Get your patient on Acetazolamide - Acetazolamide capsule, Extended Release (Acetazolamide)
Acetazolamide - Acetazolamide capsule, Extended Release prescribing information
INDICATIONS AND USAGE:
For adjunctive treatment of: chronic simple (open-angle) glaucoma, secondary glaucoma, and preoperatively in acute angle-closure glaucoma where delay of surgery is desired in order to lower intraocular pressure. Acetazolamide extended-release capsules are also indicated for the prevention or amelioration of symptoms associated with acute mountain sickness despite gradual ascent.
DOSAGE AND ADMINISTRATION:
Glaucoma
The recommended dosage is 1 capsule (500 mg) two times a day. Usually 1 capsule is administered in the morning and 1 capsule in the evening. It may be necessary to adjust the dose, but it has usually been found that dosage in excess of 2 capsules (1 g) does not produce an increased effect. The dosage should be adjusted with careful individual attention both to symptomatology and intraocular tension. In all cases, continuous supervision by a physician is advisable.
In those unusual instances where adequate control is not obtained by the twice-a-day administration of acetazolamide extended-release capsules, the desired control may be established by means of acetazolamide (tablets or parenteral). Use tablets or parenteral in accordance with the more frequent dosage schedules recommended for these dosage forms, such as 250 mg every four hours, or an initial dose of 500 mg followed by 250 mg or 125 mg every four hours, depending on the case in question.
Acute Mountain Sickness
Dosage is 500 mg to 1000 mg daily, in divided doses using tablets or extended-release capsules as appropriate. In circumstances of rapid ascent, such as in rescue or military operations, the higher dose level of 1000 mg is recommended. It is preferable to initiate dosing 24 to 48 hours before ascent and to continue for 48 hours while at high altitude, or longer as necessary to control symptoms.
CONTRAINDICATIONS:
Hypersensitivity to acetazolamide or any excipients in the formulation. Since acetazolamide is a sulfonamide derivative, cross sensitivity between acetazolamide, sulfonamides and other sulfonamide derivatives is possible.
Acetazolamide therapy is contraindicated in situations in which sodium and/or potassium blood serum levels are depressed, in cases of marked kidney and liver disease or dysfunction, in suprarenal gland failure, and in hyperchloremic acidosis. It is contraindicated in patients with cirrhosis because of the risk of development of hepatic encephalopathy.
Long-term administration of acetazolamide extended-release capsules are contraindicated in patients with chronic non-congestive angle-closure glaucoma since it may permit organic closure of the angle to occur while the worsening glaucoma is masked by lowered intraocular pressure.
ADVERSE REACTIONS:
Body as a whole:
Headache, malaise, fatigue, fever, pain at injection site, flushing, growth retardation in children, flaccid paralysis, anaphylaxis.
Digestive:
Gastrointestinal disturbances such as nausea, vomiting, diarrhea.
Hematological/Lymphatic:
Blood dyscrasias such as aplastic anemia, agranulocytosis, leukopenia, thrombocytopenic purpura, melena.
Hepato-biliary disorders:
Abnormal liver function, cholestatic jaundice, hepatic insufficiency, fulminant hepatic necrosis.
Metabolic/Nutritional:
Metabolic acidosis, electrolyte imbalance, including hypokalemia, hyponatremia, osteomalacia with long-term phenytoin therapy, loss of appetite, taste alteration, hyper/hypoglycemia.
Nervous:
Drowsiness, paresthesia (including numbness and tingling of extremities and face), depression, excitement, ataxia, confusion, convulsions dizziness.
Skin:
Allergic skin reactions including urticaria, photosensitivity, Stevens-Johnson syndrome, toxic epidermal necrolysis.
Otologic: Hearing disturbances, tinnitus.
Eye Disorders: Choroidal effusion, choroidal detachment, transient myopia. Transient myopia is the result of forward movement of the ciliary body leading to a narrowing of the angle.
Urogenital:
Crystalluria, increased risk of nephrolithiasis with long-term therapy, hematuria, glycosuria, renal failure polyuria.
Drug Interactions
Aspirin - See WARNINGS
Acetazolamide extended-release capsules modifies phenytoin metabolism with increased serum levels of phenytoin. This may increase or enhance the occurrence of osteomalacia in some patients receiving chronic phenytoin therapy. Caution is advised in patients receiving chronic concomitant therapy. By decreasing the gastrointestinal absorption of primidone, acetazolamide extended-release capsules may decrease serum concentrations of primidone and its metabolites, with a consequent possible decrease in anticonvulsant effect. Caution is advised when beginning, discontinuing, or changing the dose of acetazolamide extended-release capsules in patients receiving primidone.
Because of possible additive effects with other carbonic anhydrase inhibitors, concomitant use is not advisable.
Acetazolamide may increase the effects of other folic acid antagonists.
Acetazolamide decreases urinary excretion of amphetamine and may enhance the magnitude and duration of their effect.
Acetazolamide reduces urinary excretion of quinidine and may enhance its effect.
Acetazolamide may prevent the urinary antiseptic effect of methenamine. Acetazolamide increases lithium excretion and the lithium may be decreased.
Acetazolamide and sodium bicarbonate used concurrently increase the risk of renal calculus formation.
Acetazolamide may elevate cyclosporine levels.
DESCRIPTION:
Acetazolamide extended-release capsules are an inhibitor of the enzyme carbonic anhydrase. Acetazolamide is a white to faintly yellowish white crystalline, odorless powder, sparingly soluble in practically boiling water, very slightly soluble in water and slightly soluble in alcohol. The chemical name for acetazolamide is N-(5-Sulfamoyl-1,3,4-thiadiazol-2-yl) acetamide and has the following chemical structure: 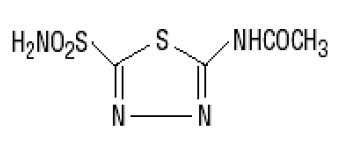 MW 222.25 C 4 H 6 N40 3 S 2 Acetazolamide are extended-release capsules, for oral administration, each containing 500 mg of acetazolamide and the following inactive ingredients: Microcrystalline cellulose, sodium lauryl sulfate and talc.
MW 222.25 C 4 H 6 N40 3 S 2 Acetazolamide are extended-release capsules, for oral administration, each containing 500 mg of acetazolamide and the following inactive ingredients: Microcrystalline cellulose, sodium lauryl sulfate and talc.
The ingredients in the capsule shell are D&C Red No. 28, D&C Yellow No. 10, FD&C Red No. 40, gelatin and titanium dioxide.
The ingredients in the imprinting ink are iron oxide black, potassium hydroxide, propylene glycol and shellac.
CLINICAL PHARMACOLOGY:
Acetazolamide is a potent carbonic anhydrase inhibitor, effective in the control of fluid secretion (e.g., some types of glaucoma), in the treatment of certain convulsive disorders (e.g., epilepsy), and in the promotion of diuresis in instances of abnormal fluid retention (e.g., cardiac edema).
Acetazolamide is not a mercurial diuretic. Rather, it is a non-bacteriostatic sulfonamide possessing a chemical structure and pharmacological activity distinctly different from the bacteriostatic sulfonamides.
Acetazolamide is an enzyme inhibitor that acts specifically on carbonic anhydrase, the enzyme that catalyzes the reversible reaction involving the hydration of carbon dioxide and the dehydration of carbonic acid. In the eye, this inhibitory action of acetazolamide decreases the secretion of aqueous humor and results in a drop in intraocular pressure, a reaction considered desirable in cases of glaucoma and even in certain non-glaucomatous conditions. Evidence seems to indicate that acetazolamide has utility as an adjuvant in treatment of certain dysfunctions of the central nervous system (e.g., epilepsy). Inhibition of carbonic anhydrase in this area appears to retard abnormal, paroxysmal, excessive discharge from central nervous system neurons. The diuretic effect of acetazolamide is due to its action in the kidney on the reversible reaction involving hydration of carbon dioxide and dehydration of carbonic acid. The result is renal loss of HCO 3 ion, which carries out sodium, water, and potassium. Alkalinization of the urine and promotion of diuresis are thus affected. Alteration in ammonia metabolism occurs due to increased reabsorption of ammonia by the renal tubules as a result of urinary alkalinization.
Acetazolamide extended-release capsules provide prolonged action to inhibit aqueous humor secretion for 18 to 24 hours after each dose, whereas tablets act for only eight to 12 hours. The prolonged continuous effect of pellets permits a reduction in dosage frequency.
Plasma concentrations of acetazolamide peak from three to six hours after administration of acetazolamide extended-release capsules, compared to one to four hours with tablets. Food does not affect bioavailability of acetazolamide extended-release capsules.
Placebo-controlled clinical trials have shown that prophylactic administration of acetazolamide at a dose of 250 mg every eight to 12 hours (or a 500 mg controlled release capsule once daily) before and during rapid ascent to altitude results in fewer and/or less severe symptoms of acute mountain sickness (AMS) such as headache, nausea, shortness of breath, dizziness, drowsiness, and fatigue. Pulmonary function (e.g., minute ventilation, expired vital capacity, and peak flow) is greater in the acetazolamide treated group, both in subjects with AMS and asymptomatic subjects. The acetazolamide treated climbers also had less difficulty in sleeping.
HOW SUPPLIED:
Acetazolamide extended-release capsules are available as:
500 mg: Orange opaque cap and orange opaque body, size ‘00’ capsule having imprinting ‘A’ on cap and ‘247’ on body with black ink, filled with white to off-white pellets.
NDC 62332-349-31 Bottle of 100 capsules
NDC 62332-349-91 Bottle of 1000 capsules
Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
You may also report side effects to Alembic Pharmaceuticals Limited at 1-866-210-9797.
Manufactured by:
Alembic Pharmaceuticals Limited
(Formulation Division),
Panelav 389350, Gujarat, India
Manufactured for:
Alembic Pharmaceuticals, Inc.
Bedminster, NJ 07921, USA
Revised: 09/2024