Get your patient on Acyclovir - Acyclovir tablet (Acyclovir)
Acyclovir - Acyclovir tablet prescribing information
INDICATIONS AND USAGE
Herpes Zoster Infections:
Acyclovir is indicated for the acute treatment of herpes zoster (shingles).
Genital Herpes:
Acyclovir is indicated for the treatment of initial episodes and the management of recurrent episodes of genital herpes.
Chickenpox:
Acyclovir is indicated for the treatment of chickenpox (varicella).
DOSAGE AND ADMINISTRATION
Acute Treatment of Herpes Zoster:
800mg every 4 hours orally, 5 times daily for 7 to 10 days.
Genital Herpes:
200mg every 4 hours, 5 times daily for 10 days.
400mg 2 times daily for up to 12 months, followed by re-evaluation. Alternative regimens have included doses ranging from 200mg 3 times daily to 200mg 5 times daily.
The frequency and severity of episodes of untreated genital herpes may change over time. After 1 year of therapy, the frequency and severity of the patient's genital herpes infection should be re-evaluated to assess the need for continuation of therapy with acyclovir.
200mg every 4 hours, 5 times daily for 5 days. Therapy should be initiated at the earliest sign or symptom (prodrome) of recurrence.
Treatment of Chickenpox:
20mg/kg per dose orally 4 times daily (80mg/kg/day) for 5 days. Children over 40kg should receive the adult dose for chickenpox.
800mg 4 times daily for 5 days.
Intravenous acyclovir is indicated for the treatment of varicella-zoster infections in immunocompromised patients.
When therapy is indicated, it should be initiated at the earliest sign or symptom of chickenpox. There is no information about the efficacy of therapy initiated more than 24 hours after onset of signs and symptoms.
Patients With Acute or Chronic Renal Impairment:
In patients with renal impairment, the dose of acyclovir Tablets should be modified as shown in Table 3.
Table 3. Dosage Modification for Renal Impairment
| Normal Dosage Regimen | Creatinine Clearance (mL/min/1.73 m 2 ) | Adjusted Dosage Regimen | |
| Dose (mg) | Dosing Interval | ||
| 200 mg every 4 hours | >10 0-10 | 200 200 | every 4 hours, 5x daily every 12 hours |
| 400 mg every 12 hours | >10 0-10 | 400 200 | every 12 hours every 12 hours |
| 800 mg every 4 hours | >25 10-25 0-10 | 800 800 800 | every 4 hours, 5x daily every 8 hours every 12 hours |
Hemodialysis:
For patients who require hemodialysis, the mean plasma half-life of acyclovir during hemodialysis is approximately 5 hours. This results in a 60 % decrease in plasma concentrations following a 6-hour dialysis period. Therefore, the patient's dosing schedule should be adjusted so that an additional dose is administered after each dialysis.
Peritoneal Dialysis:
No supplemental dose appears to be necessary after adjustment of the dosing interval.
CONTRAINDICATIONS
ADVERSE REACTIONS
Herpes Simplex:
The most frequent adverse events reported during clinical trials of treatment of genital herpes with acyclovir 200mg administered orally 5 times daily every 4 hours for 10 days were nausea and/or vomiting in 8 of 298 patient treatments (2.7%). Nausea and/or vomiting occurred in 2 of 287 (0.7%) patients who received placebo.
The most frequent adverse events reported in a clinical trial for the prevention of recurrences with continuous administration of 400mg (two 200-mg capsules) 2 times daily for 1 year in 586 patients treated with acyclovir were nausea (4.8 %) and diarrhea (2.4 %).The 589 control patients receiving intermittent treatment of recurrences with acyclovir for 1 year reported diarrhea (2.7%), nausea (2.4 %), and headache (2.2%).
Herpes Zoster:
The most frequent adverse event reported during 3 clinical trials of treatment of herpes zoster (shingles) with 800mg of oral acyclovir 5 times daily for 7 to 10 days in 323 patients was malaise (11.5%). The 323 placebo recipients reported malaise (11.1%).
Chickenpox:
The most frequent adverse event reported during 3 clinical trials of treatment of chickenpox with oral acyclovir at doses of 10 to 20mg/kg 4 times daily for 5 to 7 days or 800mg 4 times daily for 5 days in 495 patients was diarrhea (3.2%). The 498 patients receiving placebo reported diarrhea (2.2%).
Observed During Clinical Practice:
In addition to adverse events reported from clinical trials, the following events have been identified during post-approval use of acyclovir. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made. These events have been chosen for inclusion due to either their seriousness, frequency of reporting, potential causal connection to acyclovir, or a combination of these factors.
Anaphylaxis, angioedema, fever, headache, pain, peripheral edema.
Aggressive behavior, agitation, ataxia, coma, confusion, decreased consciousness, delirium, dizziness, dysarthria, encephalopathy, hallucinations, paresthesia, psychosis, seizure, somnolence, tremors. These symptoms may be marked, particularly in older adults or in patients with renal impairment (see PRECAUTIONS ).
Diarrhea, gastrointestinal distress, nausea.
Anemia, leukocytoclastic vasculitis, leukopenia, lymphadenopathy, thrombocytopenia.
Elevated liver function tests, hepatitis, hyperbilirubinemia, jaundice.
Myalgia.
Alopecia, erythema multiforme, photosensitive rash, pruritus, rash, Stevens-Johnson syndrome, toxic epidermal necrolysis, urticaria.
Visual abnormalities.
Renal failure, renal pain (may be associated with renal failure), elevated blood urea nitrogen, elevated creatinine, hematuria (see WARNINGS ).
Drug Interactions:
Coadministration of probenecid with intravenous acyclovir has been shown to increase the mean acyclovir half-life and the area under the concentration-time curve. Urinary excretion and renal clearance were correspondingly reduced.
DESCRIPTION
Acyclovir is a synthetic nucleoside analogue active against herpesviruses. Acyclovir Tablets are formulations for oral administration.
Each 800-mg tablet of acyclovir contains 800mg of acyclovir and the inactive ingredients FD&C Blue No. 2, magnesium stearate, microcrystalline cellulose, povidone, and sodium starch glycolate.
Each 400-mg tablet of acyclovir contains 400mg of acyclovir and the inactive ingredients magnesium stearate, microcrystalline cellulose, povidone and sodium starch glycolate.
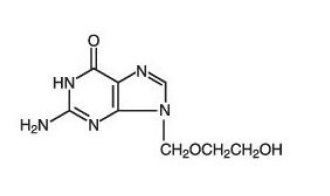
Acyclovir is a white, crystalline powder with the molecular formula C8H11N5O3 and a molecular weight of 225. The maximum solubility in water at 37°C is 2.5mg/mL. The pka's of acyclovir are 2.27 and 9.25.
The chemical name of acyclovir is 2-amino -1, 9 -dihydro -9 - [(2-hydroxyethoxy) methyl]-6 H -purin-6 -one; it has the following structural formula:
CLINICAL PHARMACOLOGY
Pharmacokinetics:
The pharmacokinetics of acyclovir after oral administration have been evaluated in healthy volunteers and in immunocompromised patients with herpes simplex or varicella-zoster virus infection. Acyclovir pharmacokinetic parameters are summarized in Table 1.
| Parameter | Range |
| Plasma protein binding | 9 % to 33% |
| Plasma elimination half-life | 2.5 to 3.3 hr |
| Average oral bio availability | 10 % to 20 % • |
| • Bio availability decreases with increasing dose. | |