Get your patient on Acyclovir - Acyclovir injection, Powder, Lyophilized, For Solution (Acyclovir)
Acyclovir - Acyclovir injection, Powder, Lyophilized, For Solution prescribing information
INDICATIONS AND USAGE
Herpes Simplex Infections in Immunocompromised Patients
Acyclovir for Injection is indicated for the treatment of initial and recurrent mucosal and cutaneous herpes simplex (HSV-1 and HSV-2) in immunocompromised patients.
Initial Episodes of Herpes Genitalis
Acyclovir for Injection is indicated for the treatment of severe initial clinical episodes of herpes genitalis in immunocompetent patients.
Herpes Simplex Encephalitis
Acyclovir for Injection is indicated for the treatment of herpes simplex encephalitis.
Neonatal Herpes Simplex Virus Infection
Acyclovir for Injection is indicated for the treatment of neonatal herpes infections.
Varicella-Zoster Infections in Immunocompromised Patients
Acyclovir for Injection is indicated for the treatment of varicella-zoster (shingles) infections in immunocompromised patients.
DOSAGE AND ADMINISTRATION
CAUTION—RAPID OR BOLUS INTRAVENOUS INJECTION MUST BE AVOIDED (see WARNINGS and PRECAUTIONS ).
INTRAMUSCULAR OR SUBCUTANEOUS INJECTION MUST BE AVOIDED (see WARNINGS ).
Therapy should be initiated as early as possible following onset of signs and symptoms of herpes infections.
A maximum dose equivalent to 20 mg/kg every 8 hours should not be exceeded for any patient.
Dosage
Herpes Simplex Infections
Mucosal and Cutaneous Herpes Simplex (HSV-1 and HSV-2) Infections in Immunocompromised Patients
Adults and Adolescents (12 years of age and older): 5 mg/kg infused at a constant rate over 1 hour, every 8 hours for 7 days.
Pediatrics (Under 12 years of age): 10 mg/kg infused at a constant rate over 1 hour, every 8 hours for 7 days.
Severe Initial Clinical Episodes of Herpes Genitalis
Adults and Adolescents (12 years of age and older): 5 mg/kg infused at a constant rate over 1 hour, every 8 hours for 5 days.
Herpes Simplex Encephalitis
Adults and Adolescents (12 years of age and older) : 10 mg/kg infused at a constant rate over 1 hour, every 8 hours for 10 days.
Pediatrics (3 months to 12 years of age): 20 mg/kg infused at a constant rate over 1 hour, every 8 hours for 10 days .
Neonatal Herpes Simplex Virus Infections (Birth to 3 months)
10 mg/kg infused at a constant rate over 1 hour, every 8 hours for 10 days. In neonatal herpes simplex infections, doses of 15 mg/kg or 20 mg/kg (infused at a constant rate over 1 hour every 8 hours) have been used; the safety and efficacy of these doses are not known.
Varicella Zoster Infections
Zoster in Immunocompromised Patients
Adults and Adolescents (12 years of age and older): 10 mg/kg infused at a constant rate over 1 hour, every 8 hours for 7 days.
Pediatrics (Under 12 years of age): 20 mg/kg infused at a constant rate over 1 hour, every 8 hours for 7 days.
Obese Patients: Obese patients should be dosed at the recommended adult dose using Ideal Body Weight.
Patients with Acute or Chronic Renal Impairment
Refer to DOSAGE AND ADMINISTRATION for recommended doses, and adjust the dosing interval as indicated in Table 5.
| Creatinine Clearance (mL/min/1.73 m 2 ) | Percent of Recommended Dose | Dosing Interval (hours) |
| >50 | 100% | 8 |
| 25 - 50 | 100% | 12 |
| 10 - 25 | 100% | 24 |
| 0 - 10 | 50% | 24 |
Hemodialysis
For patients who require dialysis, the mean plasma half-life of acyclovir during hemodialysis is approximately 5 hours. This results in a 60% decrease in plasma concentrations following a 6-hour dialysis period. Therefore, the patient’s dosing schedule should be adjusted so that an additional dose is administered after each dialysis.
Peritoneal Dialysis
No supplemental dose appears to be necessary after adjustment of the dosing interval.
Method of Preparation
Each 10 mL vial contains acyclovir sodium equivalent to 500 mg of acyclovir. Each 20 mL vial contains acyclovir sodium equivalent to 1,000 mg of acyclovir. The contents of the vial should be dissolved in Sterile Water for Injection as follows:
| Contents of Vial | Amount of Diluent |
| 500 mg | 10 mL |
| 1,000 mg | 20 mL |
The resulting solution in each case contains 50 mg acyclovir per mL (pH approximately 11). Shake the vial well to assure complete dissolution before measuring and transferring each individual dose. The reconstituted solution should be used within 12 hours. Refrigeration of reconstituted solution may result in the formation of a precipitate which will redissolve at room temperature.
DO NOT USE BACTERIOSTATIC WATER FOR INJECTION CONTAINING BENZYL ALCOHOL OR PARABENS.
Administration
The calculated dose should then be removed and added to any appropriate intravenous solution at a volume selected for administration during each 1 hour infusion. Infusion concentrations of approximately 7 mg/mL or lower are recommended. In clinical studies, the average 70 kg adult received between 60 and 150 mL of fluid per dose. Higher concentrations (e.g., 10 mg/mL) may produce phlebitis or inflammation at the injection site upon inadvertent extravasation. Standard, commercially available electrolyte and glucose solutions are suitable for intravenous administration; biologic or colloidal fluids (e.g., blood products, protein solutions, etc.) are not recommended.
Once diluted for administration, each dose should be used within 24 hours.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
CONTRAINDICATIONS
Acyclovir for Injection is contraindicated for patients who develop hypersensitivity to acyclovir or valacyclovir.
ADVERSE REACTIONS
The adverse reactions listed below have been observed in controlled and uncontrolled clinical trials in approximately 700 patients who received acyclovir at approximately 5 mg/kg (250 mg/m 2 ) three times daily, and approximately 300 patients who received approximately 10 mg/kg (500 mg/m 2 ) three times daily.
The most frequent adverse reactions reported during administration of acyclovir were inflammation or phlebitis at the injection site in approximately 9% of the patients, and transient elevations of serum creatinine or BUN in 5% to 10% (the higher incidence occurred usually following rapid [less than 10 minutes] intravenous infusion). Nausea and/or vomiting occurred in approximately 7% of the patients (the majority occurring in nonhospitalized patients who received 10 mg/kg). Itching, rash, or hives occurred in approximately 2% of patients. Elevation of transaminases occurred in 1% to 2% of patients.
The following hematologic abnormalities occurred at a frequency of less than 1%: anemia, neutropenia, thrombocytopenia, thrombocytosis, leukocytosis, and neutrophilia. In addition, anorexia and hematuria were observed.
Observed During Clinical Practice
In addition to adverse events reported from clinical trials, the following events have been identified during post-approval use of Acyclovir for Injection in clinical practice. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to either their seriousness, frequency of reporting, potential causal connection to acyclovir, or a combination of these factors.
General: Anaphylaxis, angioedema, fatigue, fever, headache, pain, peripheral edema.
Digestive: Abdominal pain, diarrhea, gastrointestinal distress, nausea.
Cardiovascular: Hypotension.
Hematologic and Lymphatic: Disseminated intravascular coagulation, hemolysis, leukocytoclastic vasculitis, leukopenia, lymphadenopathy.
Hepatobiliary Tract and Pancreas : Elevated liver function tests, hepatitis, hyperbilirubinemia, jaundice.
Musculoskeletal: Myalgia.
Nervous: Aggressive behavior, agitation, ataxia, coma, confusion, delirium, dizziness, dysarthria, encephalopathy, hallucinations, obtundation, paresthesia, psychosis, seizure, somnolence, tremor. These symptoms may be marked, particularly in older adults (see PRECAUTIONS ).
Skin: Alopecia, erythema multiforme, photosensitive rash, pruritus, rash, Stevens-Johnson syndrome, toxic epidermal necrolysis, urticaria. Severe local inflammatory reactions, including tissue necrosis, have occurred following infusion of acyclovir into extravascular tissues.
Special Senses: Visual abnormalities.
Urogenital: Renal failure, elevated blood urea nitrogen, elevated creatinine (see WARNINGS ).
To report SUSPECTED ADVERSE REACTIONS, contact Hikma Pharmaceuticals USA Inc. at 1-877-845-0689 or the FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Coadministration of probenecid with acyclovir has been shown to increase the mean acyclovir half-life and the area under the concentration-time curve. Urinary excretion and renal clearance were correspondingly reduced.
DESCRIPTION
Acyclovir is a synthetic nucleoside analog active against herpes viruses. Acyclovir for Injection, USP is a sterile lyophilized powder for intravenous administration only. Each 500 mg vial contains 500 mg of acyclovir and 49 mg of sodium, and each 1,000 mg vial contains 1,000 mg acyclovir and 98 mg of sodium. Reconstitution of the 500 mg or 1,000 mg vials with 10 mL or 20 mL, respectively, of Sterile Water for Injection, USP results in a solution containing 50 mg/mL of acyclovir. The pH of the reconstituted solution is approximately 11. Further dilution in any appropriate intravenous solution must be performed before infusion (see DOSAGE AND ADMINISTRATION: Method of Preparation and Administration ).
Acyclovir sodium is a white, crystalline powder with the molecular formula C 8 H 10 N 5 NaO 3 and a molecular weight of 247.19. The maximum solubility in water at 25°C exceeds 100 mg/mL. At physiologic pH, acyclovir sodium exists as the un-ionized form with a molecular weight of 225 and a maximum solubility in water at 37°C of 2.5 mg/mL. The pkas of acyclovir are 2.27 and 9.25.
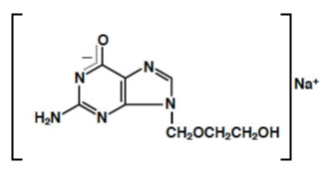
The chemical name of acyclovir sodium is 2-amino-1,9-dihydro-9-[(2-hydroxyethoxy)methyl]-6 H -purin-6-one monosodium salt; it has the following structural formula:
CLINICAL PHARMACOLOGY
Pharmacokinetics
The pharmacokinetics of acyclovir after intravenous administration have been evaluated in adult patients with normal renal function during Phase 1/2 studies after single doses ranging from 0.5 to 15 mg/kg and after multiple doses ranging from 2.5 to 15 mg/kg every 8 hours. Proportionality between dose and plasma levels is seen after single doses or at steady-state after multiple dosing. Average steady-state peak and trough concentrations from 1-hour infusions administered every 8 hours are given in Table 1.
| Dosage Regimen | C SS max | C SS trough |
| 5 mg/kg q 8 hr (n = 8) | 9.8 mcg/mL range: 5.5 to 13.8 | 0.7 mcg/mL range: 0.2 to 1 |
10 mg/kg q 8 hr (n = 7) | 22.9 mcg/mL range: 14.1 to 44.1 | 1.9 mcg/mL range: 0.5 to 2.9 |
Concentrations achieved in the cerebrospinal fluid are approximately 50% of plasma values. Plasma protein binding is relatively low (9% to 33%) and drug interactions involving binding site displacement are not anticipated.
Renal excretion of unchanged drug is the major route of acyclovir elimination accounting for 62% to 91% of the dose. The only major urinary metabolite detected is 9-carboxymethoxymethylguanine accounting for up to 14.1% of the dose in patients with normal renal function.
The half-life and total body clearance of acyclovir are dependent on renal function as shown in Table 2.
| Creatinine Clearance (mL/min/1.73 m 2 ) | Half-life (hr) | Total Body Clearance | |
| (mL/min/1.73 m 2 ) | (mL/min/kg) | ||
| >80 | 2.5 | 327 | 5.1 |
| 50 - 80 | 3 | 248 | 3.9 |
| 15 - 50 | 3.5 | 190 | 3.4 |
| 0 (Anuric) | 19.5 | 29 | 0.5 |
Special Populations
Adults with Impaired Renal Function
Acyclovir was administered at a dose of 2.5 mg/kg to 6 adult patients with severe renal failure. The peak and trough plasma levels during the 47 hours preceding hemodialysis were 8.5 mcg/mL and 0.7 mcg/mL, respectively.
Consult DOSAGE AND ADMINISTRATION for recommended adjustments in dosing based upon creatinine clearance.
Pediatrics
Acyclovir pharmacokinetics were determined in 16 pediatric patients with normal renal function ranging in age from 3 months to 16 years at doses of approximately 10 mg/kg and 20 mg/kg every 8 hours (Table 3). Concentrations achieved at these regimens are similar to those in adults receiving 5 mg/kg and 10 mg/kg every 8 hours, respectively (Table 1). Acyclovir pharmacokinetics were determined in 12 patients ranging in age from birth to 3 months at doses of 5 mg/kg, 10 mg/kg, and 15 mg/kg every 8 hours (Table 3).
| Parameter | Birth to 3 Months of Age (n = 12) | 3 Months to 12 Years of Age (n = 16) |
| CL (mL/min/kg) | 4.46 ± 1.61 | 8.44 ± 2.92 |
| VDSS (L/kg) | 1.08 ± 0.35 | 1.01 ± 0.28 |
| Elimination half-life (hr) | 3.80 ± 1.19 | 2.36 ± 0.97 |
Geriatrics
Acyclovir plasma concentrations are higher in geriatric patients compared to younger adults, in part due to age-related changes in renal function. Dosage reduction may be required in geriatric patients with underlying renal impairment (see PRECAUTIONS: Geriatric Use ).
Drug Interactions
Coadministration of probenecid with acyclovir has been shown to increase the mean acyclovir half-life and the area under the concentration-time curve. Urinary excretion and renal clearance were correspondingly reduced.
Clinical Trials
Herpes Simplex Infections in Immunocompromised Patients
A multicenter trial of acyclovir at a dose of 250 mg/m 2 every 8 hours (750 mg/m 2 /day) for 7 days was conducted in 98 immunocompromised patients (73 adults and 25 children) with orofacial, esophageal, genital, and other localized infections (52 treated with acyclovir and 46 with placebo). Acyclovir decreased virus excretion, reduced pain, and promoted healing of lesions.
Initial Episodes of Herpes Genitalis
In placebo-controlled trials, 58 patients with initial genital herpes were treated with intravenous acyclovir 5 mg/kg or placebo (27 patients treated with acyclovir and 31 treated with placebo) every 8 hours for 5 days. Acyclovir decreased the duration of viral excretion, new lesion formation, duration of vesicles, and promoted healing of lesions.
Herpes Simplex Encephalitis
Sixty-two patients ages 6 months to 79 years with brain biopsy-proven herpes simplex encephalitis were randomized to receive either acyclovir (10 mg/kg every 8 hours) or vidarabine (15 mg/kg/day) for 10 days (28 were treated with acyclovir and 34 with vidarabine). Overall mortality at 12 months for patients treated with acyclovir was 25% compared to 59% for patients treated with vidarabine. The proportion of patients treated with acyclovir functioning normally or with only mild sequelae (e.g., decreased attention span) was 32% compared to 12% of patients treated with vidarabine.
Patients less than 30 years of age and those who had the least severe neurologic involvement at time of entry into study had the best outcome with treatment with acyclovir. An additional controlled study performed in Europe demonstrated similar findings.
Neonatal Herpes Simplex Virus Infection
Two hundred and two infants with neonatal herpes simplex infections were randomized to receive either acyclovir 10 mg/kg every 8 hours (n = 107) or vidarabine 30 mg/kg/day (n = 95) for 10 days. Outcomes are presented in Table 4.
| HSV Disease Classification | Treatment Group | |
Acyclovir (n=107) | Vidarabine (n=95) | |
| SEM SEM refers to localized infection with disease limited to skin, eye, and/or mouth. (n = 85) | 0/54 | 0/31 |
| CNS CNS refers to infection of the central nervous system with compatible neurologic and CSF findings. (n = 71) | 5/35 | 5/36 |
| DISS DISS refers to visceral organ involvement such as hepatitis or pneumonitis with or without CNS involvement. (n = 46) | 11/18 | 14/28 |
Rates of neurologic sequelae at 1 year were comparable between the treatment groups.
Varicella-Zoster Infections in Immunocompromised Patients
A multicenter trial of Acyclovir for Injection at a dose of 500 mg/m 2 every 8 hours for 7 days was conducted in immunocompromised patients with zoster infections (shingles). Ninety-four (94) patients were evaluated (52 patients were treated with acyclovir and 42 with placebo). Acyclovir was superior to placebo as measured by reductions in cutaneous dissemination and visceral dissemination.
Clinical Trials
Herpes Simplex Infections in Immunocompromised Patients
A multicenter trial of acyclovir at a dose of 250 mg/m 2 every 8 hours (750 mg/m 2 /day) for 7 days was conducted in 98 immunocompromised patients (73 adults and 25 children) with orofacial, esophageal, genital, and other localized infections (52 treated with acyclovir and 46 with placebo). Acyclovir decreased virus excretion, reduced pain, and promoted healing of lesions.
Initial Episodes of Herpes Genitalis
In placebo-controlled trials, 58 patients with initial genital herpes were treated with intravenous acyclovir 5 mg/kg or placebo (27 patients treated with acyclovir and 31 treated with placebo) every 8 hours for 5 days. Acyclovir decreased the duration of viral excretion, new lesion formation, duration of vesicles, and promoted healing of lesions.
Herpes Simplex Encephalitis
Sixty-two patients ages 6 months to 79 years with brain biopsy-proven herpes simplex encephalitis were randomized to receive either acyclovir (10 mg/kg every 8 hours) or vidarabine (15 mg/kg/day) for 10 days (28 were treated with acyclovir and 34 with vidarabine). Overall mortality at 12 months for patients treated with acyclovir was 25% compared to 59% for patients treated with vidarabine. The proportion of patients treated with acyclovir functioning normally or with only mild sequelae (e.g., decreased attention span) was 32% compared to 12% of patients treated with vidarabine.
Patients less than 30 years of age and those who had the least severe neurologic involvement at time of entry into study had the best outcome with treatment with acyclovir. An additional controlled study performed in Europe demonstrated similar findings.
Neonatal Herpes Simplex Virus Infection
Two hundred and two infants with neonatal herpes simplex infections were randomized to receive either acyclovir 10 mg/kg every 8 hours (n = 107) or vidarabine 30 mg/kg/day (n = 95) for 10 days. Outcomes are presented in Table 4.
| HSV Disease Classification | Treatment Group | |
Acyclovir (n=107) | Vidarabine (n=95) | |
| SEM SEM refers to localized infection with disease limited to skin, eye, and/or mouth. (n = 85) | 0/54 | 0/31 |
| CNS CNS refers to infection of the central nervous system with compatible neurologic and CSF findings. (n = 71) | 5/35 | 5/36 |
| DISS DISS refers to visceral organ involvement such as hepatitis or pneumonitis with or without CNS involvement. (n = 46) | 11/18 | 14/28 |
Rates of neurologic sequelae at 1 year were comparable between the treatment groups.
Varicella-Zoster Infections in Immunocompromised Patients
A multicenter trial of Acyclovir for Injection at a dose of 500 mg/m 2 every 8 hours for 7 days was conducted in immunocompromised patients with zoster infections (shingles). Ninety-four (94) patients were evaluated (52 patients were treated with acyclovir and 42 with placebo). Acyclovir was superior to placebo as measured by reductions in cutaneous dissemination and visceral dissemination.
HOW SUPPLIED
Acyclovir for Injection, USP is available as sterile vials, each containing:
| NDC | Packaged | |
| Acyclovir Sodium equivalent to 500 mg of Acyclovir | 0143-9657-10 | Box of 10 vials |
| Acyclovir Sodium equivalent to 1 g of Acyclovir | 0143-9658-10 | Box of 10 vials |
Storage
Store at 15° to 25°C (59° to 77°F).
Manufactured by : HIKMA FARMACÊUTICA (PORTUGAL), S.A. Estrada do Rio da Mó, nº 8, 8A e 8B - Fervença, 2705 – 906 Terrugem SNT PORTUGAL
Distributed by: Hikma Pharmaceuticals USA Inc. Berkeley Heights, NJ 07922
Revised:February 2021 PIN428-WES/2