to the initial starting dosage of 2 mg four times a day, the dosage may be cautiously increased stepwise, but not to exceed 24 mg/day (given in divided doses).
Elderly Patients and Those Sensitive to Beta-adrenergic Stimulators
An initial dosage of 2 mg three or four times a day is recommended for elderly patients and for those with a history of unusual sensitivity to beta-adrenergic stimulators. If adequate bronchodilation is not obtained, dosage may be increased gradually to as much as 8 mg three or four times a day.
The total daily dose should not exceed 32 mg in adults and children 12 years and older.
CONTRAINDICATIONS
Albuterol tablets are contraindicated in patients with a history of hypersensitivity to albuterol, or any of its components.
ADVERSE REACTIONS
In clinical trials, the most frequent adverse reactions to albuterol tablets were:
Percent Incidence of Adverse Reactions | |
Reaction | Percent Incidence |
Central nervous system | |
Nervousness | 20% |
Tremor | 20% |
Headache | 7% |
Sleeplessness | 2% |
Weakness | 2% |
Dizziness | 2% |
Drowsiness | <1% |
Restlessness | <1% |
Irritability | <1% |
Cardiovascular | |
Tachycardia | 5% |
Palpitations | 5% |
Chest discomfort | <1% |
Flushing | <1% |
Musculoskeletal | |
Muscle cramps | 3% |
Gastrointestinal | |
Nausea | 2% |
Genitourinary | |
Difficulty in micturition | <1% |
Rare cases of urticaria, angioedema, rash, bronchospasm, and oropharyngeal edema have been reported after the use of albuterol.
In addition, albuterol, like other sympathomimetic agents, can cause adverse reactions such as hypertension, angina, vomiting, vertigo, central nervous system stimulation, unusual taste, and drying or irritation of the oropharynx.
The reactions are generally transient in nature, and it is usually not necessary to discontinue treatment with albuterol tablets. In selected cases, however, dosage may be reduced temporarily; after the reaction has subsided, dosage should be increased in small increments to the optimal dosage.
DESCRIPTION
Albuterol tablets contain albuterol sulfate USP, the racemic form of albuterol and a relatively selective beta 2 -adrenergic bronchodilator. Albuterol sulfate has the chemical name α 1 -[( tert -butylamino)methyl]-4-hydroxy- m -xylene- α,α-diol sulfate (2:1) (salt) and the following structural formula:
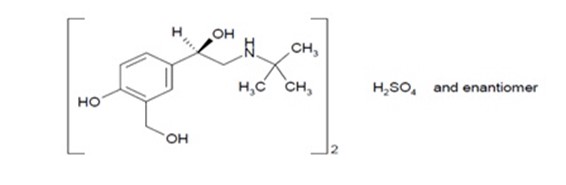
Albuterol sulfate has a molecular weight of 576.7, and the molecular formula is (C 13 H 21 NO 3 ) 2 H 2 SO 4 . Albuterol sulfate, USP is a white or almost white crystalline powder, freely soluble in water, practically insoluble or very slightly soluble in Methylene chloride and alcohol, slightly soluble in Chloroform and Ether.
The World Health Organization recommended name for albuterol base is salbutamol.
Each albuterol tablet for oral administration contains 2 mg or 4 mg of albuterol as 2.4 mg or 4.8 mg of albuterol sulfate USP, respectively. Each tablet also contains the following inactive ingredients: lactose monohydrate, pregelatinized starch, corn starch, and magnesium stearate.
CLINICAL PHARMACOLOGY
In vitro studies and in vivo pharmacologic studies have demonstrated that albuterol has a preferential effect on beta 2 -adrenergic receptors compared with isoproterenol. While it is recognized that beta 2 -adrenergic receptors are the predominant receptors in bronchial smooth muscle, data indicate that there is a population of beta 2 -receptors in the human heart existing in a concentration between 10% and 50%. The precise function of these receptors has not been established (see WARNINGS ).
The pharmacologic effects of beta-adrenergic agonist drugs, including albuterol, are at least in part attributable to stimulation through beta-adrenergic receptors of intracellular adenyl cyclase, the enzyme that catalyzes the conversion of adenosine triphosphate (ATP) to cyclic-3',5'-adenosine monophosphate (cyclic AMP). Increased cyclic AMP levels are associated with relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells.
Albuterol has been shown in most controlled clinical trials to have more effect on the respiratory tract, in the form of bronchial smooth muscle relaxation, than isoproterenol at comparable doses while producing fewer cardiovascular effects.
Albuterol is longer acting than isoproterenol in most patients by any route of administration because it is not a substrate for the cellular uptake processes for catecholamines nor for catechol- O -methyl transferase.
Preclinical
Intravenous studies in rats with albuterol sulfate have demonstrated that albuterol crosses the blood brain barrier and reaches brain concentrations amounting to approximately 5% of the plasma concentrations. In structures outside the brain barrier (pineal and pituitary glands), albuterol concentrations were found to be 100 times those in the whole brain.
Studies in laboratory animals (minipigs, rodents, and dogs) have demonstrated the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when beta-agonists and methylxanthines are administered concurrently. The clinical significance of these findings is unknown.
Pharmacokinetics
Albuterol is rapidly absorbed after oral administration of one 4 mg albuterol tablet in normal volunteers. Maximum plasma concentrations of about 18 ng/mL of albuterol are achieved within 2 hours, and the drug is eliminated with a half-life of about 5 hours.
In other studies, the analysis of urine samples of patients given 8 mg of tritiated albuterol orally showed that 76% of the dose was excreted over 3 days, with the majority of the dose being excreted within the first 24 hours. Sixty percent of this radioactivity was shown to be the metabolite. Feces collected over this period contained 4% of the administered dose.
Clinical Trials
In controlled clinical trials in patients with asthma, the onset of improvement in pulmonary function, as measured by maximum midexpiratory flow rate (MMEF), was within 30 minutes after a dose of albuterol, with peak improvement occurring between 2 and 3 hours. In controlled clinical trials in which measurements were conducted for 6 hours, clinically significant improvement (defined as maintaining a 15% or more increase in forced expiratory volume in 1 second [FEV 1 ] and a 20% or more increase in MMEF over baseline values) was observed in 60% of patients at 4 hours and in 40% at 6 hours. In other single-dose, controlled clinical trials, clinically significant improvement was observed in at least 40% of the patients at 8 hours. No decrease in the effectiveness of albuterol was reported in patients who received long-term treatment with the drug in uncontrolled studies for periods up to 6 months.
HOW SUPPLIED
Albuterol Tablets, USP are available containing albuterol sulfate, USP 2.4 mg equivalent to albuterol 2 mg or albuterol sulfate, USP 4.8 mg equivalent to albuterol 4 mg.
The 2 mg tablets are white to off white round flat face beveled edge tablets, debossed with “CE” over “84” on one side and score on the other side. They are available as follows:
Bottles of 90 tablets NDC 62135-671-90
The 4 mg tablets are white to off white round flat face beveled edge tablets, debossed with “CE” over “85” on one side and score on the other side. They are available as follows:
Bottles of 90 tablets NDC 62135-672-90
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature]. Protect from light.
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure.
Manufactured for:
Chartwell RX, LLC. Congers, NY 10920
L71378 Revised: 06/2023