Get your patient on Ala - Scalp - Hydrocortisone lotion (Hydrocortisone)
Ala - Scalp - Hydrocortisone lotion prescribing information
INDICATIONS AND USAGE
Hydrocortisone Lotion is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
DOSAGE AND ADMINISTRATION
Topical corticosteroids are generally applied to the affected area as a thin film from two to four times daily depending on the severity of the condition.
Occlusive dressings may be used for the management of psoriasis or recalcitrant conditions. If an infection develops, the use of occlusive dressings should be discontinued and appropriate antimicrobial therapy instituted.
CONTRAINDICATIONS
Hydrocortisone Lotion is contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
Rx Only
For external use only
Not for opthalmic use
ADVERSE REACTIONS
The following local adverse reactions are reported infrequently with topical corticosteroids, but may occur more frequently with the use of occlusive dressings. These reactions are listed in an aproximate decreasing order of occurrence: burning, itching, irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation, perioral dermitis, allergic contact dermatis, maceration of the skin, secondary infection, skin atrophy, striae, and miliaria.
To report SUSPECTED ADVERSE REACTIONS, contact Derm Ventures, LLC at 1-833-972-1100 or FDA at 1-800-FDA-1088 or https://fda.gov/Safety/MedWatch/
DESCRIPTION
Topical corticosteroids constitute a class of primarily synthetic steroids used a anti-inflammatory and antipruritic agents. Hydrocortisone is a member of this class. Chemically hydrocortisone is pregn-4-ene-3, 20-dione, 11, 17, 21-trihydroxy, (11Β)-. Its structural formula is:
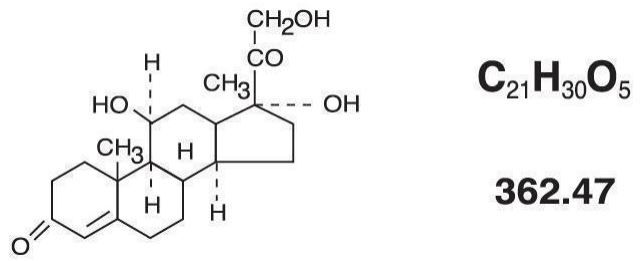
Each mL of ALA-SCALP (Hydrocortisone Lotion USP), 2% contains 20 mg of hydrocortisone USP in a vehicle of isopropyl alcohol, polysorbate 20, purified water, propylene glycol, and benzalkonium chloride.
CLINICAL PHARMACOLOGY
Topical corticosteroids share anti-inflammatory, antipruritic and vasoconstrictive actions. The mechanism of anti-inflammatory activity of the topical corticosteroids is unclear. Various laboratory methods, including vasoconstrictor assays, are used to compare and predict potencies and/or clinical efficacies of the topical corticosteroids. There is some evidence to suggest that a recognizable correlation exists between vasoconstrictor potency and therapeutic efficacy in man.
Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle, the integrity of the epidermal barrier, and the use of occlusive dressings. Topical corticosteroids can be absorbed from normal intact skin. Inflammation and/or other disease processes in the skin increase percutaneous absorption. Occlusive dressings may be a valuable therapeutic adjunct for treatment of resistant dermatoses. (See DOSAGE AND ADMINISTRATION ).
Once absorbed through the skin, topical corticosteroids are handled through pharmacokinetic pathways similar to systemically administered corticosteroids. Corticosteroids are bound to plasma proteins in varying degrees. Corticosteroids are metabolized primaily in the liver and are then excreted by the kidneys. Some of the topical corticosteroids and their matabolites are also excreted into the bile.
HOW SUPPLIED
ALA-SCALP (Hydrocortisone Lotion USP) 2% is supplied in:
2 fl oz (59.2 mL) NDC 77052-010-02