Get your patient on Albuterol Sulfate - Albuterol Sulfate aerosol, Metered (Albuterol Sulfate)
Albuterol Sulfate - Albuterol Sulfate aerosol, Metered prescribing information
USE IN SPECIFIC POPULATIONS
Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in women exposed to asthma medications during pregnancy. For more information, contact the Mothers To Baby Pregnancy Studies conducted by the Organization of Teratology Information Specialists at 1-877-311-8972 or visit http://mothertobaby.org/pregnancy-studies/.
Risk Summary
There are no randomized clinical studies of use of albuterol during pregnancy. Available data from published epidemiological studies and postmarketing case reports of pregnancy outcomes following inhaled albuterol use do not consistently demonstrate a risk of major birth defects or miscarriage. There are clinical considerations with use of albuterol in pregnant women [see Clinical Considerations]. In animal reproduction studies, when albuterol sulfate was administered subcutaneously to pregnant mice there was evidence of cleft palate at less than and up to 9 times the maximum recommended human daily inhalation dose (MRHDID) [see Data] .
The estimated background risk of major birth defects and miscarriage for the indicated population(s) are unknown. In the U.S. general population, the estimated risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
In women with poorly or moderately controlled asthma, there is an increased risk of preeclampsia in the mother and prematurity, low birth weight, and small for gestational age in the neonate. Pregnant women should be closely monitored and medication adjusted as necessary to maintain optimal control.
Labor or Delivery
Because of the potential for beta-agonist interference with uterine contractility, use of albuterol sulfate inhalation aerosol for relief of bronchospasm during labor should be restricted to those patients in whom the benefits clearly outweigh the risk. Albuterol sulfate inhalation aerosol has not been approved for the management of pre-term labor. Serious adverse reactions, including pulmonary edema, have been reported during or following treatment of premature labor with beta 2 -agonists, including albuterol.
Data
Animal Data
In a mouse reproduction study, subcutaneously administered albuterol sulfate produced cleft palate formation in 5 of 111 (4.5%) fetuses at an exposure nine-tenths of the MRHDID for adults (on a mg/m 2 basis at a maternal dose of 0.25 mg/kg) and in 10 of 108 (9.3%) fetuses at approximately 9 times the MRHDID (on a mg/m 2 basis at a maternal dose of 2.5 mg/kg). Similar effects were not observed at approximately one-eleventh the MRHDID for adults (on a mg/m 2 basis at a maternal dose of 0.025 mg/kg). Cleft palate also occurred in 22 of 72 (30.5%) fetuses from females treated subcutaneously with isoproterenol (positive control).
In a rabbit reproduction study, orally administered albuterol sulfate induced cranioschisis in 7 of 19 fetuses (37%) at approximately 750 times the MRHDID (on a mg/m 2 basis at a maternal dose of 50 mg/kg).
In a rat reproduction study, an albuterol sulfate/HFA-134a formulation administered by inhalation did not produce any teratogenic effects at exposures approximately 80 times the MRHDID (on a mg/m 2 basis at a maternal dose of 10.5 mg/kg).
A study in which pregnant rats were dosed with radiolabeled albuterol sulfate demonstrated that drug-related material is transferred from the maternal circulation to the fetus.
Lactation
Risk Summary
There are no available data on the presence of albuterol in human milk, the effects on the breastfed child, or the effects on milk production. However, plasma levels of albuterol after inhaled therapeutic doses are low in humans, and if present in breast milk, albuterol has a low oral bioavailability [see Clinical Pharmacology (12.3)] .
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for albuterol and any potential adverse effects on the breastfed child from albuterol or from the underlying maternal condition.
Pediatric Use
The safety and effectiveness of albuterol sulfate inhalation aerosol for the treatment or prevention of bronchospasm in children 12 years of age and older with reversible obstructive airway disease is based on one 6-week clinical trial in 116 patients 12 years of age and older with asthma comparing doses of 180 mcg four times daily with placebo, and one single-dose crossover study comparing doses of 90, 180, and 270 mcg with placebo in 58 patients [see Clinical Studies (14.1)] . The safety and effectiveness of albuterol sulfate inhalation aerosol for treatment of exercise-induced bronchospasm in children 12 years of age and older is based on one single-dose crossover study in 24 adults and adolescents with exercise-induced bronchospasm comparing doses of 180 mcg with placebo [see Clinical Studies (14.2)] .
The safety of albuterol sulfate inhalation aerosol in children 4 to 11 years of age is based on one 3-week clinical trial in 50 patients 4 to 11 years of age with asthma using the same formulation of albuterol as in albuterol sulfate inhalation aerosol comparing doses of 180 mcg four times daily with placebo. The effectiveness of albuterol sulfate inhalation aerosol in children 4 to 11 years of age is extrapolated from clinical trials in patients 12 years of age and older with asthma and exercise-induced bronchospasm, based on data from a single-dose study comparing the bronchodilatory effect of albuterol sulfate inhalation aerosol 90 mcg and 180 mcg with placebo in 55 patients with asthma and a 3-week clinical trial using the same formulation of albuterol as in albuterol sulfate inhalation aerosol in 95 asthmatic children 4 to 11 years of age comparing a dose of 180 mcg albuterol four times daily with placebo [see Clinical Studies (14.1)] . The safety and effectiveness of albuterol sulfate inhalation aerosol in pediatric patients below the age of 4 years have not been established.
Geriatric Use
Clinical studies of albuterol sulfate inhalation aerosol did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. Other reported clinical experience has not identified differences in responses between elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy [see Warnings and Precautions (5.4, 5.7)] .
All beta 2 -adrenergic agonists, including albuterol, are known to be substantially excreted by the kidney, and the risk of toxic reactions may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
DESCRIPTION
The active ingredient of albuterol sulfate inhalation aerosol is albuterol sulfate, a racemic salt, of albuterol. Albuterol sulfate has the chemical name α 1 -[( tert -butylamino) methyl]-4-hydroxy- m -xylene-α,α'-diol sulfate (2:1) (salt), and has the following chemical structure:
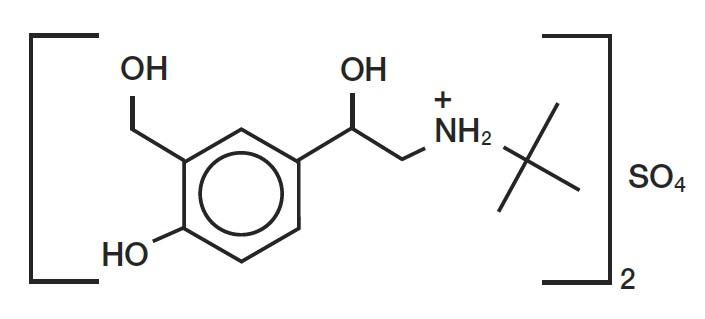
The molecular weight of albuterol sulfate is 576.7, and the empirical formula is (C 13 H 21 NO 3 ) 2 •H 2 SO 4 . Albuterol sulfate is a white to off-white crystalline powder. It is soluble in water and slightly soluble in ethanol. Albuterol sulfate is the official generic name in the United States, and salbutamol sulfate is the World Health Organization recommended generic name. Albuterol sulfate inhalation aerosol is a pressurized metered-dose aerosol unit with a dose counter. Albuterol sulfate inhalation aerosol is for oral inhalation only. It contains a microcrystalline suspension of albuterol sulfate in propellant HFA-134a (1, 1, 1, 2-tetrafluoroethane) and ethanol.
Prime the inhaler before using for the first time and in cases where the inhaler has not been used for more than 2 weeks by releasing three sprays into the air, away from the face. After priming, each actuation delivers 108 mcg albuterol sulfate, from the actuator mouthpiece (equivalent to 90 mcg of albuterol base). Each canister provides 200 actuations (inhalations).
This product does not contain chlorofluorocarbons (CFCs) as the propellant.
CLINICAL PHARMACOLOGY
Mechanism of Action
Albuterol sulfate is a beta 2 -adrenergic agonist. The pharmacologic effects of albuterol sulfate are attributable to activation of beta 2 -adrenergic receptors on airway smooth muscle. Activation of beta 2 -adrenergic receptors leads to the activation of adenylcyclase and to an increase in the intracellular concentration of cyclic-3', 5'-adenosine monophosphate (cyclic AMP). This increase of cyclic AMP is associated with the activation of protein kinase A, which in turn inhibits the phosphorylation of myosin and lowers intracellular ionic calcium concentrations, resulting in muscle relaxation. Albuterol relaxes the smooth muscle of all airways, from the trachea to the terminal bronchioles. Albuterol acts as a functional antagonist to relax the airway irrespective of the spasmogen involved, thus protecting against all bronchoconstrictor challenges. Increased cyclic AMP concentrations are also associated with the inhibition of release of mediators from mast cells in the airway. While it is recognized that beta 2 -adrenergic receptors are the predominant receptors on bronchial smooth muscle, data indicate that there are beta-receptors in the human heart, 10% to 50% of which are cardiac beta 2 -adrenergic receptors. The precise function of these receptors has not been established [see Warnings and Precautions (5.4)] .
Albuterol has been shown in most controlled clinical trials to have more effect on the respiratory tract, in the form of bronchial smooth muscle relaxation, than isoproterenol at comparable doses while producing fewer cardiovascular effects. However, inhaled albuterol, like other beta-adrenergic agonist drugs, can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or electrocardiographic changes [see Warnings and Precautions (5.4)] .
Pharmacokinetics
The systemic levels of albuterol are low after inhalation of recommended doses. In a crossover study conducted in healthy male and female volunteers, high cumulative doses of albuterol sulfate inhalation aerosol (1,080 mcg of albuterol base administered over one hour) yielded mean peak plasma concentrations (C max ) and systemic exposure (AUC inf ) of approximately 4,100 pg/mL and 28,426 pg/mL•hr, respectively compared to approximately 3,900 pg/mL and 28,395 pg/mL•hr, respectively following the same dose of an active HFA-134a albuterol inhaler comparator. The terminal plasma half-life of albuterol delivered by albuterol sulfate inhalation aerosol was approximately 6 hours. Comparison of the pharmacokinetic parameters demonstrated no differences between the products.
The pharmacokinetic profile of albuterol sulfate inhalation aerosol was evaluated in a two-way cross-over study in 11 healthy pediatric volunteers, 4 to 11 years of age. A single dose administration of albuterol sulfate inhalation aerosol (180 mcg albuterol base) yielded a least square mean (SE) C max and AUC 0-∞ of 1,100 (1.18) pg/mL and 5,120 (1.15) pg/mL•hr, respectively. The least square mean (SE) terminal plasma half-life of albuterol delivered by albuterol sulfate inhalation aerosol was 166 (7.8) minutes.
Metabolism and Elimination: Information available in the published literature suggests that the primary enzyme responsible for the metabolism of albuterol in humans is SULTIA3 (sulfotransferase). When racemic albuterol was administered either intravenously or via inhalation after oral charcoal administration, there was a 3- to 4-fold difference in the area under the concentration-time curves between the (R)- and (S)-albuterol enantiomers, with (S)-albuterol concentrations being consistently higher. However, without charcoal pretreatment, after either oral or inhalation administration the differences were 8- to 24-fold, suggesting that the (R)-albuterol is preferentially metabolized in the gastrointestinal tract, presumably by SULTIA3.
The primary route of elimination of albuterol is through renal excretion (80% to 100%) of either the parent compound or the primary metabolite. Less than 20% of the drug is detected in the feces. Following intravenous administration of racemic albuterol, between 25% and 46% of the (R)-albuterol fraction of the dose was excreted as unchanged (R)-albuterol in the urine.
Geriatric, Pediatric, Hepatic/Renal Impairment: No pharmacokinetic studies for albuterol sulfate inhalation aerosol have been conducted in neonates or elderly subjects.
The effect of hepatic impairment on the pharmacokinetics of albuterol sulfate inhalation aerosol has not been evaluated.
The effect of renal impairment on the pharmacokinetics of albuterol was evaluated in 5 subjects with creatinine clearance of 7 to 53 mL/min, and the results were compared with those from healthy volunteers. Renal disease had no effect on the half-life, but there was a 67% decline in albuterol clearance. Caution should be used when administering high doses of albuterol sulfate inhalation aerosol to patients with renal impairment [see Use in Specific Populations (8.5)] .
NONCLINICAL TOXICOLOGY
Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 2-year study in Sprague-Dawley rats, albuterol sulfate caused a dose-related increase in the incidence of benign leiomyomas of the mesovarium at and above dietary doses of 2 mg/kg (approximately 15 times and 6 times the maximum recommended human daily inhalation dose (MRHDID) for adults and children, respectively, on a mg/m 2 basis). In another study this effect was blocked by the coadministration of propranolol, a non-selective beta-adrenergic antagonist. In an 18-month study in CD-1 mice, albuterol sulfate showed no evidence of tumorigenicity at dietary doses of up to 500 mg/kg (approximately 1,900 times and 740 times the MRHDID for adults and children, respectively, on a mg/m 2 basis). In a 22-month study in Golden Hamsters, albuterol sulfate showed no evidence of tumorigenicity at dietary doses of up to 50 mg/kg (approximately 250 times and 100 times the MRHDID for adults and children, respectively, on a mg/m 2 basis).
Albuterol sulfate was not mutagenic in the Ames test or a mutation test in yeast. Albuterol sulfate was not clastogenic in a human peripheral lymphocyte assay or in an AH1 strain mouse micronucleus assay.
Reproduction studies in rats demonstrated no evidence of impaired fertility at oral doses up to 50 mg/kg (approximately 380 times the MRHDID for adults on a mg/m 2 basis).
Animal Toxicology and/or Pharmacology
Preclinical: Intravenous studies in rats with albuterol sulfate have demonstrated that albuterol crosses the blood-brain barrier and reaches brain concentrations amounting to approximately 5% of the plasma concentrations. In structures outside the blood-brain barrier (pineal and pituitary glands), albuterol concentrations were found to be 100 times those in the whole brain.
Studies in laboratory animals (minipigs, rodents, and dogs) have demonstrated the occurrence of cardiac arrhythmias and sudden death (with histologic evidence of myocardial necrosis) when β-agonists and methylxanthines were administered concurrently. The clinical significance of these findings is unknown.
Propellant HFA-134a is devoid of pharmacological activity except at very high doses in animals (380 – 1300 times the maximum human exposure based on comparisons of AUC values), primarily producing ataxia, tremors, dyspnea, or salivation. These are similar to effects produced by the structurally related chlorofluorocarbons (CFCs), which have been used extensively in metered-dose inhalers.
In animals and humans, propellant HFA-134a was found to be rapidly absorbed and rapidly eliminated, with an elimination half-life of 3 - 27 minutes in animals and 5 - 7 minutes in humans. Time to maximum plasma concentration (T max ) and mean residence time are both extremely short leading to a transient appearance of HFA-134a in the blood with no evidence of accumulation.
CLINICAL STUDIES
Bronchospasm Associated with Asthma
Adult and Adolescent Patients 12 Years of Age and Older: In a 6-week, randomized, double-blind, placebo-controlled trial, albuterol sulfate inhalation aerosol (58 patients) was compared to a matched placebo HFA inhalation aerosol (58 patients) in asthmatic patients 12 to 76 years of age at a dose of 180 mcg albuterol four times daily. An evaluator-blind marketed active comparator HFA-134a albuterol inhaler arm (56 patients) was included.
Serial FEV 1 measurements, shown below as percent change from test-day baseline at Day 1 and at Day 43, demonstrated that two inhalations of albuterol sulfate inhalation aerosol produced significantly greater improvement in FEV 1 over the pre-treatment value than the matched placebo, as well as a comparable bronchodilator effect to the marketed active comparator HFA-134a albuterol inhaler.
FEV 1 as Mean Percent Change from Test-Day Pre-Dose in a 6-Week Clinical Trial
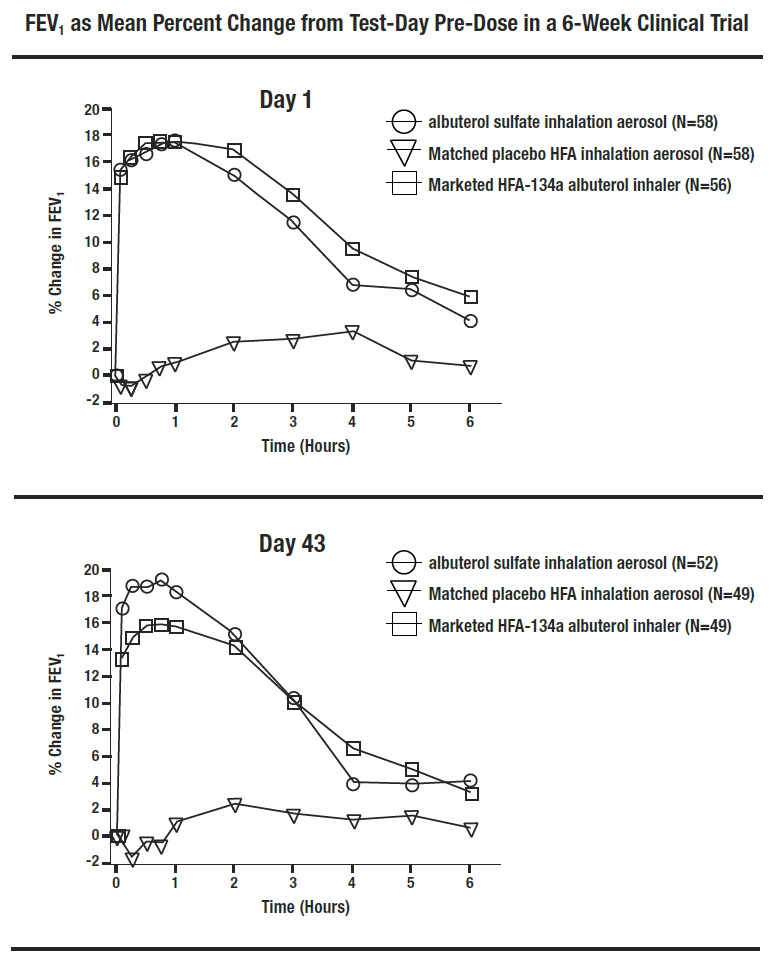
In this study, 31 of 58 patients treated with albuterol sulfate inhalation aerosol achieved a 15% increase in FEV 1 within 30 minutes post-dose on Day 1. In these patients, the median time to onset, median time to peak effect, and median duration of effect were 8.2 minutes, 47 minutes, and approximately 3 hours, respectively. In some patients, the duration of effect was as long as 6 hours.
In a placebo-controlled, single-dose, crossover study, albuterol sulfate inhalation aerosol, administered at albuterol doses of 90, 180 and 270 mcg, produced bronchodilator responses significantly greater than those observed with a matched placebo HFA inhalation aerosol and comparable to a marketed active comparator HFA-134a albuterol inhaler.
Pediatric Patients 4 to 11 Years of Age: In a 3-week, randomized, double-blind, placebo-controlled trial, the same formulation of albuterol as in albuterol sulfate inhalation aerosol (50 patients) was compared to a matched placebo HFA inhalation aerosol (45 patients) in asthmatic children 4 to 11 years of age at a dose of 180 mcg albuterol four times daily. Serial FEV 1 measurements, expressed as the maximum percent change from test-day baseline in percent predicted FEV 1 at Day 1 and at Day 22 observed within two hours post-dose, demonstrated that two inhalations of HFA albuterol sulfate produced significantly greater improvement in FEV 1 over the pre-treatment value than the matched placebo.
In this study, 21 of 50 pediatric patients treated with the same formulation of albuterol as in albuterol sulfate inhalation aerosol achieved a 15% increase in FEV 1 within 30 minutes post-dose on Day 1. In these patients, the median time to onset, median time to peak effect and median duration of effect were 10 minutes, 31 minutes, and approximately 4 hours, respectively. In some pediatric patients, the duration of effect was as long as 6 hours.
In a placebo-controlled, single-dose, crossover study in 55 pediatric patients 4 to 11 years of age, albuterol sulfate inhalation aerosol, administered at albuterol doses of 90 and 180 mcg, was compared with a matched placebo HFA inhalation aerosol. Serial FEV 1 measurements, expressed as the baseline-adjusted percent predicted FEV 1 observed over 6 hours post-dose, demonstrated that one and two inhalations of albuterol sulfate inhalation aerosol produced significantly greater bronchodilator responses than the matched placebo.
Exercise-Induced Bronchospasm
In a randomized, single-dose, crossover study in 24 adults and adolescents with exercise-induced bronchospasm (EIB), two inhalations of albuterol sulfate inhalation aerosol taken 30 minutes before exercise prevented EIB for the hour following exercise (defined as maintenance of FEV 1 within 80% of post-dose, pre-exercise baseline values) in 83% (20 of 24) of patients as compared to 25% (6 of 24) of patients when they received placebo.
Some patients who participated in these clinical trials were using concomitant steroid therapy.
HOW SUPPLIED/STORAGE AND HANDLING
Albuterol Sulfate Inhalation Aerosol is supplied as a pressurized aluminum canister with a white plastic actuator with a dose counter and white dust cap each in boxes of one. Each canister contains 8.5 g of the formulation and provides 200 actuations (NDC 45802- 088 -01). Each actuation delivers 120 mcg of albuterol sulfate from the canister valve and 108 mcg of albuterol sulfate from the actuator mouthpiece (equivalent to 90 mcg of albuterol base).
SHAKE WELL BEFORE USE. Store at 20 to 25°C (68 to 77°F) [see USP Controlled Room Temperature]. Contents under pressure. Do not puncture or incinerate. Protect from freezing temperatures and prolonged exposure to direct sunlight. Exposure to temperatures above 120°F may cause bursting. For best results, canister should be at room temperature before use. Avoid spraying in eyes. Keep out of reach of children.
See FDA-Approved Patient Labeling (17.9) for priming and cleaning instructions.
The white actuator supplied with Albuterol Sulfate Inhalation Aerosol should not be used with the canister from any other inhalation aerosol products. The Albuterol Sulfate Inhalation Aerosol canister should not be used with the actuator from any other inhalation aerosol products.
Albuterol Sulfate Inhalation Aerosol inhaler has a dose counter attached to the actuator. Patients should never try to alter the numbers for the dose counter or tamper with the parts inside the actuator. Discard the Albuterol Sulfate Inhalation Aerosol inhaler when the counter displays 0 or after the expiration date on the product, whichever comes first. The labeled amount of medication in each actuation cannot be assured after the counter displays 0, even though the canister is not completely empty and will continue to operate. Never immerse the canister into water to determine how full the canister is ("float test").
Albuterol Sulfate Inhalation Aerosol does not contain chlorofluorocarbons (CFCs) as the propellant.
Instructions for Use
Albuterol Sulfate (al bue׳ ter ol sul׳ fate) Inhalation Aerosol
Read this Instructions for Use before you start using Albuterol Sulfate Inhalation Aerosol and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
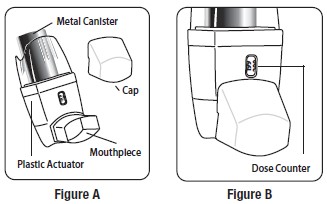
The Parts of Your Albuterol Sulfate Inhalation Aerosol Inhaler Device:
There are 2 main parts of your Albuterol Sulfate Inhalation Aerosol inhaler device including a:
- white plastic actuator that sprays the medicine from the canister. See Figure A.
- white protective dust cap that covers the mouthpiece of the actuator. See Figure A.
There is also a metal canister that holds the medicine. See Figure A.
There is also a dose counter attached to the actuator with a viewing window that shows you how many sprays of medicine you have left. See Figure B.
You will see the number 203 in the viewing window which will count down during each of 3 priming shots. See Figure B and “ Priming Your Albuterol Sulfate Inhalation Aerosol Device ” below.
- Do not use the Albuterol Sulfate Inhalation Aerosol actuator with a canister of medicine from any other inhaler.
- Do not use a Albuterol Sulfate Inhalation Aerosol canister with an actuator from any other inhaler, including another Albuterol Sulfate Inhalation Aerosol inhaler.
Priming Your Albuterol Sulfate Inhalation Aerosol Device:
Your Albuterol Sulfate Inhalation Aerosol device must be primed before you use it for the first time or if your device has not been used for more than 14 days in a row. Do not prime your Albuterol Sulfate Inhalation Aerosol device every day.
- Remove your Albuterol Sulfate Inhalation Aerosol device from its package.
- Remove the protective dust cap from the mouthpiece.
- Shake the inhaler well, and spray it into the air away from your face. See Figure C.
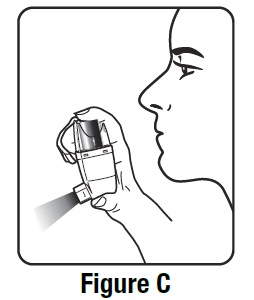
- Shake and spray the inhaler like this 2 more times to finish priming it. The dose counter on the actuator should display the number 200 after you finish priming the inhaler for the first time. See Figure D.

- Priming is necessary to make sure the correct dose of albuterol is contained in each actuation (spray). Your inhaler has 200 sprays for actual use and an extra 3 sprays for priming purposes. When the dose counter reads 203, 202, or 201, spray it into the air away from your face to prime it until the dose counter reads 200. Only begin using the new inhaler after priming, by actuating (spraying) 3 times, and when the dose counter reads 200 in the window.
Each Time You Use Your Albuterol Sulfate Inhalation Aerosol Device:
- Make sure the canister fits firmly in the plastic actuator.
- Look into the mouthpiece to make sure there are no foreign objects there, especially if the cap has not been used to cover the mouthpiece.
Reading the Dose Counter on Your Albuterol Sulfate Inhalation Aerosol Actuator
- The dose counter will count down each time a spray is released. The dose counter window shows the number of sprays left in your inhaler. See Figure D.
- When the dose counter reaches 0 with a solid red background, it will continue to show a large 0 and you should replace your Albuterol Sulfate Inhalation Aerosol device.
- The dose counter cannot be reset and is permanently attached to the actuator. Never change the numbers for the dose counter or touch the parts inside the actuator.
- Do not remove the canister from the plastic actuator except during cleaning, and make sure the cap is on the mouthpiece when reattaching the canister to the plastic actuator. Placing the cap on the mouthpiece before reattaching the canister will prevent accidental release of a dose of Albuterol Sulfate Inhalation Aerosol into the air or increasing of the number of doses on the dose counter. The dose counter will count down each time a spray is released.
Using Your Albuterol Sulfate Inhalation Aerosol Device:
Step 1. Shake the inhaler well before each spray. Take the cap off the mouthpiece of the actuator.
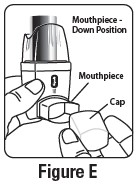
Step 2. Hold the inhaler with the mouthpiece down. See Figure E.
Step 3. Breathe out through your mouth and push as much air from your lungs as you can. Put the mouthpiece in your mouth and close your lips around it. See Figure F.
Step 4. Push the top of the canister all the way down while you breathe in deeply and slowly through your mouth. See Figure F.
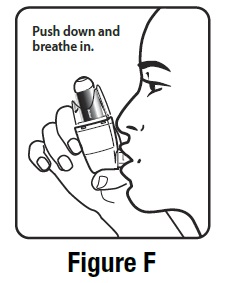
Step 5. Right after the spray comes out, take your finger off the canister. After you have breathed in all the way, take the inhaler out of your mouth and close your mouth.
Step 6. Hold your breath as long as you can , up to 10 seconds, then breathe normally. If your doctor has told you to use more sprays , wait 1 minute and shake the inhaler again. Repeat Steps 2 through Step 6.
Step 7. Put the cap back on the mouthpiece after every time you use the inhaler. Make sure the cap snaps firmly into place.
Cleaning Your Albuterol Sulfate Inhalation Aerosol Device:
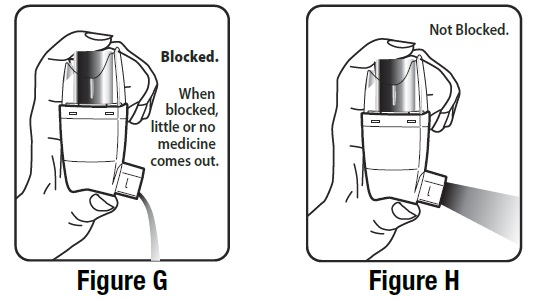
It is very important to keep the plastic actuator clean so the medicine will not build-up and block the spray. See Figure G and Figure H.
- Do not try to clean the metal canister or let it get wet . The inhaler may stop spraying if it is not cleaned correctly.
- If you have more than 1 Albuterol Sulfate Inhalation Aerosol inhaler, wash each device at separate times to prevent putting the wrong canister together with the wrong plastic actuator. This way you can be sure you will always know the correct number of remaining doses of Albuterol Sulfate Inhalation Aerosol.
- Wash the actuator at least 1 time each week as follows:
- Take the canister out of the actuator, and take the cap off the mouthpiece.
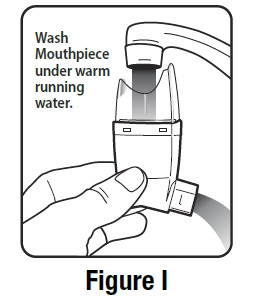
- Hold the actuator under the faucet and run warm water through it for about 30 seconds. See Figure I.
- Turn the actuator upside down and run warm water through the mouthpiece for about 30 seconds. See Figure J.
- Shake off as much water from the actuator as you can. Look into the mouthpiece to make sure any medicine build-up has been completely washed away. If there is any build-up, repeat the washing instructions.
- Let the actuator air-dry completely, such as overnight. See Figure K.
- When the actuator is dry, put the cap on the mouthpiece and then put the canister in the actuator and make sure it fits firmly. Remove the cap from the mouthpiece, shake the inhaler well and spray it twice into the air away from your face. Put the cap back on the mouthpiece.
If you need to use your inhaler before the actuator is completely dry:
- Shake as much water off the actuator as you can.
- Place the cap on the mouthpiece and then put the canister in the actuator and make sure it fits firmly.
- Remove the cap from the mouthpiece, shake the inhaler well and spray it twice into the air away from your face.
- Take your Albuterol Sulfate Inhalation Aerosol dose as prescribed.
- Follow the Cleaning Instructions above.
Replacing Your Albuterol Sulfate Inhalation Aerosol Device
- When the dose counter on the actuator says the number 20 , contact your pharmacy to refill your prescription or ask your doctor for another prescription for Albuterol Sulfate Inhalation Aerosol. When it is time to obtain a new inhaler a red flag will begin to show in the window, getting larger as the number of doses remaining approaches 0. When the dose counter reaches 0, the background color will change to solid red. The red flag is to remind you to refill your prescription or ask your doctor for another prescription.
- Throw the Albuterol Sulfate Inhalation Aerosol inhaler away as soon as the dose counter says 0 with a solid red background or after the expiration date on the Albuterol Sulfate Inhalation Aerosol packaging, whichever comes first. You should not keep using the inhaler after 200 sprays even though the canister may not be completely empty. You cannot be sure you will receive any medicine after using 200 sprays.
- Do not use the inhaler after the expiration date on the Albuterol Sulfate Inhalation Aerosol packaging.
This Patient Information and Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured for Padagis ®
Minneapolis, MN 55427
www.padagis.com
Rev 09-24
Mechanism of Action
Albuterol sulfate is a beta 2 -adrenergic agonist. The pharmacologic effects of albuterol sulfate are attributable to activation of beta 2 -adrenergic receptors on airway smooth muscle. Activation of beta 2 -adrenergic receptors leads to the activation of adenylcyclase and to an increase in the intracellular concentration of cyclic-3', 5'-adenosine monophosphate (cyclic AMP). This increase of cyclic AMP is associated with the activation of protein kinase A, which in turn inhibits the phosphorylation of myosin and lowers intracellular ionic calcium concentrations, resulting in muscle relaxation. Albuterol relaxes the smooth muscle of all airways, from the trachea to the terminal bronchioles. Albuterol acts as a functional antagonist to relax the airway irrespective of the spasmogen involved, thus protecting against all bronchoconstrictor challenges. Increased cyclic AMP concentrations are also associated with the inhibition of release of mediators from mast cells in the airway. While it is recognized that beta 2 -adrenergic receptors are the predominant receptors on bronchial smooth muscle, data indicate that there are beta-receptors in the human heart, 10% to 50% of which are cardiac beta 2 -adrenergic receptors. The precise function of these receptors has not been established [see Warnings and Precautions (5.4)] .
Albuterol has been shown in most controlled clinical trials to have more effect on the respiratory tract, in the form of bronchial smooth muscle relaxation, than isoproterenol at comparable doses while producing fewer cardiovascular effects. However, inhaled albuterol, like other beta-adrenergic agonist drugs, can produce a significant cardiovascular effect in some patients, as measured by pulse rate, blood pressure, symptoms, and/or electrocardiographic changes [see Warnings and Precautions (5.4)] .